CRISPR/Cas9 Editing of the β-Globin Locus in Sickle Cell Disease and β-Thalassemia: Mechanistic Rationale, Clinical Evidence, and Future Directions
DOI:
https://doi.org/10.48048/tis.2026.12982Keywords:
β-thalassemia, β-globin locus, CRISPR/Cas9, fetal hemoglobin, hematopoietic stem cell gene editing, sickle cell diseaseAbstract
Sickle cell disease (SCD) and β‑thalassemia rank among the world’s most frequent single-gene disorders and remain the leading causes of illness and early death, even with better supportive care. Both conditions stem from harmful mutations in the β‑globin gene (HBB) and are prime candidates for gene-editing therapies, as fetal hemoglobin (HbF) can greatly reduce disease severity. This review first explains the molecular basis of β-hemoglobinopathies and the processes that govern Hb switching, focusing on BCL11A, HBG1/HBG2 promoters, and α-globin as therapeutic targets. This review then examined CRISPR/Cas9 techniques and delivery methods used to edit hematopoietic stem and progenitor cells outside the body, contrasting direct HBB correction with approaches that reactivate HbF and with new multiplex editing strategies. Clinical data from therapies targeting the BCL11A enhancer or the HBG promoter in patients with SCD and transfusion-dependent β‑thalassemia are reviewed, highlighting high rates of transfusion independence, marked reductions in vaso-occlusive crises, and safety primarily limited by conditioning-related toxicity. The article also discusses genomic risks, ethical concerns, cost, and access challenges, and suggests future avenues, such as base and prime editing, in vivo delivery, and safer conditioning regimens. In summary, CRISPR-based editing of the β-globin locus offers a realistic functional cure for selected patients, while underscoring the need for long-term follow-up and fair global implementation.
HIGHLIGHTS
- The molecular basis of β-hemoglobinopathies and identifies key regulatory targets in the β-globin locus for CRISPR/Cas9 therapy.
- Major CRISPR/Cas9 strategies, including HBB correction, BCL11A enhancer disruption, HBG1/HBG2 promoter editing, and α-globin modulation, in the context of sickle cell disease and β-thalassemia.
- Integrating and critically evaluating clinical trial data for ex vivo edited hematopoietic stem cell products, highlighting transfusion independence, vaso-occlusive crisis reduction, and emerging safety signals.
- Genomic, ethical, and health-system challenges, including off-target risk, conditioning-related toxicity, cost, and global equity of access to gene-editing therapies, are analyzed.
- Outlines future directions, such as base and prime editing, in vivo delivery platforms, and safer conditioning regimens, to guide next-generation therapies for β-hemoglobinopathies.
GRAPHICAL ABSTRACT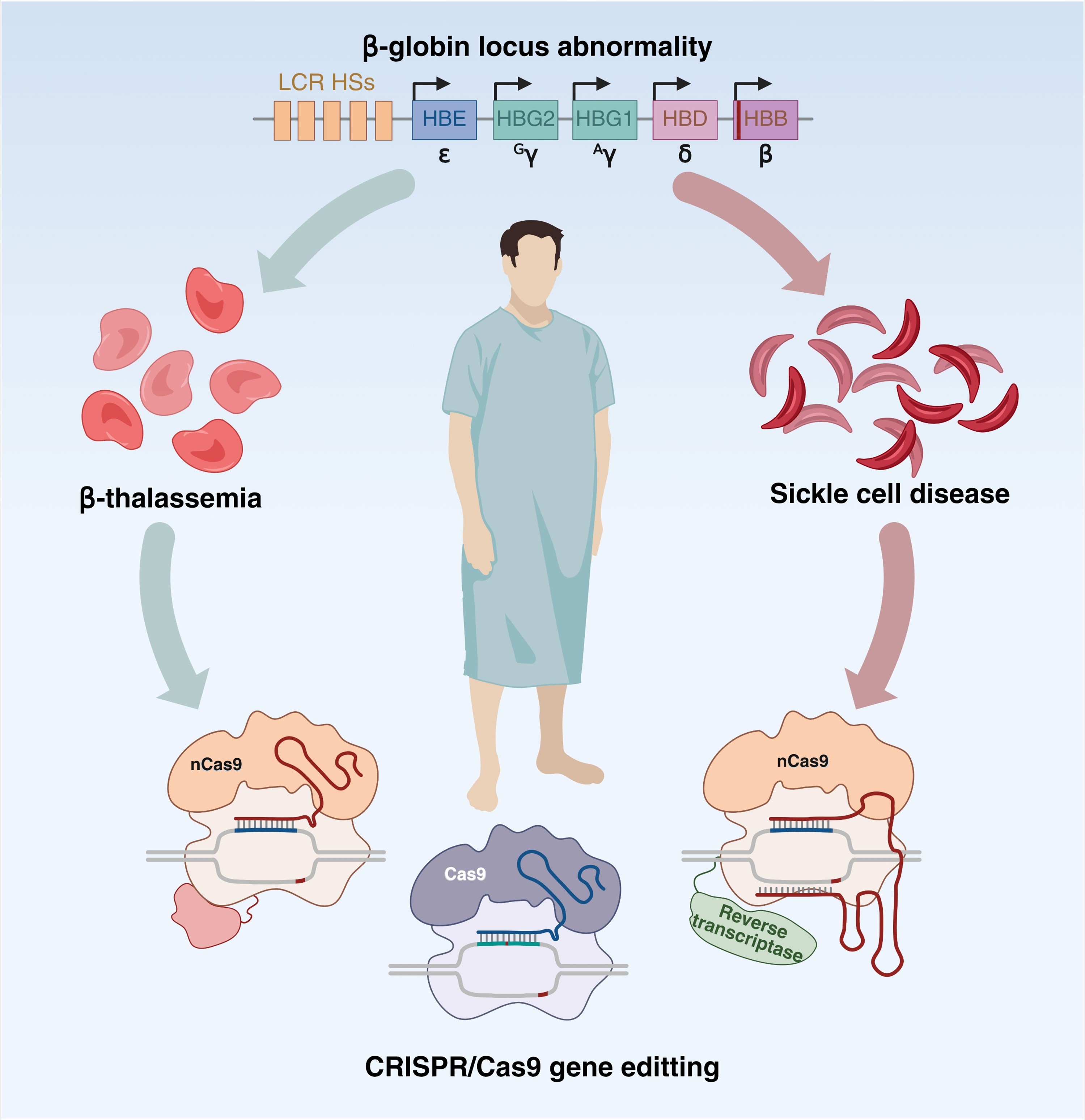
Downloads
References
M Angastiniotis and S Lobitz. Thalassemias: An overview. International Journal of Neonatal Screening 2019; 5(1), 16.
GJ Kato, FB Piel, CD Reid, MH Gaston, K Ohene-Frempong, L Krishnamurti, WR Smith, JA Panepinto, DJ Weatherall, FF Costa and EP Vichinsky. Sickle cell disease. Nature Reviews Disease Primers 2018; 4(1), 18010.
FB Piel, DC Rees, MR DeBaun, O Nnodu, B Ranque, AA Thompson, RE Ware, MR Abboud, A Abraham, EE Ambrose, B Andemariam, R Colah, R Colombatti, N Conran, FF Costa, RM Cronin, M de Montalembert, J Elion, E Esrick, AL Greenway, IM Idris, DZ Issom, D Jain, …, K Ohene-Frempong. Defining global strategies to improve outcomes in sickle cell disease: A lancet haematology commission. The Lancet Haematology 2023; 10(8), e633-e686.
A Kattamis, GL Forni, Y Aydinok and V Viprakasit. Changing patterns in the epidemiology of β-thalassemia. European Journal of Haematology 2020; 105, 692-703.
MC Walters, M Patience, W Leisenring, JR Eckman, JP Scott, WC Mentzer, SC Davies, K Ohene-Frempong, F Bernaudin, DC Matthews, R Storb and KM Sullivan. Bone marrow transplantation for sickle cell disease. New England Journal of Medicine 1996; 335(6), 369-376.
M Cavazzana, C Antoniani and A Miccio. Gene therapy for β-hemoglobinopathies. Molecular Therapy 2017; 25(5), 1142-1154.
EB Esrick, LE Lehmann, A Biffi, M Achebe, C Brendel, MF Ciuculescu, H Daley, B MacKinnon, E Morris, A Federico, D Abriss, K Boardman, R Khelladi, K Shaw, H Negre, O Negre, S Nikiforow, J Ritz, SY Pai, …, DA Williams. Post-transcriptional genetic silencing of BCL11A to treat sickle cell disease. New England Journal of Medicine 2021; 384(3), 205-215.
ADH Nguyen and MT Quang. CRISPR/Cas9 genome editing in oncology: Mechanisms, therapeutic platforms and translational challenges. Molecular Biotechnology 2025. https://doi.org/10.1007/s12033-025-01533-2
JB Papizan, SN Porter, A Sharma and SM Pruett-Miller. Therapeutic gene editing strategies using CRISPR-Cas9 for the β-hemoglobinopathies. Journal of Biomedical Research 2020; 35(2), 115.
H Frangoul, D Altshuler, MD Cappellini, YS Chen, J Domm, BK Eustace, J Foell, J de la Fuente, S Grupp, R Handgretinger, TW Ho, A Kattamis, A Kernytsky, J Lekstrom-Himes, AM Li, F Locatelli, MY Mapara, M de Montalembert, D Rondelli, …, S Corbacioglu. CRISPR-Cas9 gene editing for sickle cell disease and β-thalassemia. New England Journal of Medicine 2021; 384(3), 252-260.
B Fu, J Liao, S Chen, W Li, Q Wang, J Hu, F Yang, S Hsiao, Y Jiang, L Wang, F Chen, Y Zhang, X Wang, D Li, M Liu and Y Wu. CRISPR-Cas9-mediated gene editing of the BCL11A enhancer for pediatric β⁰/β⁰ transfusion-dependent β-thalassemia. Nature Medicine 2022; 28(8), 1573-1580.
A Quagliano, D Acevedo, P Hardigan and S Prasad. Using Clustered Regularly Interspaced Short Palindromic Repeats gene editing to induce permanent expression of fetal hemoglobin in β-thalassemia and sickle cell disease: A comparative meta-analysis. Frontiers in Medicine 2022; 9, 943631.
DV Parums. Editorial: First regulatory approvals for CRISPR-Cas9 therapeutic gene editing for sickle cell disease and transfusion-dependent β-thalassemia. Medical Science Monitor 2024; 30, e944204.
EY Adashi, PA Gruppuso and IG Cohen. CRISPR therapy of sickle cell disease: The dawning of the gene editing era. The American Journal of Medicine 2024; 137(5), 390-392.
VG Sankaran and SH Orkin. The switch from fetal to adult hemoglobin. Cold Spring Harbor Perspectives in Medicine 2013; 3(1), a011643.
E Khandros and GA Blobel. Elevating fetal hemoglobin: recently discovered regulators and mechanisms. Blood 2024; 144(8), 845-852.
DE Bauer and SH Orkin. Hemoglobin switching’s surprise: The versatile transcription factor BCL11A is a master repressor of fetal hemoglobin. Current Opinion in Genetics & Development 2015; 33, 62-70.
N Liu, VV Hargreaves, Q Zhu, JV Kurland, J Hong, W Kim and SH Orkin. Direct promoter repression by BCL11A controls the fetal to adult hemoglobin switch. Cell 2018; 173(2), 430-442.
S Rivella. Iron metabolism under conditions of ineffective erythropoiesis in β-thalassemia. Blood 2019; 133(1), 51-58.
F Longo, A Piolatto, GB Ferrero and A Piga. Ineffective erythropoiesis in β-thalassemia: Key steps and therapeutic options. International Journal of Molecular Sciences 2021; 22(13), 7229.
M Jinek, K Chylinski, I Fonfara, M Hauer, JA Doudna and E Charpentier. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012; 337(6096), 816-821.
DP Dever, RO Bak, A Reinisch, J Camarena, G Washington, CE Nicolas, M Pavel-Dinu, N Saxena, AB Wilkens, S Mantri, N Uchida, A Hendel, A Narla, R Majeti, KI Weinberg and MH Porteus. CRISPR/Cas9 β-globin gene targeting in human hematopoietic stem cells. Nature 2016; 539(7629), 384-389.
MD Hoban, D Lumaquin, CY Kuo, Z Romero, J Long, M Ho, CS Young, M Mojadidi, S Fitz-Gibbon, AR Cooper, GR Lill, F Urbinati, B Campo-Fernandez, CF Bjurstrom, M Pellegrini, RP Hollis and DB Kohn. CRISPR/Cas9-mediated correction of the sickle mutation in human CD34⁺ cells. Molecular Therapy 2016; 24(9), 1561-1569.
M Kosicki, K Tomberg and A Bradley. Repair of double-strand breaks induced by CRISPR-Cas9 leads to large deletions and complex rearrangements. Nature Biotechnology 2018; 36(8), 765-771.
J Tycko, VE Myer and PD Hsu. Methods for optimizing CRISPR-Cas9 genome editing specificity. Molecular Cell 2016; 63(3), 355-370.
RO Bak, DP Dever and MH Porteus. CRISPR/Cas9 genome editing in human hematopoietic stem cells. Nature Protocols 2018; 13(2), 358-376.
CT Charlesworth, PS Deshpande, DP Dever, J Camarena, VT Lemgart, MK Cromer, CA Vakulskas, MA Collingwood, L Zhang, NM Bode, MA Behlke, B Dejene, B Cieniewicz, R Romano, BJ Lesch, N Gomez-Ospina, S Mantri, M Pavel-Dinu, KI Weinberg and MH Porteus. Identification of preexisting adaptive immunity to Cas9 proteins in humans. Nature Medicine 2019; 25(2), 249-254.
OM Enache, V Rendo, M Abdusamad, D Lam, D Davison, S Pal, N Currimjee, J Hess, S Pantel, A Nag, AR Thorner, JG Doench, F Vazquez, R Beroukhim, TR Golub and U Ben-David. Cas9 activates the p53 pathway and selects for p53-inactivating mutations. Nature Genetics 2020; 52(7), 662-668.
MT Quang and MN Nguyen. The role and regulation of cell death in cancer. Progress in Molecular Biology and Translational Science 2025; 217, 135-161.
AC Komor, YB Kim, MS Packer, JA Zuris and DR Liu. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 2016; 533(7603), 420-424.
J Zeng, Y Wu, C Ren, J Bonanno, AH Shen, D Shea, JM Gehrke, K Clement, K Luk, Q Yao, R Kim, SA Wolfe, JP Manis, L Pinello, JK Joung and DE Bauer. Therapeutic base editing of human hematopoietic stem cells. Nature Medicine 2020; 26(4), 535-541.
LC Cosenza, J Gasparello, N Romanini, M Zurlo, C Zuccato, R Gambari and A Finotti. Efficient CRISPR-Cas9-based genome editing of the β-globin gene in erythroid cells from β⁰³⁹-thalassemia patients. Molecular Therapy - Methods & Clinical Development 2021; 21, 507-523.
S Mettananda, CA Fisher, D Hay, M Badat, L Quek, K Clark, P Hublitz, D Downes, J Kerry, M Gosden, J Telenius, JA Sloane-Stanley, P Faustino, A Coelho, J Doondeea, B Usukhbayar, P Sopp, JA Sharpe, JR Hughes, ..., DR Higgs. Editing an α-globin enhancer in primary human hematopoietic stem cells as a treatment for β-thalassemia. Nature Communications 2017; 8(1), 424.
MC Canver, EC Smith, F Sher, L Pinello, NE Sanjana, O Shalem, DD Chen, PG Schupp, DS Vinjamur, SP Garcia, S Luc, R Kurita, Y Nakamura, Y Fujiwara, T Maeda, GC Yuan, F Zhang, SH Orkin and DE Bauer. BCL11A enhancer dissection by Cas9-mediated in situ saturating mutagenesis. Nature 2015; 527(7577), 192-197.
G Pavani, A Fabiano, M Laurent, F Amor, E Cantelli, A Chalumeau, G Maule, A Tachtsidi, JP Concordet, A Cereseto, F Mavilio, G Ferrari, A Miccio and M Amendola. Correction of β-thalassemia by CRISPR/Cas9 editing of the α-globin locus in human hematopoietic stem cells. Blood Advances 2021; 5(5), 1137-1153.
C Antoniani, V Meneghini, A Lattanzi, T Felix, O Romano, E Magrin, L Weber, G Pavani, S El Hoss, R Kurita, Y Nakamura, TJ Cradick, AS Lundberg, M Porteus, M Amendola, W El Nemer, M Cavazzana, F Mavilio and A Miccio. Induction of fetal hemoglobin synthesis by CRISPR/Cas9-mediated editing of the human β-globin locus. Blood 2018; 131(17), 1960-1973
N Psatha, A Reik, S Phelps, Y Zhou, D Dalas, E Yannaki, DN Levasseur, FD Urnov, MC Holmes and T Papayannopoulou. Disruption of the BCL11A erythroid enhancer reactivates fetal hemoglobin in β-thalassemia. Molecular Therapy - Methods & Clinical Development 2018; 10, 313-326.
L Weber, G Frati, T Felix, G Hardouin, A Casini, C Wollenschlaeger, V Meneghini, C Masson, A De Cian, A Chalumeau, F Mavilio, M Amendola, I Andre-Schmutz, A Cereseto, W El Nemer, JP Concordet, C Giovannangeli, M Cavazzana and A Miccio. Editing a γ-globin repressor binding site restores fetal hemoglobin synthesis and corrects the sickle cell disease phenotype. Science Advances 2020; 6(7), eaay9392.
EA Traxler, Y Yao, YD Wang, KJ Woodard, R Kurita, Y Nakamura, JR Hughes, RC Hardison, GA Blobel, C Li and MJ Weiss. A genome-editing strategy to treat β-hemoglobinopathies that recapitulates a benign genetic condition. Nature Medicine 2016; 22(9), 987-990.
D Lu, Z Xu, Z Peng, Y Yang, B Song, Z Xiong, Z Ma, H Guan, B Chen, Y Nakamura, J Zeng, N Liu, X Sun and D Chen. Induction of fetal hemoglobin by introducing natural hereditary persistence of fetal hemoglobin mutations in the γ-globin gene promoters for genome editing therapies for β-thalassemia. Frontiers in Genetics 2022; 13, 881937.
A Sharma, JJ Boelens, M Cancio, JS Hankins, P Bhad, M Azizy, A Lewandowski, X Zhao, S Chitnis, R Peddinti, Y Zheng, N Kapoor, F Ciceri, T Maclachlan, Y Yang, Y Liu, J Yuan, U Naumann, VWC Yu, …, JL LaBelle. CRISPR-Cas9 editing of the HBG1 and HBG2 promoters to treat sickle cell disease. New England Journal of Medicine 2023; 389(9), 820-832.
NS Ravi, B Wienert, SK Wyman, HW Bell, A George, G Mahalingam, JT Vu, K Prasad, BP Bandlamudi and KM Mohankumar. Identification of novel HPFH-like mutations by CRISPR base editing that elevate the expression of fetal hemoglobin. eLife 2022; 11, e65421.
L Wang, H Xu, J Liang, Y Li, L Shi, B Xiao, J Huang, X Xu and X Zhang. Initial safety and efficacy study of RM-001 autologous HBG1/2 promoter-modified CD34⁺ cells in transfusion-dependent β-thalassemia. HemaSphere 2022; 6, 1347-1348.
R Liu, L Wang, H Xu, X Yin, J Liang, W Xie, G Yang, Y Li, Y Zhou, L Shi, B Xiao, L Shi, Z Shi, X Zhou, J Fang, X Xu, Y Lai, J Huang and X Zhang. Safety and efficacy of RM-001 in transfusion-dependent β-thalassemia: Early results. HemaSphere 2023; 7(S3), e613965e.
M Laurent, M Geoffroy, G Pavani and S Guiraud. CRISPR-based gene therapies: From preclinical to clinical treatments. Cells 2024; 13(10), 800.
L Li and PK Mandal. Recent. Recent advancements in gene therapy for sickle cell disease and β-thalassemia. Frontiers in Hematology 2024; 3, 1468952.
A Aprile, MR Lidonnici and G Ferrari. Gene therapy for hemoglobinopathies: Clinical trial results and biology of hematopoietic stem cell and the bone marrow niche. Cell Reports Medicine 2025; 6(12), 102419.
H Frangoul, F Locatelli, A Sharma, M Bhatia, M Mapara, L Molinari, D Wall, RI Liem, P Telfer, AJ Shah, M Cavazzana, S Corbacioglu, D Rondelli, R Meisel, L Dedeken, S Lobitz, M de Montalembert, MH Steinberg, MC Walters, …, SA Grupp. Exagamglogene autotemcel for severe sickle cell disease. New England Journal of Medicine 2024; 390(18), 1649-1662.
F Locatelli, P Lang, D Wall, R Meisel, S Corbacioglu, AM Li, J de la Fuente, AJ Shah, B Carpenter, JL Kwiatkowski, M Mapara, RI Liem, MD Cappellini, M Algeri, A Kattamis, S Sheth, S Grupp, R Handgretinger, P Kohli, ..., H Frangoul. Exagamglogene autotemcel for transfusion-dependent β -thalassemia. New England Journal of Medicine 2024; 390(18), 1663-1676.
R Handgretinger and M Mezger. An evaluation of exagamglogene autotemcel for the treatment of sickle cell disease and transfusion-dependent beta-thalassaemia. Expert Opinion on Biological Therapy 2024; 24(9), 883-888.
JD Finn, AR Smith, MC Patel, L Shaw, MR Youniss, J van Heteren, T Dirstine, C Ciullo, R Lescarbeau, J Seitzer, RR Shah, A Shah, D Ling, J Growe, M Pink, E Rohde, KM Wood, WE Salomon, WF Harrington, ..., DV Morrissey. A single administration of CRISPR/Cas9 lipid nanoparticles achieves robust and persistent in vivo genome editing. Cell Reports 2018; 22(9), 2227-2235.
JD Gillmore, E Gane, J Taubel, J Kao, M Fontana, ML Maitland, J Seitzer, D O’Connell, KR Walsh, K Wood, J Phillips, Y Xu, A Amaral, AP Boyd, JE Cehelsky, MD McKee, A Schiermeier, O Harari, A Murphy, ..., D Lebwohl. CRISPR-Cas9 in vivo gene editing for transthyretin amyloidosis. New England Journal of Medicine 2021; 385(6), 493-502.
B Fu, X Zhang, L Wang, J Liao, S Chen, B Zheng, W Li, F Wang, D Li, M Liu and Y Wu. Updated follow-up of BRL-101 CRISPR-Cas9-mediated BCL11A enhancer editing for transfusion-dependent β-thalassemia. HemaSphere 2023; 7(S3), e406095b.
A Czechowicz, R Palchaudhuri, A Scheck, Y Hu, J Hoggatt, B Saez, WW Pang, MK Mansour, TA Tate, YY Chan, E Walck, G Wernig, JA Shizuru, F Winau, DT Scadden and DJ Rossi. Selective hematopoietic stem cell ablation using CD117-antibody-drug-conjugates enables safe and effective transplantation with immunity preservation. Nature Communications 2019; 10(1), 617.
TTT Huynh, TN Nguyen, TA Nguyen, ADH Nguyen and MT Quang. Convergence of gene therapy and vaccine platforms in the post-pandemic era: A mini review. Trends in Sciences 2026; 23(3), 12197.
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






