Convergence of Gene Therapy and Vaccine Platforms in the Post-Pandemic Era: A Mini Review
DOI:
https://doi.org/10.48048/tis.2026.12197Keywords:
Gene therapy, mRNA vaccines, Adenoviral vectors, Platform convergence, Post-pandemic therapeutics, Translational medicine, Gene therapy, mRNA vaccines, Adenoviral vectors, Platform convergence, Post-pandemic therapeutics, Translational medicineAbstract
The COVID-19 pandemic accelerated the convergence of vaccine and gene therapy research, demonstrating the potential for gene-encoded platforms to be rapidly reconfigured for both preventive and therapeutic applications. This real-world evaluation validated the principle of adaptable, platform-based molecular medicines. Convergence is facilitated by three key components: Chemically modified nucleotides that reduce innate-immune sensing while maintaining high translation efficiency, lipid nanoparticles refined for targeted and low-toxicity delivery, and viral vectors with adjustable immunogenicity. Emerging technologies, including self-amplifying RNA, selectively targeted lipid nanoparticles, and advanced adenoviruses, have expanded the therapeutic toolkit to include in vivo genome editing and personalized neoantigen vaccines. The rapid scale-up of production during the pandemic has had a lasting impact, establishing cGMP manufacturing lines, emergency-use regulatory frameworks, and extensive safety data. However, the field still faces challenges such as context-dependent immunogenicity, the need for thermostable products to improve global distribution, and widening disparities in technology access between high- and low-income regions. Gene-encoded platforms have evolved from a proof-of-concept to a multifaceted therapeutic framework capable of addressing infectious, genetic, and malignant diseases. Their full public health potential will depend on coordinated investment in distributed manufacturing, adaptive regulation, and ethical governance that prioritizes equity alongside technical advancements.
HIGHLIGHTS
- The COVID-19 pandemic served as a major catalyst, accelerating the convergence of gene therapy and vaccine development into a unified field of adaptable, gene-encoded molecular medicines.
- A bidirectional flow of innovation is detailed, where foundational gene therapy research (e.g., modified nucleotides, lipid nanoparticles) enabled rapid vaccine success, which in turn established manufacturing and regulatory precedents that now benefit therapeutic development.
- The validation of mRNA and adenoviral vector platforms is expanding the therapeutic toolkit, driving progress in protein replacement therapies, in vivo genome editing, and personalized cancer immunotherapy.
- Future progress depends on overcoming shared challenges, including managing context-dependent immunogenicity, ensuring thermostability for global distribution, and establishing equitable access through distributed manufacturing and adaptive governance.
GRAPHICAL ABSTRACT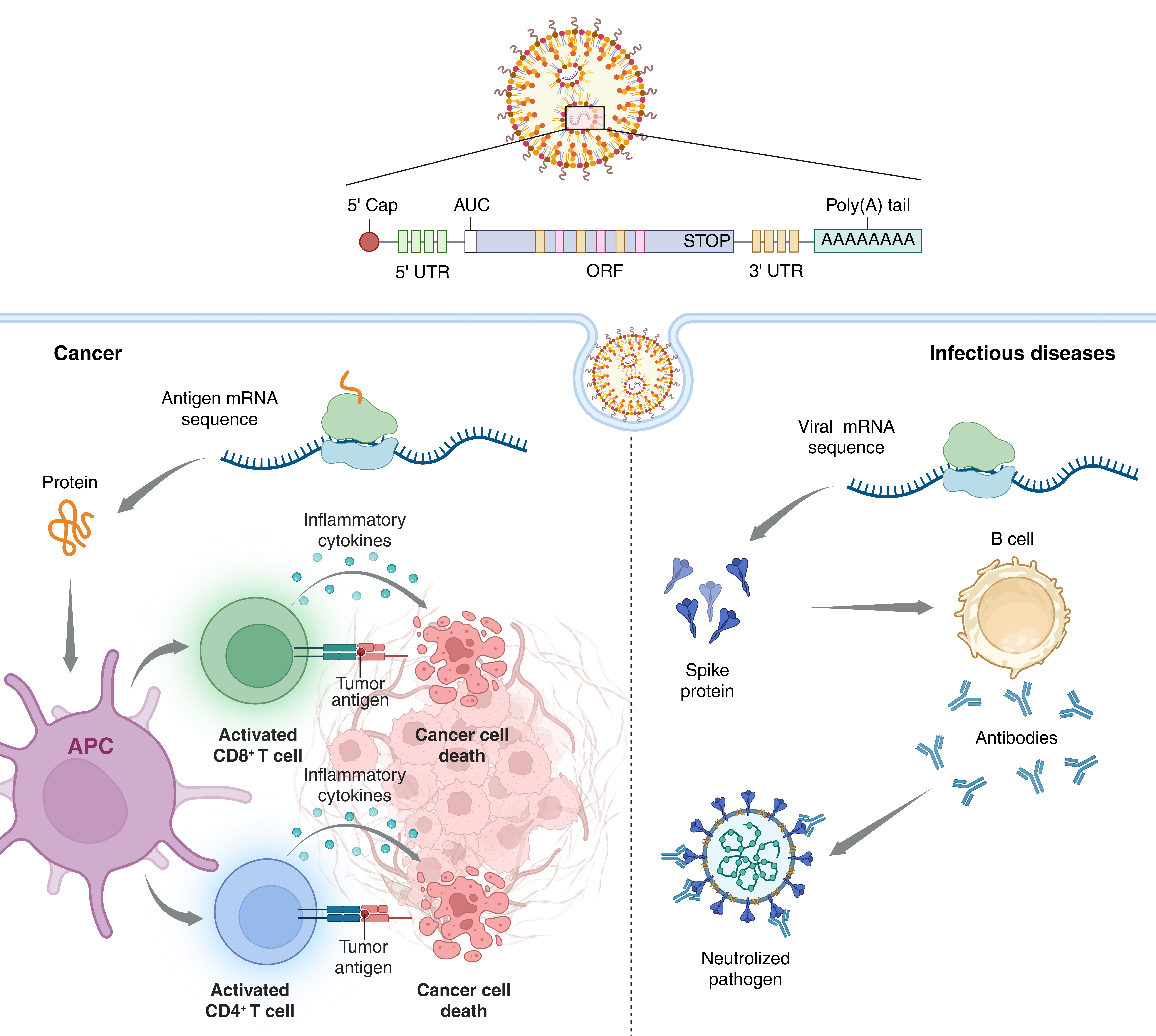
Downloads
References
J Tang, MA Amin and JL Campian. Past, present, and future of viral vector vaccine platforms: A comprehensive review. Vaccines 2025; 13(5), 524.
X Hou, T Zaks, R Langer and Y Dong. Lipid nanoparticles for mRNA delivery. Nature Reviews Materials 2021; 6(12), 1078-1094.
H Parhiz, EN Atochina-Vasserman and D Weissman. mRNA-based therapeutics: Looking beyond COVID-19 vaccines. The Lancet 2024; 403(10432), 1192-1204.
K Bloom, F van den Berg and P Arbuthnot. Self-amplifying RNA vaccines for infectious diseases. Gene Therapy 2021; 28(3), 117-129.
Y Shi, M Shi, Wang Y and J You. Progress and prospects of mRNA-based drugs in pre-clinical and clinical applications. Signal Transduction and Targeted Therapy 2024; 9(1), 322.
W Abu El Kheir-Mataria, Z Khadr, H El Fawal and S Chun. COVID-19 vaccine intercountry distribution inequality and its underlying factors: A combined concentration index analysis and multiple linear regression analysis. Frontiers in Public Health 2024; 12, 1348088.
K Karikó. Modified uridines are the key to a successful message. Nature Reviews Immunology 2021; 21(10), 619-619.
R Ostad Ali Dehaghi, A Khadem Broojerdi, A Magdy, M Valentin, J Dahlan, O Malik, RH Siggers, E Nkansah and HB Sillo. Strengthening vaccine regulation: Insights from COVID-19 vaccines, best practices, and lessons for future public health emergencies. Vaccines 2025; 13(6), 638.
M Slaoui and M Hepburn. Developing safe and effective covid vaccines - operation warp speed’s strategy and approach. New England Journal of Medicine 2020; 383(18), 1701-1703.
HS Lalani, S Nagar, A Sarpatwari, RE Barenie, J Avorn, BN Rome and AS Kesselheim. US public investment in development of mRNA covid-19 vaccines: Retrospective cohort study. BMJ 2023; 380, e073747.
A Talbot, TF de Koning-Ward and D Layton. Left out in the cold - inequity in infectious disease control due to cold chain disparity. Vaccine 2025; 45, 126648.
SM Iqbal, AM Rosen, D Edwards, A Bolio, HJ Larson, M Servin, M Rudowitz, A Carfi and F Ceddia. Opportunities and challenges to implementing mRNA-based vaccines and medicines: lessons from COVID-19. Frontiers in Public Health 2024; 12, 1429265.
R Karam, K Iskandar, M Watfa and A Zeitoun. Serious adverse events following immunization with COVID-19 vaccines in Lebanon: a retrospective analysis of the National Pharmacovigilance Database. BMC Public Health 2024; 24(1), 2905.
FP Polack, SJ Thomas, N Kitchin, J Absalon, A Gurtman, S Lockhart, JL Perez, G Pérez Marc, ED Moreira, C Zerbini and R Bailey. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. New England journal of medicine 2020; 383(27), 2603-2615.
LR Baden, HM El Sahly, B Essink, K Kotloff, S Frey, R Novak, D Diemert, SA Spector, N Rouphael, CB Creech and J McGettigan. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. New England Journal of Medicine 2021; 384(5), 403-416.
M Voysey, SAC Clemens, SA Madhi, LY Weckx, PM Folegatti, PK Aley, B Angus, VL Baillie, SL Barnabas, QE Bhorat, S Bibi, C Briner, P Cicconi, AM Collins, R Colin-Jones, CL Cutland, TC Darton, K Dheda, CJA Duncan, …, Oxford COVID Vaccine Trial Group. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. The Lancet 2021; 397(10269), 99-111.
J Sadoff, G Gray, A Vandebosch, V Cárdenas, G Shukarev, B Grinsztejn, PA Goepfert, C Truyers, H Fennema, B Spiessens, K Offergeld, G Scheper, KL Taylor, ML Robb, J Treanor, DH Barouch, J Stoddard, MF Ryser, MA Marovich, …, Ensemble Study Group. Safety and efficacy of single-dose Ad26.COV2.S vaccine against covid-19. New England Journal of Medicine 2021; 384(23), 2187-2201.
SH Ko, P Radecki, F Belinky, JN Bhiman, S Meiring, J Kleynhans, D Amoako, V Guerra Canedo, M Lucas, D Kekana, N Martinson, L Lebina, J Everatt, S Tempia, T Bylund, R Rawi, PD Kwong, N Wolter, A von Gottberg, C Cohen and EA Boritz. Rapid intra-host diversification and evolution of SARS-CoV-2 in advanced HIV infection. Nature Communications 2024; 15(1), 7240.
S Kochhar, HS Izurieta, RE Chandler, A Hacker, RT Chen and B Levitan. Benefit-risk assessment of vaccines. Vaccine 2024; 42(4), 969-971.
K Faksova, D Walsh, Y Jiang, J Griffin, A Phillips, A Gentile, JC Kwong, K Macartney, M Naus, Z Grange, S Escolano, G Sepulveda, A Shetty, A Pillsbury, C Sullivan, Z Naveed, NZ Janjua, N Giglio, J Perälä, …, A Hviid. COVID-19 vaccines and adverse events of special interest: A multinational Global Vaccine Data Network (GVDN) cohort study of 99 million vaccinated individuals. Vaccine 2024; 42(9), 2200-2211.
CP Bown and TJ Bollyky. How COVID-19 vaccine supply chains emerged in the midst of a pandemic. World Economics 2022; 45(2), 468-522.
E Nourafkan, Z Yang, M Maamra and Z Kis. Advancing continuous encapsulation and purification of mRNA vaccines and therapeutics. European Journal of Pharmaceutical Sciences 2025; 212, 107183.
S Zilberzwige-Tal, A Levin, A Ezra, U Elia, G Finkelstein-Zuta, T Kreiser, L Gershon, M Goldsmith, E Kon, Y Navon, O Cohen, TP Knowles, D Peer and E Gazit. On-chip de novo production of mRNA vaccine in lipid nanoparticles. Small 2025; 21(32), e2500114.
A Azizi, D Ogbeni, G Kamuyu, LM Schwartz, C Clark, P Spencer and V Bernasconi. Standardized immunological assays for assessing COVID-19 vaccines by the CEPI-Centralized Laboratory Network. npj Vaccines 2024; 9(1), 128.
KVS Hodel, BSD Fiuza, RS Conceição, ACM Aleluia, TN Pitanga, LMDS Fonseca, CO Valente, CS Minafra-Rezende and BAS Machado. Pharmacovigilance in vaccines: Importance, main aspects, perspectives, and challenges - a narrative review. Pharmaceuticals 2024; 17(6), 807.
PA Krieg and DA Melton. Functional messenger RNAs are produced by SP6 in vitro transcription of cloned cDNAs. Nucleic Acids Research 1984; 12(18), 7057-7070.
JA Wolff, RW Malone, P Williams, W Chong, G Acsadi, A Jani and PL Felgner. Direct gene transfer into mouse muscle in vivo. Science 1990; 27(4949), 1465-1468.
JM Henderson, A Ujita, E Hill, S Yousif-Rosales, C Smith, N Ko, T McReynolds, CR Cabral, JR Escamilla-Powers and ME Houston. Cap 1 messenger RNA synthesis with co-transcriptional CleanCap® analog by in vitro transcription. Current Protocols 2021; 1(2), e39.
E Rohner, R Yang, KS Foo, A Goedel and KR Chien. Unlocking the promise of mRNA therapeutics. Nature Biotechnology 2022; 40(11), 1586-1600.
H Zhang, L Zhang, A Lin, C Xu, Z Li, K Liu, B Liu, X Ma, F Zhao, H Jiang, C Chen, H Shen, H Li, DH Mathews, Y Zhang and L Huang. Algorithm for optimized mRNA design improves stability and immunogenicity. Nature 2023; 621(7978), 396-403.
S Castillo-Hair, S Fedak, B Wang, J Linder, K Havens, M Certo and G Seelig. Optimizing 5’UTRs for mRNA-delivered gene editing using deep learning. Nature Communications 2024; 15(1), 5284.
K Swetha, NG Kotla, L Tunki, A Jayaraj, SK Bhargava, H Hu, SR Bonam and R Kurapati. Recent advances in the lipid nanoparticle-mediated delivery of mRNA vaccines. Vaccines 2023; 11(3), 658.
MS Padilla, K Mrksich, Y Wang, RM Haley, JJ Li, EL Han, R El-Mayta, EH Kim, S Dias, N Gong, SV Teerdhala, X Han, V Chowdhary, L Xue, Z Siddiqui, HM Yamagata, D Kim, IC Yoon, JM Wilson, R Radhakrishnan and MJ Mitchell. Branched endosomal disruptor (BEND) lipids mediate delivery of mRNA and CRISPR-Cas9 ribonucleoprotein complex for hepatic gene editing and T cell engineering. Nature Communications 2025; 16(1), 996.
K Karikó, M Buckstein, H Ni and D Weissman. Suppression of RNA recognition by Toll-like receptors: The impact of nucleoside modification and the evolutionary origin of RNA. Immunity 2005; 23(2), 165-175.
YV Svitkin, YM Cheng, T Chakraborty, V Presnyak, M John and N Sonenberg. N1-methyl-pseudouridine in mRNA enhances translation through eIF2α-dependent and independent mechanisms by increasing ribosome density. Nucleic Acids Research 2017; 45(10), 6023-6036.
JM Bergelson. Receptors mediating adenovirus attachment and internalization. Biochemical Pharmacology 1999; 57(9), 975-979.
G Miller, S Lahrs, VG Pillarisetty, AB Shah and RP DeMatteo. Adenovirus infection enhances dendritic cell immunostimulatory properties and induces natural killer and T-cell-mediated tumor protection. Cancer Research 2002; 62(18), 5260-5266.
N Pardi, MJ Hogan, FW Porter and D Weissman. mRNA vaccines - a new era in vaccinology. Nature Reviews Drug Discovery 2018; 17(4), 261-279.
M Aldén, F Olofsson Falla, D Yang, M Barghouth, C Luan, M Rasmussen and Y De Marinis. Intracellular Reverse Transcription of Pfizer BioNTech COVID-19 mRNA Vaccine BNT162b2 In Vitro in Human Liver Cell Line. Current Issues in Molecular Biology 2022; 44(3), 1115-1126.
S Jt, H M, B Wam, B Ac and N Sa. Adenoviral vectors for cardiovascular gene therapy applications: a clinical and industry perspective. Journal of Molecular Medicine (Berl) 2022; 100(6), 875-901.
U Kafle, HQ Truong, CT Nguyen and F Meng. Development of thermally stable mRNA-LNP delivery systems: Current Progress and Future Prospects. Molecular Pharmaceutics 2024; 21(12), 5944-5959.
L Ai, Y Li, L Zhou, W Yao, H Zhang, Z Hu, J Han, W Wang, J Wu, P Xu and R Wang. Lyophilized mRNA-lipid nanoparticle vaccines with long-term stability and high antigenicity against SARS-CoV-2. Cell Discovery 2023; 9(1), 9.
X Wang, S Liu, Y Sun, X Yu, SM Lee, Q Cheng, T Wei, J Gong, J Robinson, D Zhang and X Lian. Preparation of selective organ-targeting (SORT) lipid nanoparticles (LNPs) using multiple technical methods for tissue-specific mRNA delivery. Nature Protocols 2023; 18(1), 265-291.
Y Sun, S Chatterjee, X Lian, Z Traylor, SR Sattiraju, Y Xiao, SA Dilliard, YC Sung, M Kim, SM Lee and S Moore. In vivo editing of lung stem cells for durable gene correction in mice. Science 2024; 384(6701), 1196-1202.
T Vallet and M Vignuzzi. Self-amplifying RNA: advantages and challenges of a versatile platform for vaccine development. Viruses 2025; 17(4), 566.
JG Low, R De Alwis, S Chen, S Kalimuddin, YS Leong, TK Mah, N Yuen, HC Tan, SL Zhang, JX Sim and YF Chan. A phase I/II randomized, double-blinded, placebo-controlled trial of a self-amplifying Covid-19 mRNA vaccine. npj Vaccines 2022; 7(1), 161.
X Huang, C Liu, SN Sharma, X You, S Chen, Y Li, HJ Liu, B Liu, Q Saiding, W Chen and Y Lee. Oral delivery of liquid mRNA therapeutics by an engineered capsule for treatment of preclinical intestinal disease. Science Translational Medicine 2025; 17(807), eadu1493.
LA Rojas, Z Sethna, KC Soares, C Olcese, N Pang, E Patterson, J Lihm, N Ceglia, P Guasp, A Chu and R Yu. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature 2023; 618(7963), 144-150.
S Liu, Y Wen, X Shan, X Ma, C Yang, X Cheng, Y Zhao, J Li, S Mi, H Huo and W Li. Charge-assisted stabilization of lipid nanoparticles enables inhaled mRNA delivery for mucosal vaccination. Nature Communications 2024; 15(1), 9471.
A Vander Straeten, M Sarmadi, JL Daristotle, M Kanelli, LH Tostanoski, J Collins, A Pardeshi, J Han, D Varshney, B Eshaghi and J Garcia. A microneedle vaccine printer for thermostable COVID-19 mRNA vaccines. Nature Biotechnology 2024; 42(3), 510-517.
PJ Schürmann, SP van Breda Vriesman, JA Castro‐Alpízar, SA Kooijmans, EE Nieuwenhuis, RM Schiffelers and SA Fuchs. Therapeutic application of mRNA for genetic diseases. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology 2025; 17(3), e70019.
D Koeberl, A Schulze, N Sondheimer, GS Lipshutz, T Geberhiwot, L Li, R Saini, J Luo, V Sikirica, L Jin and M Liang. Interim analyses of a first-in-human phase 1/2 mRNA trial for propionic acidaemia. Nature 2024; 628(8009), 872-877.
A Fontanellas, P Berraondo, F Urigo, D Jericó, PG Martini, F Pastor and MA Avila. RNA-based therapies in liver metabolic diseases. Gut 2025; 74, 1514-1527.
SM Rowe, JB Zuckerman, D Dorgan, J Lascano, K McCoy, M Jain, MS Schechter, S Lommatzsch, V Indihar, N Lechtzin and K McBennett. Inhaled mRNA therapy for treatment of cystic fibrosis: Interim results of a randomized, double‐blind, placebo‐controlled phase 1/2 clinical study. Journal of Cystic Fibrosis 2023; 22(4), 656-664.
LW Plasschaert, KD MacDonald and JS Moffit. Current landscape of cystic fibrosis gene therapy. Frontiers in pharmacology 2024; 15, 1476331.
JD Gillmore, E Gane, J Taubel, J Kao, M Fontana, ML Maitland, J Seitzer, D O’Connell, KR Walsh, K Wood and J Phillips. CRISPR-Cas9 in vivo gene editing for transthyretin amyloidosis. New England Journal of Medicine 2021; 385(6), 493-502.
Y Jiang, S Chen, S Hsiao, H Zhang, D Xie, ZJ Wang, W Ren, M Liu, J Liao and Y Wu. Efficient and safe in vivo treatment of primary hyperoxaluria type 1 via LNP-CRISPR-Cas9-mediated glycolate oxidase disruption. Molecular Therapy 2025; 33(1), 104-118.
TL Hunter, Y Bao, Y Zhang, D Matsuda, R Riener, A Wang, JJ Li, F Soldevila, DS Chu, DP Nguyen and QC Yong. In vivo CAR T cell generation to treat cancer and autoimmune disease. Science 2025; 388(6753), 1311-1317.
Y Lee, M Jeong, J Park, H Jung and H Lee. Immunogenicity of lipid nanoparticles and its impact on the efficacy of mRNA vaccines and therapeutics. Experimental & Molecular Medicine 2023; 55(10), 2085-2096.
JH Skerritt. Considerations for mRNA Product development, regulation and deployment across the lifecycle. Vaccines 2025; 13(5), 473.
AJ Bett, W Haddara, L Prevec and FL Graham. An efficient and flexible system for construction of adenovirus vectors with insertions or deletions in early regions 1 and 3. Proceedings of the National Academy of Sciences 1994; 91(19), 8802-8806.
B Ghebremedhin. Human adenovirus: Viral pathogen with increasing importance. European Journal of Microbiology and Immunology 2014; 4(1), 26-33.
K Toth, M Kuppuswamy, K Doronin, OA Doronina, DL Lichtenstein, AE Tollefson and WS Wold. Construction and characterization of E1-minus replication-defective adenovirus vectors that express E3 proteins from the E1 region. Virology 2002; 301(1), 99-108.
C Li, H Wang, S Gil, A Germond, C Fountain, A Baldessari, J Kim, Z Liu, A Georgakopoulou, S Radtke and T Raskó. Safe and efficient in vivo hematopoietic stem cell transduction in nonhuman primates using HDAd5/35++ vectors. Molecular Therapy Methods & Clinical Development 2022; 24, 127-141.
D Weklak, D Pembaur, G Koukou, F Jönsson, C Hagedorn and F Kreppel. Genetic and chemical capsid modifications of adenovirus vectors to modulate vector-host interactions. Viruses 2021; 13(7), 1300.
JH Wang, DJ Gessler, W Zhan, TL Gallagher and G Gao. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduction and Targeted Therapy 2024; 9(1), 78.
J Zhu, X Huang and Y Yang. Innate immune response to adenoviral vectors is mediated by both Toll-like receptor-dependent and-independent pathways. Journal of Virology 2007; 81(7), 3170-3180.
DH Barouch, MG Pau, JH Custers, W Koudstaal, S Kostense, MJ Havenga, DM Truitt, SM Sumida, MG Kishko, JC Arthur and B Korioth-Schmitz. Immunogenicity of recombinant adenovirus serotype 35 vaccine in the presence of pre-existing anti-Ad5 immunity. The Journal of Immunology 2004; 172(10), 6290-6297.
TW Geisbert, M Bailey, L Hensley, C Asiedu, J Geisbert, D Stanley, A Honko, J Johnson, S Mulangu, MG Pau and J Custers. Recombinant adenovirus serotype 26 (Ad26) and Ad35 vaccine vectors bypass immunity to Ad5 and protect nonhuman primates against ebolavirus challenge. Journal of Virology 2011; 85(9), 4222-4233.
MD Dicks, AJ Spencer, NJ Edwards, G Wadell, K Bojang, SC Gilbert, AV Hill and MG Cottingham. A novel chimpanzee adenovirus vector with low human seroprevalence: Improved systems for vector derivation and comparative immunogenicity. PLoS One 2012; 7(7), e40385.
A Greinacher, T Thiele, TE Warkentin, K Weisser, PA Kyrle and S Eichinger. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. New England Journal of Medicine 2021; 384(22), 2092-2101.
M Scully, D Singh, R Lown, A Poles, T Solomon, M Levi, D Goldblatt, P Kotoucek, W Thomas and W Lester. Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV-19 vaccination. New England Journal of Medicine 2021; 384(23), 2202-2211.
JJ Wang, L Schönborn, TE Warkentin, T Chataway, L Grosse, P Simioni, S Moll, A Greinacher and TP Gordon. Antibody fingerprints linking adenoviral anti-PF4 disorders. New England Journal of Medicine 2024; 390(19), 1827-1829.
AT Baker, RJ Boyd, D Sarkar, A Teijeira-Crespo, CK Chan, E Bates, K Waraich, J Vant, E Wilson, CD Truong and M Lipka-Lloyd. ChAdOx1 interacts with CAR and PF4 with implications for thrombosis with thrombocytopenia syndrome. Science Advances 2021; 7(49), eabl8213.
HE Mudrick, SC Lu, J Bhandari, ME Barry, JR Hemsath, FG Andres, OX Ma, MA Barry and VS Reddy. Structure-derived insights from blood factors binding to the surfaces of different adenoviruses. Nature communications 2024; 15(1), 9768.
AT Sampson, M Hlaváč, AC Gillman, B Douradinha and SC Gilbert. Developing the next-generation of adenoviral vector vaccines. Human Vaccines & Immunotherapeutics 2025; 21(1), 2514356.
F Vigant, D Descamps, B Jullienne, S Esselin, E Connault, P Opolon, T Tordjmann, E Vigne, M Perricaudet and K Benihoud. Substitution of hexon hypervariable region 5 of adenovirus serotype 5 abrogates blood factor binding and limits gene transfer to liver. Molecular Therapy 2008; 16(8), 1474-1480.
CF Shen, A Rodenbrock, S Lanthier, E Burney and M Loignon. Optimization of culture media and feeding strategy for high titer production of an adenoviral vector in HEK 293 fed-batch culture. Vaccines 2024; 12(5), 524.
C Velikonja, L Steenbakkers, J How, M Enns, B Corbett, C McCready, J Nease, P Mhaskar and D Latulippe. Expediting adenovirus titer assays via an algorithmic live-cell imaging technique. Journal of Biotechnology 2024; 395, 216-227.
Y Wu, E Barbieri, WK Smith, A Minzoni, RE Kilgore, W Chu, MA Daniele and S Menegatti. Integrating affinity chromatography in the platform process for Adenovirus purification. Biotechnology and Bioengineering 2025; 122(7), 1780-1792.
CC Joe, RR Segireddy, C Oliveira, A Berg, Y Li, D Doultsinos, S Scholze, A Ahmad, P Nestola, J Niemann and AD Douglas. Accelerated and intensified manufacturing of an adenovirus‐vectored vaccine to enable rapid outbreak response. Biotechnology and Bioengineering 2024; 121(1), 176-191.
F Mathot, E Lefebvre, BG Francq, D Guillaume and E Bourlès. A lyophilised formulation of chimpanzee adenovirus vector for long-term stability outside the deep-freeze cold chain. Communications Medicine 2025; 5(1), 23.
D Wang, CE Porter, B Lim, A Rosewell Shaw, CS Robertson, ML Woods, Y Xu, GG Biegert, D Morita, T Wang and BJ Grilley. Ultralow-dose binary oncolytic/helper-dependent adenovirus promotes antitumor activity in preclinical and clinical studies. Science Advances 2023; 9(13), eade6790.
A Muravyeva and S Smirnikhina. Adenoviral vectors for gene therapy of hereditary diseases. Biology 2024; 13(12), 1052.
S Mathlouthi, L Kuryk, M Prygiel, MG Lupo, AA Zasada, C Pesce, N Ferri, B Rinner, S Salmaso and M Garofalo. Extracellular vesicles powered cancer immunotherapy: Targeted delivery of adenovirus-based cancer vaccine in humanized melanoma model. Journal of Controlled Release 2024; 376, 777-793.
C Cheng, Q Wang and S Zhang. Synergy of oncolytic adenovirus and immune checkpoint inhibitors: transforming cancer immunotherapy paradigms. Frontiers in Immunology 2025; 16, 1610858.
J Olaghere, DA Williams, J Farrar, H Büning, C Calhoun, T Ho, MS Inamdar, D Liu, J Makani, K Nyarko and S Ruiz. Scientific advancements in gene therapies: Opportunities for global regulatory convergence. Biomedicines 2025; 13(3), 758.
J Zengel, YX Wang, JW Seo, K Ning, JN Hamilton, B Wu, M Raie, C Holbrook, S Su, DR Clements and S Pillay. Hardwiring tissue-specific AAV transduction in mice through engineered receptor expression. Nature Methods 2023; 20(7), 1070-1081.
AE Hamouda, J Filtjens, E Brabants, D Kancheva, A Debraekeleer, J Brughmans, L Jacobs, PM Bardet, E Knetemann, P Lefesvre and L Allonsius. Intratumoral delivery of lipid nanoparticle-formulated mRNA encoding IL-21, IL-7, and 4-1BBL induces systemic anti-tumor immunity. Nature Communications 2024; 15(1), 10635.
Z Najjari, F Sadri and J Varshosaz. Smart stimuli-responsive drug delivery systems in spotlight of COVID-19. Asian Journal of Pharmaceutical Sciences 2023; 18(6), 100873.
EI Azhar, SA El-Kafrawy and ZA Memish. Strengthening global health Equity: The imperative of the WHO pandemic agreement. New Microbes and New Infections 2025; 66, 101607.
K Yang. Accelerating vaccine development: Plug-and-play platforms for emerging infectious diseases. Virus Research 2025. https://doi.org/10.1016/j.virusres.2025.199601.
AJL Abreu, CAM Mpande, M Helble, MW Nicholson, MÁ Cortés, MEP Ponsa, IR Blumenthal, F Caccavo, T Pippo, J Rius Sanjuan and C Nannei. Investment opportunities for mRNA technology in low- and middle-income countries: Key findings and future perspectives. Vaccines 2025; 13(2), 112.
AA Saied, AA Metwally, M Dhawan, OP Choudhary and H Aiash. Strengthening vaccines and medicines manufacturing capabilities in Africa: Challenges and perspectives. EMBO Molecular Medicine 2022; 14(8), e16287.
K Lundstrom. COVID-19 vaccines: Where did we stand at the end of 2023? Viruses 2024; 16(2), 203.
PJ Klasse, RW Sanders, AB Ward, IA Wilson and JP Moore. The HIV-1 envelope glycoprotein: Structure, function and interactions with neutralizing antibodies. Nature Reviews Microbiology 2025; 23, 734-752.
X Li, S Jin, S Guo, D Yang, W Sai, X Qiu, X Zhao, L Wang, T Wang and M Li. Platform technology in global vaccine regulation: Development, applications, and regulatory strategies with insights from China. Vaccines 2024; 12(12), 1436.
JH Skerritt, C Tucek-Szabo, B Sutton and T Nolan. The platform technology approach to mRNA product development and regulation. Vaccines 2024; 12(5), 528.
B Canter, S Sussman, S Colvill, N Arad, E Staton and A Rai. Introducing biosimilar competition for cell and gene therapy products. Journal of Law and the Biosciences 2024; 11(2), lsae015.
MB Petersen, A Bor, F Jørgensen and MF Lindholt. Transparent communication about negative features of COVID-19 vaccines decreases acceptance but increases trust. Proceedings of the National Academy of Sciences 2021; 118(29), e2024597118.
MC Parikh. Gene editing: Developments, Ethical considerations, and future directions. Journal of Community Hospital Internal Medicine Perspectives 2025; 15(1), 1.
ADH Nguyen and MT Quang. CRISPR/Cas9 genome editing in oncology: Mechanisms, therapeutic platforms and translational challenges. Molecular Biotechnology 2025. https://doi.org/10.1007/s12033-025-01533-2.
MT Quang, MN Nguyen and VT Than. The role and regulation of cell death in cancer. Progress in Molecular Biology and Translational Science 2025; 217, 135-161.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






