New Onoceranoid-Triterpenoids from the Stem Bark of Lansium domesticum Corr. cv. Piedjietan: Structural Characterization and Their Cytotoxic Activity Evaluated by In Vitro and In Silico Approaches
DOI:
https://doi.org/10.48048/tis.2026.12463Keywords:
Onoceranoid-triterpenoid, Lansium domesticum Corr. cv. Piedjietan, In silico, In vitro, Breast cancer, MCF-7Abstract
This study presents the isolation, characterization and cytotoxic potential of four onoceranoid triterpenoids from the stem bark of Lansium domesticum Corr. cv. Piedjietan, including a newly identified compound, methyl lansic (1), and three known compounds: α,γ-onoceradienedione (2), methyl ester lansiolate (3), and lansiolic acid (4), but reported for the first time in this cultivar. Methyl lansic was tested for cytotoxicity against MCF-7 breast cancer cells. The Results showed that methyl lansic exhibited strong cytotoxic activity with an IC₅₀ value of 7.78 µg/mL, while its IC₅₀ on normal CV-1 cells was substantially higher (620.30 µg/mL), indicating good selectivity toward cancer cells. Molecular docking analysis indicated that methyl lansic and α,γ-onoceradienedione had strong binding affinities to ERα, suggesting their potential as estrogen receptor inhibitors. Molecular dynamics, RMSD/RMSF profiles, persistent key interactions, and favourable MM/GBSA binding energy collectively demonstrate that the methyl lansic–ERα complex is structurally stable and exhibits characteristics consistent with a promising candidate for further evaluation in ERα-positive breast cancer models. These findings expand the phytochemical and pharmacological understanding of Lansium domesticum cv. Piedjietan and highlight its promise as a source of bioactive compounds for the development of anticancer agents.
HIGHLIGHTS
- Methyl lansic was isolated for the first time from Lansium domesticum cv. Piedjietan, expanding the onoceranoid triterpenoid class.
- This study provides the first phytochemical investigation of the Piedjietan cultivar and reports four onoceranoid compounds.
- Methyl lansic demonstrated strong cytotoxicity against MCF-7 cells with an IC₅₀ value of 7.78 µg/mL, indicating its potential as an anticancer agent. The comparison of IC₅₀ values between breast cancer cells MCF-7 and normal CV-1 cells (620.30 µg/mL) shows that methyl lansic is selectively cytotoxic toward breast cancer cells.
- In silico studies including molecular docking, molecular dynamics, and ADMET analyses demonstrated strong binding to ERα and favorable pharmacokinetic properties.
- Bioavailability and safety predictions indicate that methyl lansic is a promising lead compound for the development of therapies targeting estrogen-receptor-positive breast cancer.
GRAPHICAL ABSTRACT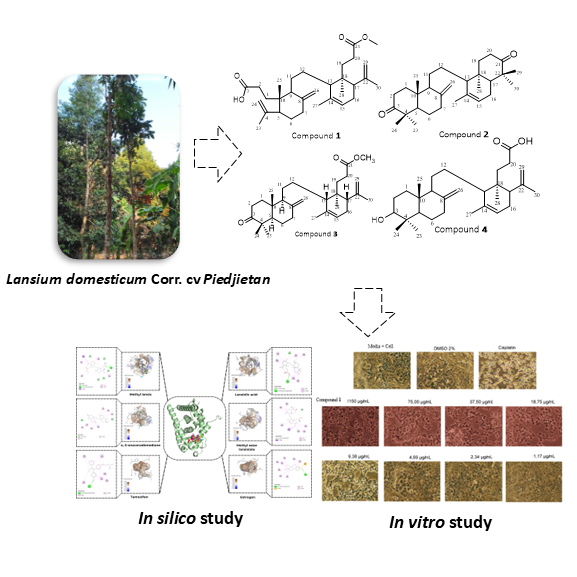
Downloads
References
R Yadav, A Pednekar, A Avalaskar, M Rathi and Y Rewachandani. A comprehensive review on meliaceae family. World Journal of Pharmaceutical Sciences 2015; 3(8), 1572-1577.
K Yulita. Genetic variations of Lansium domesticum Corr. accessions from Java, Bengkulu and ceram based on random amplified ploymorhic DNA. Biodiversitas 2011; 12(3), 125-130.
TK Lim. Edible medicinal and Non-Medicinal plants. Springer, Dordrecht, 2012, p. 265-268.
T Mayanti, SE Sinaga and U Supratman. Phytochemistry and biological activity of Lansium domesticum Corr. species: A review. Journal of Pharmacy and Pharmacology 2022; 74(11), 1568-1587.
R Septiyanti, R Maharani, MA Nafiah and T Mayanti. A review of onoceranoids from the Lansium and Lycopodium genera: phytochemistry and their biological activities. Trends in Sciences 2025; 22(10), 10509.
S Omar, M Marcotte, P Fields, PE Sanchez, L Poveda, R Mata, A Jimenez, T Durst, J Zhang, S MacKinnon, D Leaman, JT Arnason and BJR Philogène. Antifeedant activities of terpenoids isolated from tropical Rutales. Journal of Stored Products Research 2007; 43(1), 92.
T Mayanti, R Tjokronegoro, U Supratman, MR Mukhtar, K Awang and AHA Hadi. Antifeedant triterpenoids from the seeds and bark of Lansium domesticum cv Kokossan (Meliaceae). Molecules 2011; 16(4), 2785-2795.
M Nishizawa, H Nishide and Y Hayashi. Total synthesis of (±)-α,γ-onoceradienedione and lansic acid. Tetrahedron Letters 1984; 25(44), 5071-5074.
R Ramadhan, W Worawalai and P Phuwapraisirisan. Furofuran lignans as a new series of antidiabetic agents exerting α-glucosidase inhibition and radical scarvenging: Semisynthesis, kinetic study and molecular modeling. Bioorganic Chemistry 2019; 87, 783-793.
T Mayanti, SA Azahra, TP Syafriadi, Nurlelasari, R Maharani, E Julaeha, U Supratman, SE Sinaga, S Ekawardhani, and S Fajriah. Onoceranoid triterpenes of Lansium domesticum Corr. cv. Kokossan and their cytotoxicity against MCF-7 breast cancer cells. Trends in Sciences 2025; 22(1), 9000.
A Hardianto, SS Mardetia, W Destiarani, YP Budiman, D Kurnia and T Mayanti. Unveiling the anti-cancer potential of onoceranoid triterpenes from Lansium domesticum Corr. cv. kokosan: An in silico study against estrogen receptor alpha. International Journal of Molecular Sciences 2023; 24(19), 15033.
T Mayanti, LD Ramadhanti, W Safriansyah, Darwati, Nurlelasari, D Harneti, SE Sinaga, E Bachtiar, S Hartati, S Fajriah and U Supratman. Lansioside E, an onoceranoid-type triterpenoid isolated from the fruit peel of Lansium domesticum Corr. cv. Kokossan and its cytotoxic activity against MCF-7 breast cancer cells. Journal of Asian Natural Products Research 2025; 27(11), 1720-1728.
HM Abdallah, GA Mohamed and SRM Ibrahim. Lansium domesticum—A Fruit with Multi-Benefits: Traditional uses, phytochemicals, nutritional value, and bioactivities. Nutrients 2022; 14(7), 1531.
T Matsumoto, T Kitagawa, S Teo, Y Anai, R Ikeda, D Imahori, HSB Ahmad and T Watanabe. Structures and antimutagenic effects of onoceranoid-type triterpenoids from the leaves of Lansium domesticum. Journal of Natural Products 2018; 81(10), 2187-2194.
I Gupta, O Hussein, KS Sastry, S Bougarn, N Gopinath, E Chin-Smith, Y Sinha and HM Koras. Deciphering the complexities of cancer cell immune evasion: Mechanism and therapeutic implications. Advances in Cancer Biology - Metastasis 2023; 8, 100107.
H Qayoom, M Alkhanani, A Almilaibary, SA Alsagaby and MA Mir. Mechanistic elucidation of juglanthraquinone c targeting breast cancer: A network pharmacology-based investigation. Saudi Journal of Biological Sciences 2023; 30(7), 103705.
Mary Ann Liebert, Inc. Antisense & nucleic acid drug development. Mary Ann Liebert, Inc., New Rochelle, 2023.
T Mayanti, Zulfikar, S Fawziah, AA Naini, R Maharani, K Farabi, Nurlelasari, M Yusuf, D Harneti, D Kurnia and U Supratman. New triterpenoids from Lansium domesticum Corr. cv kokossan and their cytotoxic activity. Molecules 2023; 28(5), 2144.
M Nishizawa, H Nishide, S Kosela and Y Hayashi. Structure of lansiosides: Biologically active new triterpene glycosides from Lansium domesticum. The Journal of Organic Chemistry 1983; 48(24), 4462-4466.
LJ Brandes. N,N-diethyl-2-[4-(phenylmethyl) phenoxy] ethanamine (DPPE; tesmilifene), a chemopotentiating agent with hormetic effects on DNA synthesis in vitro, may improve survival in patients with metastatic breast cancer. Human & Experimental Toxicology 2008; 27(2), 143-147.
SA Putri, R Maharani, IP Maksum and TJ Siahaan. Peptide design for enhanced anti-melanogenesis: Optimizing molecular weight, polarity, and cyclization. Drug Design, Development and Therapy 2025; 19, 645-670.
T Barkhem, B Carlsson, Y Nilsson, E Enmark, JÅ Gustafsson and S Nilsson. Differential response of estrogen receptor a and estrogen receptor 13 to partial estrogen agonists/antagonists. Molecular Pharmacology 1998; 54, 105-112.
K Ramasamy, C Samayoa, N Krishnegowda and RR Tekmal. Abstract P1-04-01: Estrogen receptor β agonists inhibits syngeneic mammary tumor growth through cell-cycle arrest by modulating cell-cycle regulators. Cancer Research 2019; 79(S4), P1-04-01.
CA Lipinski. Lead- and drug-like compounds: The rule-of-five revolution. Drug Discovery Today Technologies 2004; 1(4), 337-341.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






