Synthesis and Evaluation of HKHK-PKKKRKV and Its Lipopeptide as New Gene Delivery Candidates
DOI:
https://doi.org/10.48048/tis.2025.9605Keywords:
Gene delivery, NLS peptide, Lipopeptide, Solid phase peptide synthesisAbstract
A major challenge in the delivery of genetic material is finding safe and effective vectors to deliver genetic material into target cells. Although virus-based vectors have proven to be effective in delivering genes to the cell nucleus, they often turn out to be harmful and interact with host cells. On the other hand, naturally available or chemically synthesized nonviral vectors are relatively safer but have low delivery efficiency. Research regarding the use of lipopeptide-based compounds as non-viral vectors for the delivery of genetic material is still limited. This study aims to synthesize NLS-based peptide, HKHK-PKKKRKV (P) and its lipopeptide, C16-HKHK-PKKKRKV (LP), and to evaluate their capability to transport genetic material into the nucleus in vitro. Synthesis of peptide and its lipopeptide was performed using the solid-phase method on 2-chlorotrityl chloride resin with Fmoc chemistry. HATU/HOAt was used as coupling reagents for amide bond formation between 2 amino acids, and DIC/Oxyma was used for fatty acid conjugation to the peptidyl resin. The yields of peptide and lipopeptide obtained were 33 and 26.4 %, respectively. Their structures were confirmed by ESI-TOF-MS, 1H-NMR, and 13C-NMR. The physicochemical analysis of peptide and lipopeptide showed that they were capable of condensing and protecting DNA from enzyme degradation at a ratio of 1:8 to DNA. The peptide and lipopeptide were found to be less efficient for transfection in HEK-293T cells because they required a high concentration (16,000 ng/μL). Nonetheless, peptide and lipopeptide concentrations of 250 - 16,000 ng/μL showed no toxicity to the cells.
HIGHLIGHTS
- Successful synthesis of NLS-based peptide, HKHK-PKKKRKV, and its amphiphilic lipopeptide C16-HKHK-PKKKRKV using solid-phase method on 2-chlrotrityl resin with Fmoc chemistry.
- Implementation of efficient synthesis lipopeptide techniques, which overcame difficult aggregation and hydrophobic interaction issues throughout the assembly process.
- Demonstration of the lipopeptide’s capability as a versatile non-viral vector for gene delivery with adaptable properties for nuclear localization, cellular entrance, and nucleic acid binding.
GRAPHICAL ABSTRACT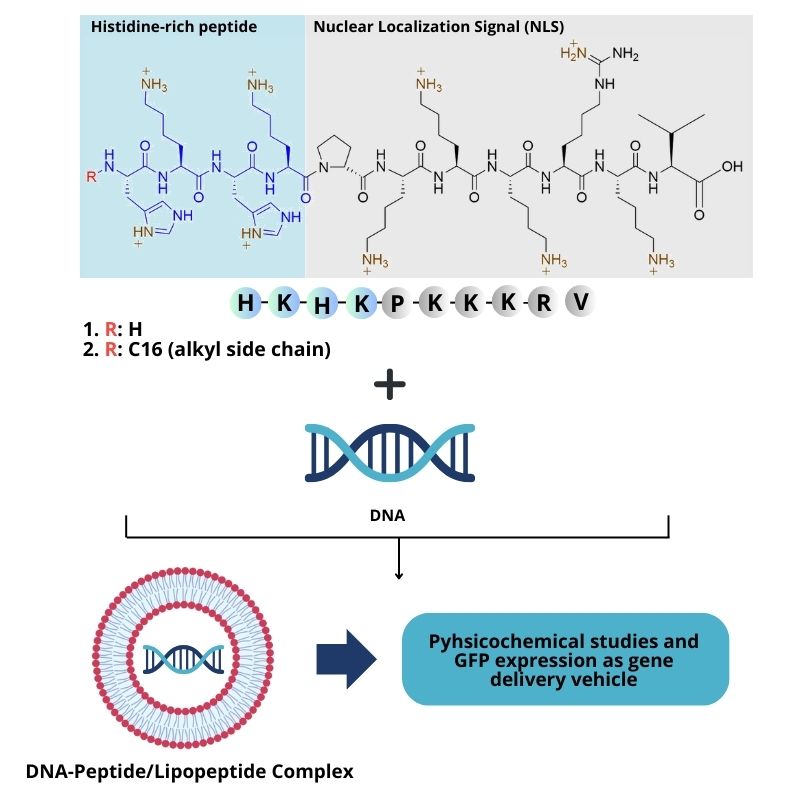
Downloads
References
SL Ginn, M Mandwie, IE Alexander, M Edelstein and MR Abedi. Gene therapy clinical trials worldwide to 2023 - an update. The Journal of Gene Medicine 2024; 26(8), 3721.
JA Kulkarni, D Witzigmann, SB Thomson, S Chen, BR Leavitt, PR Cullis and R van der Meel. The current landscape of nucleic acid therapeutics. Nature Nanotechnology 2021; 16(6), 630-643.
C Wang, C Pan, H Yong, F Wang, T Bo, Y Zhao, B Ma, W He and M Li. Emerging non-viral vectors for gene delivery. Journal of Nanobiotechnology 2023; 21(1), 272.
L Jiao, Z Sun, Z Sun, J Liu, G Deng and X Wang. Nanotechnology-based non-viral vectors for gene delivery in cardiovascular diseases. Frontiers in Bioengineering and Biotechnology 2024; 12, 1349077.
S Tarvirdipour, M Skowicki, CA Schoenenberger and CG Palivan. Peptide-assisted nucleic acid delivery systems on the rise. International Journal of Molecular Sciences 2021; 22(16), 9092.
Y Liu, Z Zhao and M Li. Overcoming the cellular barriers and beyond: Recent progress on cell penetrating peptide modified nanomedicine in combating physiological and pathological barriers. Asian Journal of Pharmaceutical Sciences 2022; 17(4), 523-543.
R Hadianamrei and X Zhao. Current state of the art in peptide-based gene delivery. Journal of Controlled Release 2022; 343, 600-619.
J Yang and GF Luo. Peptide-based vectors for gene delivery. Chemistry 2023; 5(3), 1696-1718.
P Belguise-Valladier and JP Behr. Nonviral gene delivery: Towards artificial viruses. Cytotechnology 2001; 35(3), 197-201.
J He, S Xu, Q Leng and AJ Mixson. Location of a single histidine within peptide carriers increases mRNA delivery. The Journal of Gene Medicine 2021; 23(2), 3295.
A Kichler, AJ Mason and B Bechinger. Cationic amphipathic histidine-rich peptides for gene delivery. Biochim Biophys Acta 2006; 1758(3), 301-307.
J Lu, T Wu, B Zhang, S Liu, W Song, J Qiao and H Ruan. Types of nuclear localization signals and mechanisms of protein import into the nucleus. Cell Communication and Signaling 2021; 19(1), 60.
H Bai, GMS Lester, LC Petishnok and DA Dean. Cytoplasmic transport and nuclear import of plasmid DNA. Bioscience Reports 2017; 37(6), BSR20160616.
EM Cross, N Akbari, H Ghassabian, M Hoad, S Pavan, D Ariawan, CM Donnelly, E Lavezzo, GF Petersen, JK Forwood and G Alvisi. A functional and structural comparative analysis of large tumor antigens reveals evolution of different importin α-dependent nuclear localization signals. Protein Science 2024; 33(2), e4876.
IW Hamley. Lipopeptides: From self-assembly to bioactivity. Chemical Communications 2015; 51(41), 8574-8583.
Tarwadi, JA Jazayeri, RJ Prankerd and CW Pouton. Preparation and in vitro evaluation of novel lipopeptide transfection agents for efficient gene delivery. Bioconjugate Chemistry 2008; 19(4), 940-950.
W Zeng, KJ Horrocks, G Robevska, CY Wong, K Azzopardi, M Tauschek, RM Robins-Browne and DC Jackson. A modular approach to assembly of totally synthetic self-adjuvanting lipopeptide-based vaccines allows conformational epitope building. The Journal of Biological Chemistry 2011; 286(15), 12944-12951.
HY Wang, JX Chen, YX Sun, JZ Deng, C Li, XZ Zhang and RX Zhuo. Construction of cell-penetrating peptide vectors with N-terminal stearylated nuclear localization signal for targeted delivery of DNA into the cell nuclei. Journal of Controlled Release 2011; 155(1), 26-33.
M Paradís-Bas, J Tulla-Puche and F Albericio. The road to the synthesis of “difficult peptides.” Chemical Society Reviews 2016; 45(3), 631-654.
LK Mueller, AC Baumruck, H Zhdanova and AA Tietze. Challenges and perspectives in chemical synthesis of highly hydrophobic peptides. Frontiers in Bioengineering and Biotechnology 2020; 8, 162.
AT Hidayat, R Maharani, AY Chaerunisaa, FF Masduki, R Aditama, H Setiawana and Tarwadi. Synthesis, characterization, and in vitro evaluation of short cationic peptides for gene delivery vehicle candidate. Current Bioactive Compounds 2024; 20(3), 14-24.
CW Chan and PD White. Fmoc solid phase peptide synthesis. Oxford University Press, Nottingham, 2000.
L Cseri, S Kumar, P Palchuber and G Szekely. NMR chemical shifts of emerging green solvents, acids, and bases for facile trace impurity analysis. ACS Sustainable Chemistry & Engineering 2023; 11(14), 5696-5725.
JC Hoch, K Baskaran, H Burr, J Chin, HR Eghbalnia, T Fujiwara, MR Gryk, T Iwata, C Kojima, G Kurisu, D Maziuk, Y Miyanoiri, JR Wedell, C Wilburn, H Yao and M Yokochi. Biological magnetic resonance data bank. Nucleic Acids Research 2023; 51(D1), D368-D376.
V Ravula, YL Lo, LF Wang and SV Patri. Gemini lipopeptide bearing an ultrashort peptide for enhanced transfection efficiency and cancer-cell-specific cytotoxicity. ACS Omega 2021; 6(35), 22955-22968.
H Zhang, Z Chen, M Du, Y Li and Y Chen. Enhanced gene transfection efficiency by low-dose 25 kDa polyethylenimine by the assistance of 1.8 kDa polyethylenimine. Drug Delivery 2018; 25(1), 1740-1745.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






