A Review of Onoceranoids from the Lansium and Lycopodium Genera: Phytochemistry and Their Biological Activities
DOI:
https://doi.org/10.48048/tis.2025.10509Keywords:
Lansium genus, Lycopodium genus, Natural product, Onoceranoid triterpenoid, Pharmacological properties, Phytochemistry, Secondary metabolitesAbstract
Onoceranoids, a subgroup of triterpenoids, have demonstrated considerable potential in medical applications due to their diverse biological activities. They exhibit antioxidant, cytotoxic, antimutagenic, antibacterial and antifeedant properties. This study aimed to review the phytochemical composition and biological activities of onoceranoids found in the Lansium and Lycopodium genera. The chemical structures, biological activities, and plant species serving as sources of onoceranoids are analyzed, emphasizing their potential applications in medicine and implications for future research. Experimental approach: Relevant scientific literature on onoceranoids was collected from databases including SciFinder, Google Scholar, and PubMed, covering research from 1967 to 2025. Fifty onoceranoid compounds were identified and analyzed to determine their biological activities and distribution among plant species. The analysis from this review that 96% onoceranoid compounds were found in the Lansium genus, establishing these compounds as marker constituents of this genus. Onoceranoids demonstrate extensive biological potential due to their multifunctional bioactivities. These findings support further research on onoceranoid compounds, including their structural modification and potential therapeutic applications in medicine.
HIGHLIGHTS
- Onoceranoids, a subgroup of triterpenoids, exhibit a wide range of biological activities, including antioxidant, cytotoxic, antimutagenic, antibacterial, and antifeedant properties.
- This review focuses on the phytochemical composition and biological activities of onoceranoids found in the Lansium and Lycopodium genera.
- From this review 96 percent of the identified onoceranoid compounds were isolated from the Lansium genus, establishing them as chemical markers for this genus.
- A total of 50 onoceranoid compounds were identified and analyzed from scientific literature spanning 1967 to 2025.
- The multifunctional bioactivities of onoceranoids highlight their promising potential for therapeutic applications and future structural modification studies.
GRAPHICAL ABSTRACT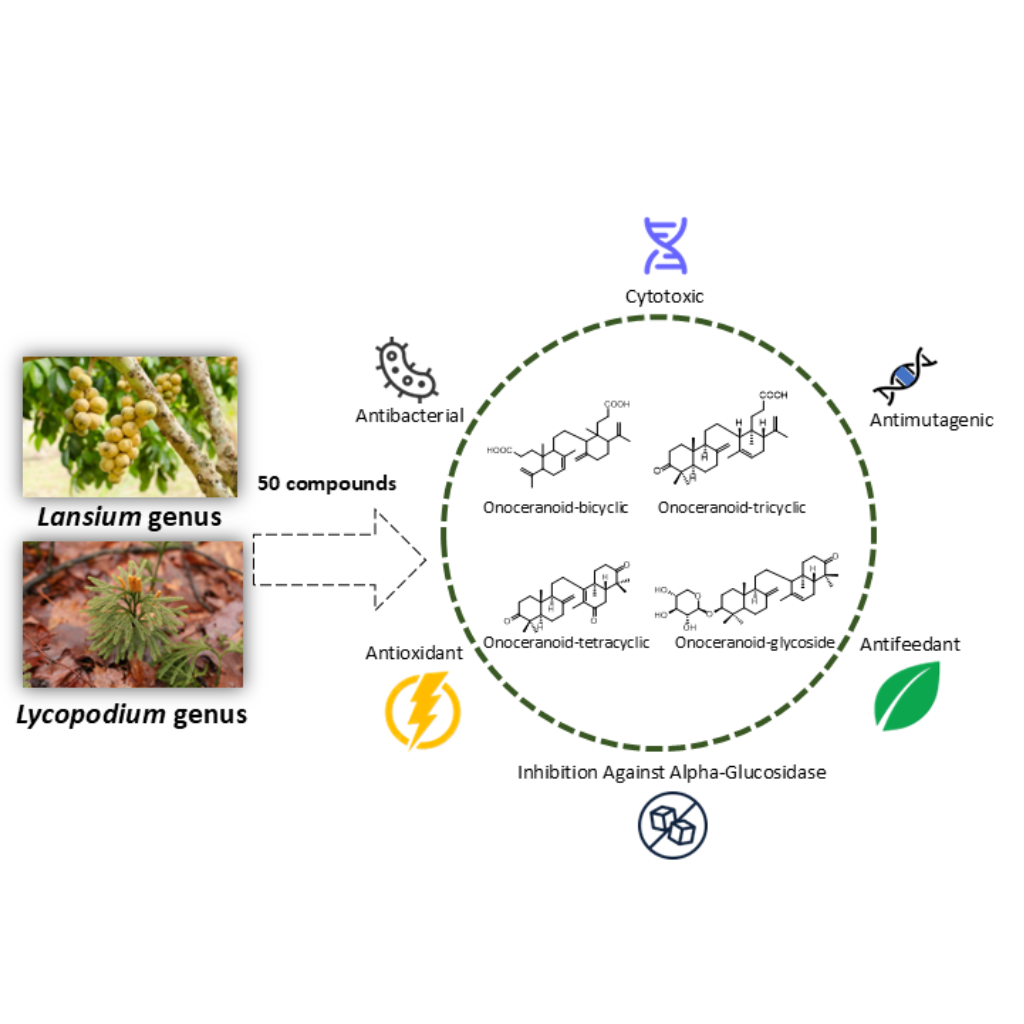
Downloads
References
PM Dewick. Medicinal natural products: a biosynthetic approach. John Wiley & Sons 2002.
Sandeep and S Ghosh. Triterpenoids: Structural diversity, biosynthetic pathway, and bioactivity. Studies in Natural Products Chemistry 2020; 67, 411-461.
J Tang and Y Matsuda. Discovery of fungal onoceroid triterpenoids through domainless enzyme-targeted global genome mining. Nature Communications 2024; 15(1), 4312.
S Yang and J Yue. Discovery of structurally diverse and bioactive compounds from plant resources in China. Acta Pharmacologica Sinica 2012; 33(9), 1147-1158.
Y Chen, Q Yang and Y Zhang. Lycopodium japonicum: A comprehensive review on its phytochemicals and biological activities. Arabian Journal of Chemistry 2020; 13(5), 5438-5450.
S Dong, C Zhang, L Dong, Y Wu and J Yue. Onoceranoid-type triterpenoids from Lansium domesticum. Journal of Natural Products 2011; 74(5), 1042-1048.
S Omar, M Marcotte, P Fields, PE Sanchez, L Poveda, R Mata, A Jimenez, T Durst, J Zhang, S MacKinnon, D Leaman, JT Arnason and BJR Philogene. Antifeedant activities of terpenoids isolated from tropical Rutales. Journal of Stored Products Research 2007; 43(1), 92-96.
T Mayanti, R Tjokronegoro, U Supratman, MR Mukhtar, K Awang and AHA Hadi. Antifeedant triterpenoids from the seeds and bark of Lansium domesticum cv kokossan (Meliaceae). Molecules 2011; 16(4), 2785-2795.
M Nishizawa, H Nishide and Y Hayashi. The structure of lansioside: a novel triterpene glycoside with amino-sugar from Lansium domesticum. Tetrahedron Letters 1982; 23(13), 1349-1350.
R Ramadhan, W Worawalai and P Phuwapraisirisan. New onoceranoid xyloside from lansium parasiticum. Natural Product Research 2019; 33(20), 2917-2924.
T Mayanti, L Abdillah, D Darwati, TP Wikayani, N Qomarilla and DI Dinata. Senyawa triterpenoid 3β-hidroksi-tirukal-7-en dari ekstrak daun kapi nango (Dysoxylum arborescens) dan aktivitas sitotoksiknya terhadap sel kanker payudara MCF-7. Chemica et Natura Acta 2016; 4(3), 138-141.
R Onuki, H Kawasaki, T Baba and K Taira. Analysis of a mitochondrial apoptotic pathway using bid-targeted ribozymes in human MCF-7 cells in the absence of a caspase-3-dependent pathway. Antisense and Nucleic Acid Drug Development 2003; 13(2), 75-82.
HM Abdallah, GA Mohamed and SRM Ibrahim. Lansium domesticum—a fruit with multi-benefits: traditional uses, phytochemicals, nutritional value, and bioactivities. Nutrients 2022; 14(7), 1531.
T Matsumoto, T Kitagawa, S Teo, Y Anai, R Ikeda, D Imahori, HSB Ahmad and T Watanabe. Structures and antimutagenic effects of onoceranoid-type triterpenoids from the leaves of Lansium domesticum. Journal of Natural Products 2018; 81(10), 2187-2194.
M Fellermeier, M Raschke, S Sagner, J Wungsintaweekul, CA Schuhr, S Hecht, K Kis, T Radykewicz, P Adam, F Rohdich, W Eisenreich, A Bacher, D Arigoni and MH Zenk. Studies on the nonmevalonate pathway of terpene biosynthesis. European Journal of Biochemistry 2001; 268(23), 6302-6310.
J Gershenzon and N Dudareva. The function of terpene natural products in the natural world. Nature Chemical Biology 2007; 3(7), 408-414.
CE Vickers, J Gershenzon, MT Lerdau and F Loreto. A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nature Chemical Biology 2009; 5(5), 283-291.
D Tholl. Biosynthesis and biological functions of terpenoids in plants. Springer Nature, Cham, Switzerland, 2015.
T Mayanti, SE Sinaga and U Supratman. Phytochemistry and biological activity of Lansium domesticum Corr. species: A review. Journal of Pharmacy and Pharmacology 2022; 74(11), 1568-1587.
KC Camargo, MGD Aguilar, ARA Moraes, RGD Castro, D Szczerbowski, ELM Migue, LR Oliveira, GF Sousa, DM Vidal and LP Duarte. Pentacyclic triterpenoids isolated from Celastraceae: a focus in the 13C-NMR Data. Molecules 2022; 27(3), 959.
Harizon, B Pujiastuti, D Kurnia, D Sumiarsa and Y Shiono. Triterpenoid lupan dari kulit batang Sonneratia Alba (Lythraceae). Jurnal Ilmu-Ilmu Hayati dan Fisik 2014; 16(1), 25-29.
AA Naini, T Mayanti and U Supratman. Triterpenoids from Dysoxylum genus and their biological activities. Archives of Pharmacal Research 2022; 45(2), 63-89.
S Boonya-udtayan, N Thasana, N Jarussophon and S Ruchirawat. Serratene triterpenoids and their biological activities from Lycopodiaceae plants. Fitoterapia 2019; 136, 104181.
YP Yang, Tasneem, Shumailaa, D Muhammada, Z Liua, J Yan-Zhea, J Yu-Qinga, L Bina and W Weia. Lanostane tetracyclic triterpenoids as important sources for anti-inflammatory drug discovery. World Journal of Traditional Chinese Medicine 2020; 6(3), 229-238.
M Lin, X Liu, J Chen, J Huang and L Zhou. Insecticidal triterpenes in Meliaceae III: Plant species, molecules, and activities in Munronia–Xylocarpus. International Journal of Molecular Sciences 2024; 25(14), 7818.
AK Kiang, EL Tan, FY Lim, K Habaguchi, K Nakanishi, L Fachan and G Ourisson. Lansic acid a bicyclic triterpene. Tetrahedron Lett 1967; 8(37), 3571-3574.
T Potipiranun, W Worawalai and P Phuwapraisirisan. Lamesticumin G, a new α-glucosidase inhibitor from the fruit peels of Lansium parasiticum. Natural Product Research 2018; 32(16), 1881-1886.
Y Zhao, T Deng, Y Chen, X Liu and GZ Yang. Two new triterpenoids from Lycopodium obscurum L. Journal of Asian Natural Products Research 2010; 12(8), 666-671.
HA Noushahi, HA Noushahi, AH Khan, UF Noushahi, M Hussain, T Javed, M Zafar, M Batool, U Ahmed, K Liu, MT Harrison, S Saud, S Fahad and S Shu. Biosynthetic pathways of triterpenoids and strategies to improve their biosynthetic efficiency. Plant Growth Regulation 2022; 97(3), 439-454.
J Liu, X Yin, C Kou, R Thimmappa, X Hua and Z Xue. Classification, biosynthesis, and biological functions of triterpene esters in plants. Plant Communications 2024; 5(4), 100845.
D Cox-Georgian, N Ramadoss, C Dona and C Basu. Therapeutic and medicinal uses of terpenes. Springer Nature, Cham, Switzerland, 2019.
MO Oyedeji-Amusa, NJ Sadgrove and BEV Wyk. The ethnobotany and chemistry of south african meliaceae: A review. Plants 2021; 10(9), 1796.
AN Muellner, H Schaefer and R Lahaye. Evaluation of candidate DNA barcoding loci for economically important timber species of the mahogany family (Meliaceae). Molecular Ecology Resources 2011; 11(3), 450-460.
J Gershenzon and TJ Mabry. Secondary metabolites and the higher classification of angiosperms. Nordic Journal of Botany 1983; 3(1), 5-34.
DJ Mabberley. Florae malesianae praecursores LXVII. meliaceae (divers genera). Blumea: Biodiversity, Evolution and Biogeography of Plants 1985; 31(1), 129-152.
C Techavuthiporn. Langsat lansium domesticum. Elsevier Science, Amsterdam, Netherlands, 2018.
N Saewan, JD Sutherland and K Chantrapromma. Antimalarial tetranortriterpenoids from the seeds of Lansium domesticum Corr. Phytochemistry 2006; 67(20), 2288-2293.
A Ludwiczuk, K Skalicka-Wozniak and MI Georgiev. Terpenoids. Elsevier Science, Amsterdam, Netherlands, 2017.
S Sunarti. Anatomi daun dan taksonomi duku kokosan dan pisitan. Floribunda 1997; 1,1-15.
DTT Yapp and SY Yap. Lansium domesticum: Skin and leaf extracts of this fruit tree interrupt the lifecycle of Plasmodium falciparum, and are active towards a chloroquine-resistant strain of the parasite (T9) in vitro. Journal of Ethnopharmacology 2003; 85(1), 145-150.
FM Fawwaz, SR Meilanie, Zulfikar, K Farabi, T Herlina, JA Anshori and T Mayanti. Kokosanolide D: A new tetranortriterpenoid from fruit peels of Lansium domesticum Corr. cv kokossan. Molbank 2021; 2021(2), M1232.
H Fun, S Chantrapromma, N Boonnak, K Chaiyadej, K Chantrapromma and X Yu. Dukunolide F: A new tetranortriterpenoid from Lansium domesticum Corr. Acta Crystallographica Section E: Structure Reports Online 2006; 62(9), o3725-o3727.
M Nishizawa, Y Nademoto, S Sastrapradja, M Shiro and Y Hayashi. Structure of dukunolide A: A tetranortriterpenoid with a new carbon skeleton from Lansium domesticum. Journal of the Chemical Society, Chemical Communications 1985; 7, 395-396.
SE Sinaga, T Mayanti, AA Naini, D Harneti, N Nurlelasari, R Maharani, K Farabi, U Supratman, S Fajriah and MN Azmi. Sesquiterpenoids from the stem bark of Lansium domesticum corr. cv. kokossan and their cytotoxic activity against MCF-7 breast cancer cell lines. Indonesian Journal of Chemistry 2022; 22(4), 1035-1042.
SE Sinaga, FF Abdullah, U Supratman, T Mayanti and R Maharani. Isolation and structure determination of stigmaterol from the stem bark of Lansium domesticum Corr. Cv. Kokossan. Chimica et Natura Acta 2022; 10(3), 106-111.
JP Benca. Cultivation techniques for terrestrial clubmosses (Lycopodiaceae): Conservation, research, and horticultural opportunities for an early-diverging plant lineage. American Fern Journal 2014; 104(2), 25-48.
RB Primack. Growth patterns of five species of Lycopodium. American Fern Journal 1973; 63(1), 3-7.
RJ Hickey. The lycopodium obscurum complex in North America. American Fern Journal 1977; 67(2), 45-48.
X Ma and D R Gang. The lycopodium alkaloids. Natural Product Reports 2004; 21(6), 752-772.
KM Laemmerhold and B Breit. Total synthesis of (+)‐clavolonine, (−)‐deacetylfawcettiine, and (+)‐acetylfawcettiine. Angewandte Chemie International Edition 2010; 49(13), 2367-2370.
S Wittayalai, S Sathalalai, S Thorroad, P Worawittayanon, S Ruchirawat and N Thasana. Lycophlegmariols A–D: Cytotoxic serratene triterpenoids from the club moss Lycopodium phlegmaria L. Phytochemistry 2012; 76, 117-123.
T Mayanti, Zulfikar, S Fawziah, AA Naini, R Maharani, K Farabi, Nurlelasari, M Yusuf, D Harneti, D Kurnia and U Supratman. New triterpenoids from Lansium domesticum Corr. cv kokossan and their cytotoxic activity. Molecules 2023; 28(5), 2144.
T Tanaka, M Ishibashi, H Fujimoto, E Okuyama, T Koyano, T Kowithayakorn, M Hayashi and K Komiyama. New onoceranoid triterpene constituents from Lansium domesticum. Journal of Natural Products 2002; 65(11), 1709-1711.
AK Kiang, EL Tan, FY Lim, K Habaguchi, K Nakanishi, L Fachan and G Ourisson. Lansic acid, a bicyclic triterpene. Tetrahedron Letters 1967; 8(37), 3571-3574.
B Wang, C Guan and Q Fu. The traditional uses, secondary metabolites, and pharmacology of Lycopodium species. Phytochemistry Reviews 2002; 21, 1-79.
M Nishizawa, H Nishide, K Kuriyama and Y Hayashi. Regioselective reduction α, γ-onoceradienedione: Synthesis of lansiolic acid. Chemical and Pharmaceutical Bulletin 1986; 34(10), 4443-4446.
R Tjokronegero, T Mayanti, U Supratman, MR Mukhtar and SW Ng. 8,14-Secogammacera-7,14(27)-diene-3,21-dione–8,14-secogammacera-7,14-diene-3,21-dione (1.5/0.5) from the bark of Lansium domesticum Corr. Acta Crystallographica 2009; 65(6), o1448.
T Mayanti, U Supratman, MR Mukhtar, K Awang and SW Ng. Kokosanolide from the seed of Lansium domesticum Corr. Acta Crystallographica 2009; 65(4), o750.
Zulfikar, NK Putri, S Fajriah, M Yusuf, R Maharani, JA Anshori, U Supratman and T Mayanti. 3-Hydroxy-8, 14-secogammacera-7, 14-dien-21-one: a new onoceranoid triterpenes from Lansium domesticum Corr. cv kokossan. Molbank 2020; 2020(4), M1157.
T Mayanti, LD Ramadhanti, W Safriansyah, Darwati, Nurlelasari, D Harneti, SE Sinaga, E Bachtiar, S Hartati, S Fajriah and U Supratman. Lansioside E, an onoceranoid-type triterpenoid isolated from the fruit peel of Lansium domesticum Corr. cv. kokossan and its cytotoxic activity against MCF-7 breast cancer cells. Elsevier Science, Amsterdam, Netherlands, 2025
A Jeyasankar, S Premalatha and K Elumalai. Antifeedant and insecticidal activities of selected plant extracts against Epilachna beetle, Henosepilachna vigintioctopunctata (Coleoptera: Coccinellidae). Advances in Entomology 2014; 2(1), 14-19.
LM Schoonven. Biological aspect of antifeedants. Entomologia Experimentalis et Applicata 1982; 31(1), 57-69.
K Munakata. Insect antifeeding substances in plant leaves. Elsevier Science, Amsterdam, Netherlands, 1975.
O Koul. Insect Antifeedants. CRC Press, Florida, 2004.
AF Sandoval‐Mojica and JL Capinera. Antifeedant effect of commercial chemicals and plant extracts against Schistocerca americana (Orthoptera: Acrididae) and Diaprepes abbreviatus (Coleoptera: Curculionidae). Pest Management Science 2011; 67(7), 860-868.
O Koul. Phytochemicals and insect control: An antifeedant approach. Critical Reviews in Plant Sciences 2008; 27(1), 1-24.
M Hasanuzzaman, MHMB Bhuyan, F Zulfiqar, A Raza, SM Mohsin, JA Mahmud, M Fujita and V Fotopoulos. Reactive oxygen species and antioxidant defense in plants under abiotic stress: Revisiting the crucial role of a universal defense regulator. Antioxidants 2020; 9(8), 681.
R Apak, M Ozyurek, K Guclu and E Capanoglu. antioxidant activity/capacity measurement. 2. hydrogen atom transfer (HAT)-based, mixed-mode (Electron Transfer (ET)/HAT), and lipid peroxidation assays. Journal of Agricultural and Food Chemistry 2016; 64(5), 1028-1045.
A Hunyadi. The mechanism(s) of action of antioxidants: from scavenging reactive oxygen/nitrogen species to redox signaling and the generation of bioactive secondary metabolites. Medicinal Research Reviews 2019; 39(6), 2505-2533,
S Losada-Barreiro, Z Sezgin-Bayindir, F Paiva-Martins and C Bravo-Diaz. Biochemistry of antioxidants: Mechanisms and pharmaceutical applications. Biomedicines 2022; 10(12), 3051.
P Chaudhary, P Janmeda, AO Docea, B Yeskaliyeva, AFA Razis, B Modu, D Calina and J Sharifi-Rad. Oxidative stress, free radicals and antioxidants: potential crosstalk in the pathophysiology of human diseases. Frontiers in Chemistry 2023; 11, 1158198.
N Francenia Santos-Sanchez, R Salas-Coronado, C Villanueva-Canongo and B Hernandez-Carlos. Antioxidant compounds and their antioxidant mechanism. IntechOpen Limited, London, 2019.
R Carpenter, YC O’Callaghan, MN O’Grady, JP Kerry and NM O’Brien. Modulatory effects of resveratrol, citroflavan-3-ol, and plant-derived extracts on oxidative stress in U937 cells. Journal of Medicinal Food 2006; 9(2), 187-195.
AM Dirir, M Daou, AF Yousef and LF Yousef. A review of alpha-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochemistry Reviews 200; 21, 1049-1079..
Y Feng, H Nan, H Zhou, P Xi and B Li. Mechanism of inhibition of α-glucosidase activity by bavachalcone. Food Science and Technology 2022; 42, e123421.
NR Perron and JL Brumaghim. A review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochemistry and Biophysics 2009; 53(2), 75-100.
W Worawalai, S Wacharasindhu and P Phuwapraisirisan. N-arylmethylaminoquercitols, a new series of effective antidiabetic agents having α-glucosidase inhibition and antioxidant activity. Bioorganic & Medicinal Chemistry Letters 2015; 25(12), 2570-2573.
R Ramadhan and P Phuwapraisirisan. New arylalkanones from Horsfieldia macrobotrys, effective antidiabetic agents concomitantly inhibiting α-glucosidase and free radicals. Bioorganic & Medicinal Chemistry Letters 2015; 25(20), 4529-4533.
B Sottero, S Gargiulo, I Russo, C Barale, G Poli and F Cavalot. Postprandial dysmetabolism and oxidative stress in type 2 diabetes: pathogenetic mechanisms and therapeutic strategies. Medicinal Research Reviews 2015; 35(5), 968-1031.
AHM Lin, BH Lee and WJ Chang. Small intestine mucosal α-glucosidase: A missing feature of in vitro starch digestibility. Food Hydrocolloids 2016; 53, 163-171.
P Osadebe, E Odoh and P Uzor. Oral anti-diabetic agents-review and updates. Journal of Advances in Medicine and Medical Research 2015; 5(2), 134-159.
CM Payne, C Bernstein and H Bernstein. Apoptosis overview emphasizing the role of oxidative stress, DNA damage and signal transduction pathways. Leukemia & Lymphoma 1995. 19(1-2), 43-93.
S Rajabi, A Ramazani, M Hamidi and T Naji. Artemia salina as a model organism in toxicity assessment of nanoparticles. DARU Journal of Pharmaceutical Sciences 2015; 23(1), 20.
T Efferth, PCH Li, VSB Konkimalla and B Kaina. From traditional Chinese medicine to rational cancer therapy. Trends in Molecular Medicine 2007; 13(8), 353-361.
MJ Ramirez-Exposito, MP Carrera-Gonzalez, MD Mayas and JM Martinez-Martos. Gender differences in the antioxidant response of oral administration of hydroxytyrosol and oleuropein against N-ethyl-N-nitrosourea (ENU)-induced glioma. Food Research International 2021; 140, 110023.
A Allegra, G Pioggia, A Tonacci, M Casciaro, C Musolino and S Gangemi. Synergic crosstalk between inflammation, oxidative stress, and genomic alterations in BCR–ABL-negative myeloproliferative neoplasm. Antioxidants 2020; 9(11), 1037.
Q Labibah, KNW Tun, NS Aminah, AN Kristanti, R Ramadhan, Y Takaya, CAC Abdullah and MJ Masarudin. Cytotoxic constituent in the fruit peel of Lansium domesticum. Rasayan Journal of Chemistry 2021; 14(2), 1336-1340.
NEH Daoud, P Borah, PK Deb, KN Venugopala, W Hourani, M Alzweiri, SK Bardaweel, and V Tiwari. ADMET profiling in drug discovery and development: perspectives of in silico, in vitro and integrated approaches. Current Drug Metabolism 2021; 22(7), 503-522.
A Hardianto, SS Mardetia, W Destiarani, YP Budiman, D Kurnia and T Mayanti. Unveiling the anti-cancer potential of onoceranoid triterpenes from Lansium domesticum Corr. cv. kokosan: An in-silico study against estrogen receptor alpha. International Journal of Molecular Sciences 2023; 24(19), 15033.
AI Arshad, P Ahmad, MI Karobari, JA Asif, MK Alam, Z Mahmood, NA Rahman, N Mamat and MA Kamal. Antibiotics: A bibliometric analysis of top 100 classics. Antibiotics 2020; 9(5), 219.
MI Hutchings, AW Truman and B Wilkinson. Antibiotics: Past, present and future. Current Opinion in Microbiology 2019; 51, 72-80.
E Setiawan, ME Hermanto, N Abdulgani, EN Prasetyo, C Riani, D Wulandari and A Budiharjo. A mini review on analysis of potential antibacterial activity of symbiotic bacteria from indonesian freshwater sponge: An unexplored and a hidden potency. Journal of Tropical Biodiversity and Biotechnology 2024; 9(1), 1-17.
J Sikkema, JA de-Bont and B Poolman. Mechanisms of membrane toxicity of hydrocarbons. Microbiology and Molecular Biology Reviews 1995; 59(2), 201-222.
EJ Prenner, RNAH Lewis and RN McElhaney. The interaction of the antimicrobial peptide gramicidin S with lipid bilayer model and biological membranes. Biochimica et Biophysica Acta (BBA) – Biomembranes 1999; 1462(1-2), 201-221.
JH Weisburger. Antimutagenesis and anticarcinogenesis, from the past to the future. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 2001; 480-481, 23-35.
LA Mitscher, H Telikepalli, E McGhee and DM Shankel. Natural antimutagenic agents. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 1996, 350(1), 143-152.
S Gautam, S Saxena, and S Kumar. Fruits and vegetables as dietary sources of antimutagens. Journal of Food Chemistry and Nanotechnology 2016. 2(3), 97-114.
T Matsumoto and T Watanabe. Isolation and structure elucidation of constituents of citrus limon, Isodon japonicus, and Lansium domesticum as the cancer prevention agents. Genes and Environment 2020; 42(1), 17.
B Godlewska-Zylkiewicz, R Swislocka, M Kalinowska, A Golonko, G Swiderski, Z Arciszewska, E Nalewajko-Sieliwoniuk, M Naumowicz and W Lewandowski. Biologically active compounds of plants: structure-related antioxidant, microbiological and cytotoxic activity of selected carboxylic acids. Materials 2020; 13(19), 4454.
SG Hillier and R Lathe. Terpenes, hormones and life: Isoprene rule revisited. Journal of Endocrinology 2019; 242(2), R9-R22.
LH Reddy and P Couvreur. Squalene: A natural triterpene for use in disease management and therapy. Advanced Drug Delivery Reviews 2009; 61(15), 1412-1426.
K Vavitsas, M Fabris and C Vickers. Terpenoid metabolic engineering in photosynthetic microorganisms. Genes 2018; 9(11), 520.
S Fatima, AF Zahoor, SG Khan, SAR Naqvi, SM Hussain, U Nazeer, A Mansha, H Ahmad, AR Chaudhry and A Irfan. Baeyer–Villiger oxidation: A promising tool for the synthesis of natural products: A review. RSC Advances 2024. 14(32), 23423-23458.
P Weyerstahl, H Marschall-Weyerstahl, J Penninger and L Walther. Terpenes and terpene derivatives. Tetrahedron 1987; 43(22), 5287-5298.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






