Synthesis of Nodupetide Analogue: Substitution of β- to α-hydroxy Acid
DOI:
https://doi.org/10.48048/tis.2024.8342Keywords:
Nodupetide, [(2S,3S)-Hmp]2-nodupetide, SPPS, Cyclopeptide, AntimicrobialAbstract
Nodupetide, a cyclodepsipeptide with the potential as an antimicrobial agent, was isolated from the fermentation of the fungi Nodulisporium sp strain IFB-A163. It possessed antibacterial activity against Pseudomonas aeruginosa and revealed an insecticidal activity. In our effort to synthesized nodupeptide, the preparation of the (3S,4S)-3-hydroxy-4-methylhexanoic acid (HMHA) as one of the nodupetide residue was found to be challenging, where it is not commercially available and the synthetic trial of the HMHA was not successful. This led us to synthesize nodupetide derivative by replacing HMHA with its a-hydroxy acid to give the analogue [(2S,3S)-Hmp]2-nodupetide. A combination of solid- and solution-phase peptide synthesis was applied in the synthesis nodupetide analogue. Esterification on the resin for synthesis [(2S,3S)-Hmp]2-nodupetide was achieved by placing the reaction at the last stage of the linear peptide synthesis and obtained in overall yield of 4.3 %. We also conducted antimicrobial activity screening for [(2S,3S)-Hmp]2-nodupetide and its linear precursor, showing inactive activity. The alter of hydrophobicity on the nodupetide analogue has very important role in the difference of bioactivity compared to their parent compound.
HIGHLIGHTS
- The synthesized analogue compound has not been reported previously
- we studied the synthesis strategy of cyclodepsipeptide.
- We studied the change of beta hydroxy carboxylic acid residue to alpha hydroxy carboxylic acid, which has a very significant effect on biological activity.
GRAPHICAL ABSTRACT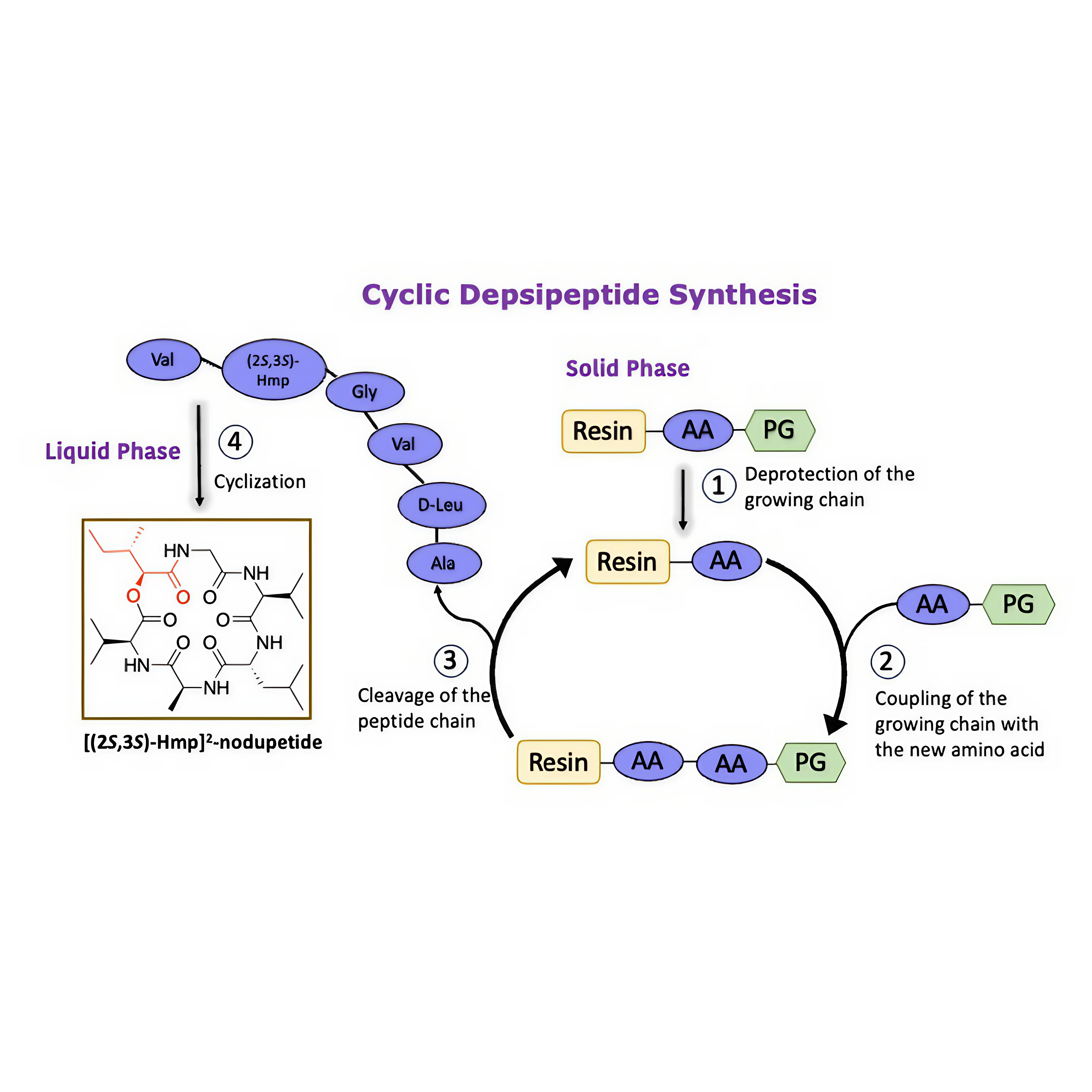
Downloads
References
Y Ding, JP Ting, J Liu, S Al-Azzam, P Pandya and S Afshar. Impact of non-proteinogenic amino acids in the discovery and development of peptide therapeutics. Amino Acids 2020; 52, 1207-26.
C Domhan, P Uhl, C Kleist, S Zimmermann, F Umstatter, K Leotta, W Mier and M Wink. Replacement of L-amino acids by D-amino acids in the antimicrobial peptide ranalexin and its consequences for antimicrobial activity and biodistribution. Molecules 2019; 24, 2987.
HM Wu, LP Lin, QL Xu, WB Han, S Zhang, ZW Liu, YN Mei, ZJ Yao and RX Tan. Nodupetide, a potent insecticide and antimicrobial from Nodulisporium sp. associated with Riptortus pedestris. Tetrahedron Lett. 2017; 58, 663-5.
A Rahmadani, MA Masruhim, L Rijai, A Tatang, U Supratman and R Maharani. Total synthesis of cyclohexadepsipeptides exumolides A and B. Tetrahedron 2021; 83, 131987.
R Maharani, RTC Brownlee, AB Hughes and BM Abbott. A total synthesis of a highly N-methylated cyclodepsipeptide [2S,3S-Hmp]-aureobasidin L using solid-phase methods. Tetrahedron 2014; 70, 2351-8.
I Shin, MR Lee, J Lee, M Jung, W Lee and J Yoon. Synthesis of optically active phthaloyl D-aminooxy acids from L-amino acids or L-hydroxy acids as building blocks for the preparation of aminooxy peptides. J. Org. Chem. 2000; 65, 7667-75.
MM Nguyen, N Ong and L Suggs. A general solid phase method for the synthesis of depsipeptides. Org. Biomol. Chem. 2013; 11, 1167-70.
XM Gao, YH Ye and MBB Kutscher. Studies on the synthesis of cyclic pentapeptides as LHRH antagonist and the factors that influence cyclization yield. J. Pept. Sci. 2002; 8, 418-30.
MI Muhajir, A Hardianto, JA Anshori, D Sumiarsa, T Mayanti, N Nurlelasari, D Harneti, A Hidayat, U Supratman and R Maharani. Total synthesis of nocardiotide A by using a combination of Solid‐ and Solution‐Phase methods. ChemistrySelect 2021; 6, 12941-6.
E Strandberg, P Wadhwani and AS Ulrich. Antibiotic potential and biophysical characterization of amphipathic β-stranded [XZ]n peptides with alternating cationic and hydrophobic residues. Front. Med. Tech. 2021; 3, 622096.
Y Chen, MT Guarnieri, AI Vasil, ML Vasil, CT Mant and RS Hodges. Role of peptide hydrophobicity in the mechanism of action of α-helical antimicrobial peptides. Antimicrob. Agents Chemother. 2007; 51, 1398-406.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






