Phenolic Derivatives from Meliaceae Family and Their Biological Activities
DOI:
https://doi.org/10.48048/tis.2025.10855Keywords:
Meliaceae, Phenolic, Secondary metabolites, Bioactive compoundsAbstract
Phenolic compounds are an important class of secondary metabolites found in the Meliaceae family, which is widely distributed in tropical and subtropical regions. This study presents a comprehensive review of 73 scientific publications from 1985 to 2024, focusing on the occurrence, classification, and biological activities of phenolic compounds isolated from various plant parts such as leaves, barks, seeds, roots, fruits, and flowers. A total of 147 phenolic compounds were identified, categorized into major groups including simple phenolics, coumarins, stilbenes, flavonoids, and lignans. Lignans were the most abundant class, accounting for 45.6% of the total compounds reported. These phenolic constituents exhibit a wide range of biological activities, such as antioxidant, anti-inflammatory, antimicrobial, anticancer, and agonistic activity. Notably, several lignans, such as methyl rocaglate and its derivatives, displayed potent cytotoxicity in the nanomolar range (IC₅₀ = 0.0023 - 0.068 µM), highlighting their potential as anticancer agents. Data were collected and analyzed using established scientific databases including PubMed, Scopus, Reaxys, and SciFinder. The findings underscore the rich chemical diversity and therapeutic potential of Meliaceae-derived phenolics, particularly lignans, as promising candidates for pharmaceutical development. Further pharmacological and clinical investigations are recommended to validate their efficacy and safety.
HIGHLIGHTS
- A comprehensive review of 147 phenolic compounds from 17 genera within the Meliaceae family.
- Lignans are the predominant class of phenolic compounds, representing 46.3% of total isolates.
- Several compounds demonstrated potent antioxidant, cytotoxic, anti-inflammatory, antimicrobial, and agonistic activities.
- The study identifies Meliaceae-derived phenolics as promising leads for pharmaceutical and therapeutic development.
GRAPHICAL ABSTRACT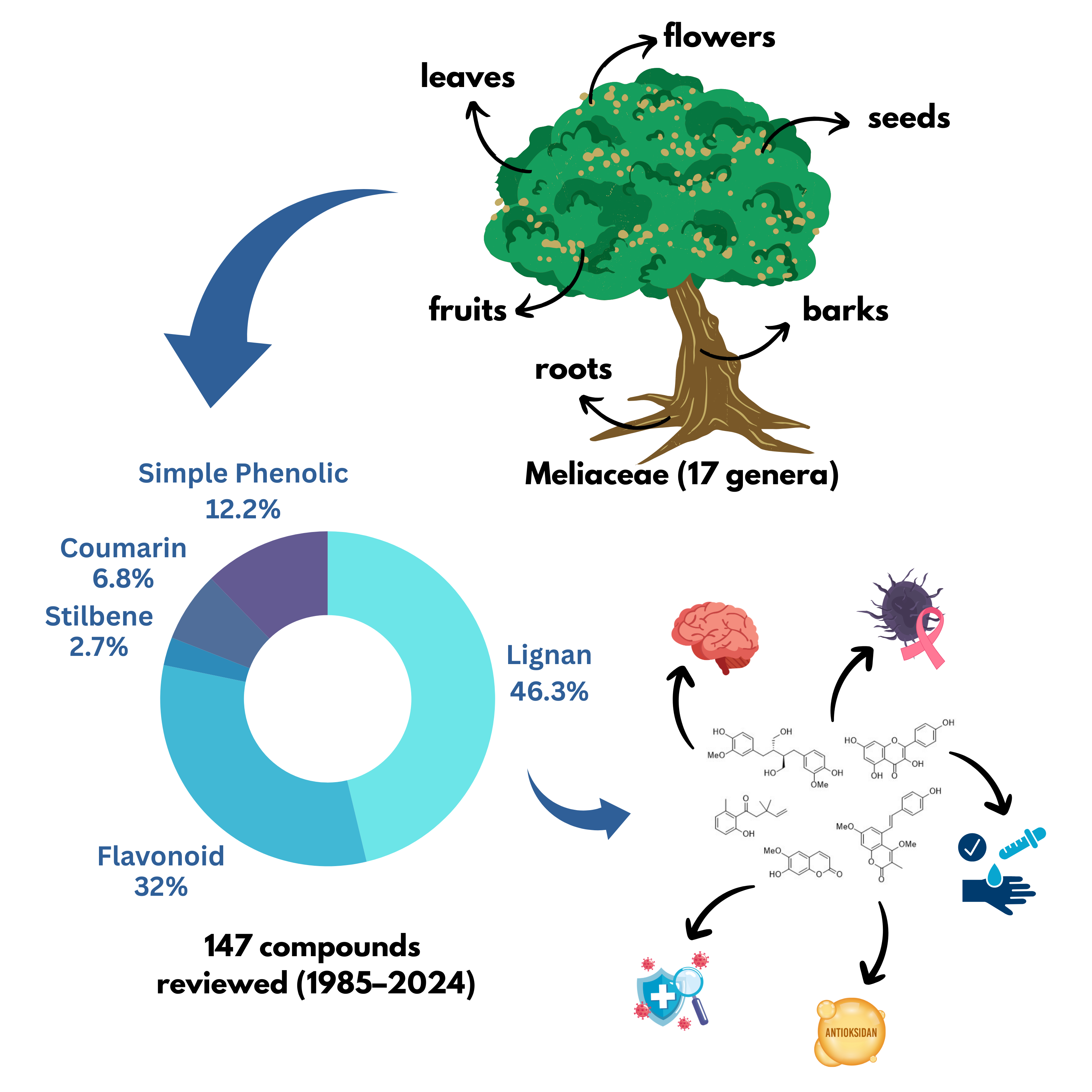
Downloads
References
AN Muellner-Riehl and BM Rojas-Andrés. Biogeography of neotropical Meliaceae: Geological connections, fossil and molecular evidence revisited. Brazilian Journal of Botany 2022; 45(1), 527-543.
TT Hieu, PT Thuy and DX Duc. Isolation and bioactivities of limonoids from Meliaceae family: A review. Current Organic Chemistry 2022; 26(14), 1359-1430.
U Supratman, W Naibaho, S Salam, R Maharani, AT Hidayat, D Harneti, Nurlelasari and Y Shiono. Cytotoxic triterpenoids from the bark of Chisocheton patens Blume (Meliaceae). Phytochemistry Letters 2019; 30, 81-87.
SA Riyadi, AA Naini and U Supratman. Sesquiterpenoids from Meliaceae family and their biological activities. Molecules 2023; 28(12), 4874.
Supriatno, Nurlelasari, T Herlina, D Harneti, R Maharani, AT Hidayat, T Mayanti, U Supratman, MN Azmi and Y Shiono. A new limonoid from stem bark of Chisocheton pentandrus (Meliaceae). Natural Product Research 2018; 32(21), 2610-2616.
SA Riyadi, AA Naini, T Mayanti, R Lesmana, MN Azmi, S Fajriah, S Jungsuttiwong and U Supratman. Alliaxylines A-E: Five new mexicanolides from the stem barks of Dysoxylum alliaceum (Blume) Blume ex A. Juss. Journal of Natural Medicines 2024; 78(3), 558-567.
AA Naini, T Mayanti, R Maharani, D Harneti, Nulelasari, K Farabi, S Fajriah, Hilmayanti, K Kabayama, A Shimoyama, Y Manabe, K Fukase, S Jungsuttiwong, TAK Prescott and U Supratman. Paraxylines AG: Highly oxygenated preurianin-type limonoids with immunomodulatory TLR4 and cytotoxic activities from the stem bark of Dysoxylum parasiticum. Phytochemistry 2024; 220, 114009.
R Ahmad, N Ahmad, AA Naqvi, V Exarchou, A Upadhyay, E Tuenter, K Foubert, S Apers, N Hermans and L Pieters. Antioxidant and antiglycating constituents from leaves of Ziziphus oxyphylla and Cedrela serrata. Antioxidants 2016; 5(1), 9.
PH Yen, NA Bang, DTH Yen, DT Dung, PTT Huong, NH Hoang, BH Tai, LT Anh and PV Kiem. Undescribed 2,9-deoxyflavonoids and flavonol-diamide [3+2] adduct from the leaves of Aglaia odorata Lour. inhibit nitric oxide production. Phytochemistry 2023; 214, 113792.
NKT Dharmayani, T Yoshimura, E Hermawati, LD Juliawaty and YM Syah. Antibacterial and antifungal two phenolic sesquiterpenes from Dysoxylum densiflorum. Zeitschrift für Naturforschung C 2020; 75(1-2), 1-5.
H Wang, CA Geng, HB Xu, XY Huang, YB Ma, CY Yang, XM Zhang and JJ Chen. Lignans from the fruits of Melia toosendan and their agonistic activities on melatonin receptor MT1. Planta Medica 2015; 81(10), 847-854.
B Elya, FS Budiarso, M Hanafi, MA Gani and PW Prasetyaningrum. Two tetrahydroxyterpenoids and a flavonoid from Xylocarpus moluccensis M.Roem. and their α-glucosidase inhibitory and antioxidant capacity. Journal of Pharmacy and Pharmacognosy Research 2024; 12(3), 453-476.
L Peng, WX Fu, CX Zeng, L Zhou, MF Bao and XH Cai. Two new lignans from twigs of Aglaia odorata. Journal of Asian Natural Products Research 2016; 18(2), 147-152.
JY Cho, JH Moon, KY Seong and KH Park. Antimicrobial activity of 4-hydroxybenzoic acid and trans 4-hydroxycinnamic acid isolated and identified from rice hull. Bioscience, Biotechnology, and Biochemistry 1998; 62(11), 2273-2276.
L Wang, F Li, CY Yang, AA Khan, X Liu and MK Wang. Neolignans, lignans and glycoside from the fruits of Melia toosendan. Fitoterapia 2014; 99, 92-98.
ME Cartea, M Francisco, P Soengas and P Velasco. Phenolic compounds in Brassica vegetables. Molecules 2010; 16(1), 251-280.
AJ Aladesanmi and SA Odediran. Antimicrobial activity of Trichilia heudelotti leaves. Fitoterapia 2000; 71(2), 179-182.
Q Li, F Song, M Zhu, Q Wang, Y Han, Y Ling, L Qiao, N Zhong and L Zhang. Hyperoside: A review of pharmacological effects. F1000 Research 2022; 11, 635.
O Myint, S Wattanapongpitak, S Kothan, C Udomtanakunchai, S Tima and M Tungjai. Modulation of p-glycoprotein-mediated efflux pirarubicin in living multidrug-resistant K562/Dox cell lines by 4-hydroxybenzoic acid and 4-hydroxy-3-methoxybenzoic acid via impairment of the cellular energetic state. Toxicology Reports 2022; 9, 1443-1451.
MS Abdelfattah, K Toume, F Ahmed, SK Sadhu and M Ishibashi. Cucullamide, a new putrescine bisamide from Amoora cucullata. Chemical and Pharmaceutical Bulletin 2010; 58(8), 1116-1118.
GPM Kemayou, GM Happi, YAT Ngandjui, JC Tchouankeu, N Sewald, MS Ali and SF Kouam. Senegalin, a new phenylpropanoid and other secondary metabolites from the stem bark of Ekebergia senegalensis A. Juss. (Meliaceae). Natural Product Research 2021; 35(21), 3694-3700.
J Phongmaykin, T Kumamoto, T Ishikawa, R Suttisri and E Saifah. A new sesquiterpene and other terpenoid constituents of Chisocheton penduliflorus. Archives of Pharmacal Research 2008; 31, 21-27.
DA Mulholland, HA Mahomed and S Iourine. A comparison of extractives from the bark of Ekebergia capensis and Ekebergia senegalensis. South African Journal of Botany 1997; 63(5), 259-260.
MY Li, XB Yang, JY Pan, G Feng, Q Xiao, J Sinkkonen, T Satyanandamurty and J Wu. Granatumins A-G, limonoids from the seeds of a krishna mangrove, Xylocarpus granatum. Journal of Natural Products 2009; 72(12), 2110-2114.
KJ Wang, CR Yang and YJ Zhang. Phenolic antioxidants from Chinese toon (fresh young leaves and shoots of Toona sinensis). Food Chemistry 2007; 101(1), 365-371.
SK Sadhu, P Phattanawasin, MSK Choudhuri, T Ohtsuki and M Ishibashi. A new lignan from Aphanamixis polystachya. Journal of Natural Medicines 2006; 60(3), 258-260.
JY Salib, HN Michael and SI El-Nogoumy. New lactoyl glycoside Quercetin from Melia azedarach leaves. Chemistry of Natural Compounds 2008; 44, 13-15.
MDR Camacho, JD Phillipson, SL Croft, GC Kirby, DC Warhurst and PN Solis. Terpenoids from Guarea rhophalocarpa. Phytochemistry 2001; 56(2), 203-210.
BS Siddiqui, M Rasheed, F Ilyas, T Gulzar, RM Tariq and SNUH Naqvi. Analysis of insecticidal Azadirachta indica A. Juss. fractions. Zeitschrift für Naturforschung C 2004; 59(1-2), 104-112.
D Chávez, HB Chai, TE Chagwedera, Q Gao, NR Farnsworth, GA Cordell, JM Pezzuto and AD Kinghorn. Novel stilbenes isolated from the root bark of Ekebergia benguelensis. Tetrahedron Letters 2001; 42(22), 3685-3688.
S Roy, B Banerjee and JR Vedasiromoni. Cytotoxic and apoptogenic effect of Swietenia mahagoni (L.) Jacq. leaf extract in human leukemic cell lines U937, K562 and HL-60. Environmental Toxicology and Pharmacology 2014; 37(1), 234-247.
W Kitdamrongtham, K Ishii, K Ebina, J Zhang, M Ukiya, K Koike, H Akazawa, A Manosroi and T Akihisa. Limonoids and flavonoids from the flowers of Azadirachta indica var. siamensis, and their melanogenesis-inhibitory and cytotoxic activities. Chemistry and Biodiversity 2014; 11(1), 73-84.
J Sianturi, M Purnamasari, T Mayanti, D Harneti, U Supratman, K Awang and H Hayashi. Flavonoid compounds from the bark of Aglaia eximia (Meliaceae). Makara Journal of Science 2015; 19(1), 2.
LS Sidjui, YOD Nganso, RMK Toghueo, BNK Wakeu, JT Dameue, P Mkounga, A Adhikari, M Lateef, GN Folefoc and MS Ali. Kostchyienones A and B, new antiplasmodial and cytotoxicity of limonoids from the roots ofPseudocedrela kotschyi (Schweinf.) Harms. Zeitschrift für Naturforschung C 2018; 73(3-4), 153-160.
SA Jain and SK Srivastava. 8-c-methyl-quercetin-3-o-β/-d-xylopyranoside, a new flavone glycoside from the roots of Amoora rohituka. Journal of Natural Products 1985; 48(2), 299-301.
J Puripattanavong, S Weber, V Brecht and AW Frahm. Phytochemical investigation of Aglaia andamanica. Planta Medica 2000; 66(8), 740-745.
C Pereira, CBB Júnior, RM Kuster, NK Simas, CM Sakuragui, A Porzel and L Wessjohann. Flavonoids and a neolignan glucoside from Guarea macrophylla (Meliaceae). Química Nova 2012; 35, 1123-1126.
W Tang, H Hioki, K Harada, M Kubo and Y Fukuyama. Antioxidant phenylpropanoid-substituted epicatechins from Trichilia catigua. Journal of Natural Products 2007; 70(12), 2010-2013.
K He, BN Timmermann, AJ Aladesanmi and L Zeng. A biflavonoid from Dysoxylum lenticellare gillespie. Phytochemistry 1996; 42(4), 1199-1201.
RB Teponno, S Kusari and M Spiteller. Recent advances in research on lignans and neolignans. Natural Product Reports 2016; 33(9), 1044-1092.
SP Voravuthikunchai, T Kanchanapoom, N Sawangjaroen and N Hutadilok-Towatana. Antioxidant, antibacterial and antigiardial activities of Walsura robusta Roxb. Natural Product Research 2010; 24(9), 813-824.
DAG Cortez, JB Fernandes, QB Cass, PC Vieira, MFGD Silva, AG Ferreira and JR Pirani. Lignan glycosides from Trichilia estipulata bark. Natural Product Letters 1998; 11(4), 255-262.
J Sianturi, D Harneti, Darwati, T Mayanti, U Supratman and K Awang. A New (-)-5′,6-dimethoxyisolariciresinol-(3′′,4′′-dimethoxy)-3α-O-β-d-glucopyranoside from the bark of Aglaia eximia (Meliaceae). Natural Product Research 2016; 30(19), 2204-2208.
BG Wang, H Peng, HL Huang, XM Li, G Eck, X Gong and P Proksch. Rocaglamide, aglain, and other related derivatives from Aglaia testicularis (Meliaceae). Biochemical Systematics and Ecology 2004; 32(12), 1223-1226.
N Fuzzati, W Dyatmiko, A Rahman, F Achmad and K Hostettmann. Triterpenoids, lignans and a benzofuran derivative from the bark of Aglaia elaeagnoidea. Phytochemistry 1996; 42(5), 1395-1398.
SK Lee, B Cui, RR Mehta, AD Kinghorn and JM Pezzuto. Cytostatic mechanism and antitumor potential of novel 1H-cyclopenta[b]benzofuran lignans isolated from Aglaia elliptica. Chemico-Biological Interactions 1998; 115(3), 215-228.
IS Lee, HJ Kim, UJ Youn, QC Chen, JP Kim, DT Ha, TM Ngoc, BS Min, SM Lee, HJ Jung, MK Na and KH Bae. Dihydrobenzofuran norlignans from the leaves of Cedrela sinensis A. Juss. Helvetica Chimica Acta 2010; 93(2), 272-276.
DR Kar, P Ghosh, P Suresh, S Chandra and D Paul. Review on phyto-chemistry & pharmacological activity of Melia azedarach. International Journal of Experimental Research and Review 2022; 28, 38-46.
TJ Hsieh, TZ Liu, YC Chia, CL Chern, FL Lu, MC Chuang, SY Mau, SH Chen, YH Syu and CH Chen. Protective effect of methyl gallate from Toona sinensis (Meliaceae) against hydrogen peroxide-induced oxidative stress and DNA damage in MDCK cells. Food and Chemical Toxicology 2004; 42(5), 843-850.
CAVD Heijden, PJ Janssen and JJ Strik. Toxicology of gallates: A review and evaluation. Food and Chemical Toxicology 1986; 24(10-11), 1067-1070.
SA Pshenichnyuk, YN Elkin, NI Kulesh, EF Lazneva and AS Komolov. Low-energy electron interaction with retusin extracted from Maackia amurensis: Towards a molecular mechanism of the biological activity of flavonoids. Physical Chemistry Chemical Physics 2015; 17(26), 16805-16812.
WJ Choung, SH Hwang, DS Ko, SB Kim, SH Kim, SH Jeon, HD Choi, SS Lim and JH Shim. Enzymatic synthesis of a novel kaempferol-3-O-β-D-glucopyranosyl-(1→4)-O-α-D-glucopyranoside using cyclodextrin glucanotransferase and its inhibitory effects on aldose reductase, inflammation, and oxidative stress. Journal of Agricultural and Food Chemistry 2017; 65(13), 2760-2767.
A Szakiel, L Voutquenne-Nazabadioko and M Henry. Isolation and biological activities of lyoniside from rhizomes and stems of Vaccinium myrtillus. Phytochemistry Letters 2011; 4(2), 138-143.
X Li, W Cao, Y Shen, N Li, XP Dong, KJ Wang and YX Cheng. Antioxidant compounds from Rosa laevigata fruits. Food Chemistry 2012; 130(3), 575-580.
CW Song, SM Wang, LL Zhou, FF Hou, KJ Wang and QB Han. Isolation and identification of compounds responsible for antioxidant capacity of Euryale ferox seeds. Journal of Agricultural and Food Chemistry 2011; 59(4), 1199-1204.
Y Yu, ZB Wang, YD Zhai, PY Song, QH Wang, BY Yang and HX Kuang. Lignan glycosides from Gentianella acuta (Michx.) Hultén. and their protective effects against H₂O₂-induced apoptosis in H9c2 cardiomyoblasts. Records of Natural Products 2014; 8(3), 234-241.
J Xiong, VB Bui, XH Liu, ZL Hong, GX Yang and JF Hu. Lignans from the stems of Clematis armandii (“Chuan-Mu-Tong”) and their anti-neuroinflammatory activities. Journal of Ethnopharmacology 2014; 153(3), 737-743.
JJ Chen, HB Wei, YZ Xu, J Zeng and K Gao. Antioxidant lignans from the roots of Vladimiria muliensis. Planta Medica 2013; 79(15), 1470-1473.
K Prasad. Antioxidant activity of secoisolariciresinol diglucoside-derived metabolites, secoisolariciresinol, enterodiol, and enterolactone. International Journal of Angiology 2000; 9(4), 220-225.
HA Ali, AKA Chowdhury, AKM Rahman, T Borkowski, L Nahar and SD Sarker. Pachypodol, a flavonol from the leaves of Calycopteris floribunda, inhibits the growth of CaCo2 colon cancer cell line in vitro. Phytotherapy Research 2008; 22(12), 1684-1687.
EK Kim, JH Kim, S Jeong, YW Choi, HJ Choi, CY Kim and YM Kim. Pachypodol, a methoxyflavonoid isolated from Pogostemon cablin Bentham exerts antioxidant and cytoprotective effects in HepG2 cells: Possible role of ERK-dependent Nrf2 activation. International Journal of Molecular Sciences 2019; 20(17), 4082.
H Wang, R Li, X Xi, T Meng, M Zhou, L Wang, Y Zhang, T Chen and C Shaw. Senegalin: A novel antimicrobial/myotropic hexadecapeptide from the skin secretion of the African running frog, Kassina senegalensis. Amino Acids 2013; 44(5), 1347-1355.
N Mahmood, S Piacente, C Pizza, A Burke, AI Khan and AJ Hay. The anti-HIV activity and mechanisms of action of pure compounds isolated from Rosa damascena. Biochemical and Biophysical Research Communications 1996; 229(1), 73-79.
F Zhao, L Chen, C Bi, M Zhang, W Jiao and X Yao. In vitro anti-inflammatory effect of picrasmalignan A by the inhibition of iNOS and COX-2 expression in LPS-activated macrophage RAW 264.7 cells. Molecular Medicine Reports 2013; 8(5), 1575-1579.
SK Mun, KY Kang, HY Jang, YH Hwang, SG Hong, SJ Kim, HW Cho, DJ Chang, JS Hur and ST Yee. Atraric acid exhibits anti-inflammatory effect in lipopolysaccharide-stimulated RAW264.7 cells and mouse models. International Journal of Molecular Sciences 2020; 21(19), 7070.
A Sudha, J Jeyakanthan and P Srinivasan. Protective effect of 5-hydroxy-3′,4′,7-trimethoxyflavone against inflammation induced by lipopolysaccharide in RAW 264.7 macrophage: In vitro study and in silico validation. Medical Chemistry Research 2016; 25, 1754-1767.
J Attoumbré, S Charlet, S Baltora-Rosset, C Hano, SRL Grandic, F Gillet, L Bensaddek, F Mesnard and MA Fliniaux. High accumulation of dehydrodiconiferyl alcohol-4-β-D-glucoside in free and immobilized Linum usitatissimum cell cultures. Plant Cell Reports 2006; 25, 859-864.
L Hu, XM Tian, CC Wang, Q Olga, D Yan, PF Tang, LN Zhang, LY Kong and J Luo. New triterpenoids, steroids and lignan from the stem barks of Entandrophragma utile. Fitoterapia 2020; 143, 104546.
SM Yang, QS Song, C Qing, DG Wu and XK Liu. Anticancer activity of tirucallane triterpenoids from Amoora dasyclada. Zeitschrift für Naturforschung C 2006; 61(3-4), 193-195.
WDS Terra, IJC Vieira, R Braz-Filho, WRD Freitas, MM Kanashiro and MCM Torres. Lepidotrichilins A and B, new protolimonoids with cytotoxic activity from Trichilia lepidota (Meliaceae). Molecules 2013; 18(10), 12180-12191.
SA Jonker, MHH Nkunya, L Mwamtobe, J Geenevasen and GJ Koomen. A new coumarin and polyhydroxysqualenes from Ekebergia benguelensis. Natural Product Letters 1997; 10(4), 245-248.
BN Irungu, JA Orwa, A Gruhonjic, PA Fitzpatrick, G Landberg, F Kimani, J Madiwo, M Erdélyi and A Yenesew. Constituents of the roots and leaves of Ekebergia capensis and their potential antiplasmodial and cytotoxic activities. Molecules 2014; 19(9), 14235-14246.
T Murata, T Miyase, FW Muregi, Y Naoshima-Ishibashi, K Umehara, T Warashina and A Ishih. Antiplasmodial triterpenoids from Ekebergia capensis. Journal of Natural Products 2008; 71(2), 167-174.
FP Chang, W Chao, SY Wang, HC Huang, PJ Sung, JJ Chen, MJ Cheng, GJ Huang and YH Kuo. Three new iridoid derivatives have been isolated from the stems of Neonauclea reticulata (Havil.) Merr. with cytotoxic activity on hepatocellular carcinoma cells. Molecules 2018; 23(9), 2297.
N Li, F Yang, DY Liu, JT Guo, N Ge and SY Sun. Scoparone inhibits pancreatic cancer through PI3K/Akt signaling pathway. World Journal of Gastrointestinal Oncology 2021; 13(9), 1164-1183.
H Morita, T Dota and J Kobayashi. Antimitotic activity of glaupalol-related coumarins from Glaucidium palmatum. Bioorganic and Medical Chemistry Letters 2004; 14(14), 3665-3668.
W Wu, R Li, X Li, J He, S Jiang, S Liu and L Yang. Quercetin as an antiviral agent inhibits Influenza A virus (IAV) entry. Viruses 2015; 8(1), 6.
GD Yao, J Wang, XY Song, L Zhou, LL Lou, WY Zhao, B lin, XX Huang and SJ Song. Stereoisomeric guaiacylglycerol-β-coniferyl aldehyde ether induces distinctive apoptosis by downregulation of MEK/ERK pathway in hepatocellular carcinoma cells. Bioorganic Chemistry 2018; 81, 382-388.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






