Onoceranoid Triterpenes of Lansium domesticum Corr. cv. Kokossan and Their Cytotoxicity against MCF-7 Breast Cancer Cells
DOI:
https://doi.org/10.48048/tis.2025.9000Keywords:
Lansium domesticum Corr. cv. Kokossan, MCF-7 breast cancer cell, OnoceranoidsAbstract
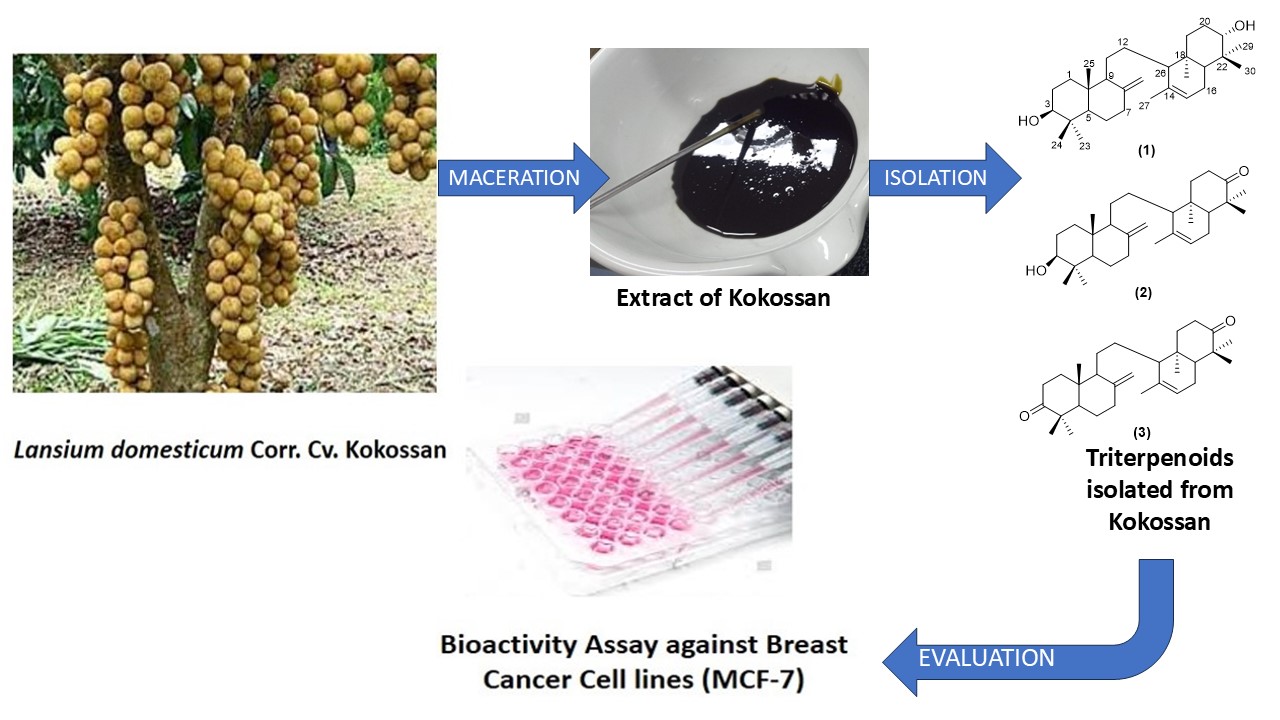
The onoceranoid triterpenes isolated from the kokossan fruit peels were studied in vitro using MCF-7 breast cancer cells. Three onoceranoids, namely 8,14-secogammacera-8(26),14-dien-3,21-diol (1), 3β-hydroxyonocera-8(26),14-dien-21-one (2) and α,γ-onoceradienedione (3), were obtained from the ethyl acetate extract of kokossan fruit peels. Compounds 1 - 3 were obtained for the first time from this cultivar. Several spectroscopic methods, including IR, HR-TOFMS and 1,2 D-NMR, were used to determine the chemical structures of compounds (1 - 3), and the results were compared to spectrum data that had previously been published. Additionally, the activity of compounds 1 - 3 was assessed in vitro using MCF-7 cells. With an IC50 value of 17.11 µg/mL, compound 2 demonstrated the highest activity in the activity test findings, followed by compounds 3 and 1, with IC50 values of 19.66 and > 150 µg/mL, respectively. When compared to cisplatin as a positive control, compounds 2 and 3 showed considerable potency, while compound 1 showed no activity. Compounds 2 and 3 were the most promising candidates for the anticancer drug; nevertheless, more testing is necessary to ascertain their biological mechanism for future research.
HIGHLIGHTS
- Lansium domesticum is a tropical fruit-bearing tree with significant economic and medicinal value. It has been traditionally used for various ailments, including fever, dysentery, and malaria, due to its rich phytochemical composition.
- Kokossan is a cultivar of Lansium domesticum that has not been widely studied for its onoseronoid content, especially in the skin of the fruit. 8,14-sekogammacera-8(26),14-dien-3,21-diol (1), 3β-hydroxyonocera-8(26),14-dien-21-one (2), and α,γ-onoceradienedione (3). For the first time, these two compounds (1 and 2) were separated from this genus; earlier, this compound was the outcome of the reduction (synthesis) of compound 3.
- 3β-hydroxyonocera-8(26),14-dien-21-one exhibited the highest IC50 value compared to α,γ-onoceradienedione and . 8,14-sekogammacera-8(26),14-dien-3,21-diol.
- Compounds 2 and 3 demonstrated potential as anticancer agents, with activity comparable to the standard drug cisplatin. This provides insight that onoceranoids have the potential as chemopreventive agents to treat breast cancer.
GRAPHICAL ABSTRACT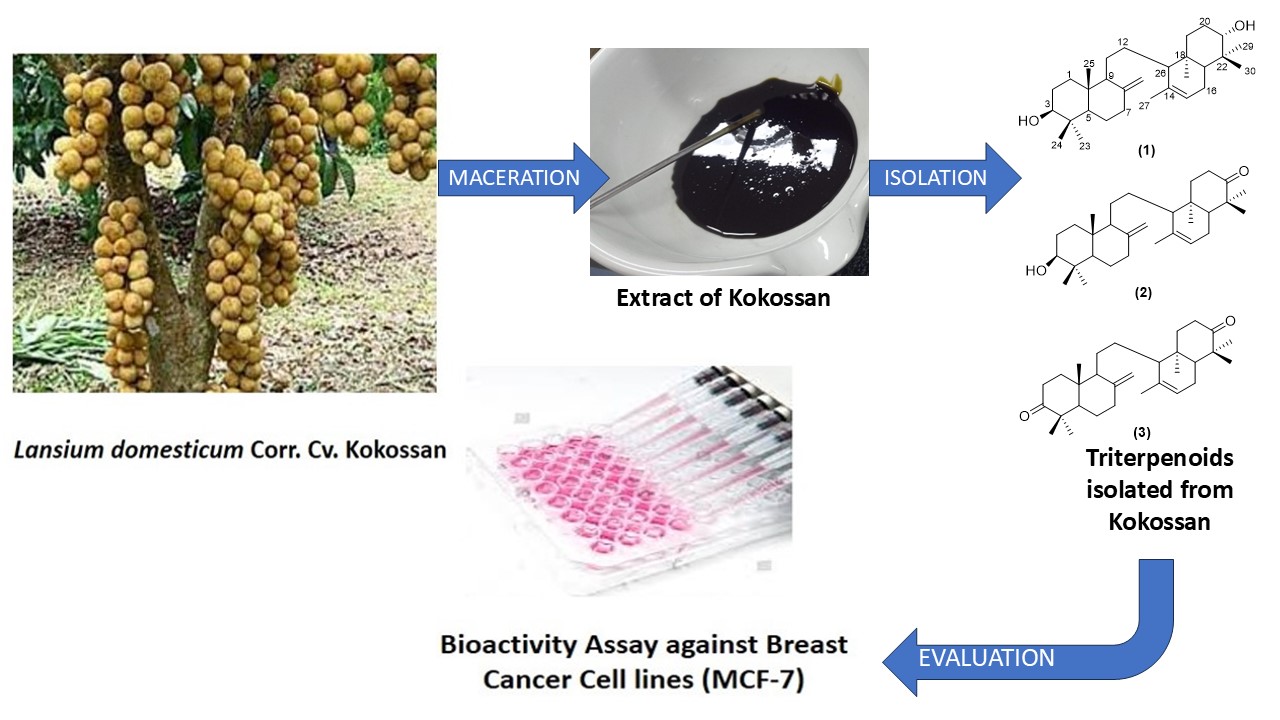
Downloads
References
T Mayanti, SE Sinaga and U Supratman. Phytochemistry and biological activity of Lansium domesticum Corr. species: A review. Journal of Pharmacy and Pharmacology 2022; 74(11), 1568-1587.
T Mayanti, R Tjokronegoro, U Supratman, MR Mukhtar, K Awang and AHA Hadi. Antifeedant triterpenoids from the seeds and bark of Lansium domesticum cv Kokossan (Meliaceae). Molecules 2011; 16(4), 2785-2795.
QG Tan and XD Luo. Meliaceous limonoids: Chemistry and biological activities. Chemical Reviews 2011; 111(11), 7437-7522.
W Safriansyah, SE Sinaga, U Supratman and D Harneti. Phytochemistry and biological activities of Guarea genus (Meliaceae). Molecules 2022; 27(24), 8758.
Y Mulyani, SE Sinaga and U Supratman. Phytochemistry and biological activities of endophytic fungi from the Meliaceae family. Molecules 2023; 28(2), 778.
SE Sinaga, M Fajar, T Mayanti and U Supratman. Bioactivities screening and elucidation of terpenoid from the stembark extracts of Lansium domesticum Corr. cv. Kokosan (Meliaceae). Sustainability 2023; 15(3), 2140.
FM Fauzi, SR Meilanie, Zulfikar, K Farabi, T Herlina, J Al Anshori and T Mayanti. Kokosanolide d: A new tetranortriterpenoid from fruit peels of Lansium domesticum Corr. cv Kokossan. Molbank 2021; 2021(2), M1232.
Zulfikar, NK Putri, S Fajriah, M Yusuf, R Maharani, J Al Anshori, U Supratman and T Mayanti. 3-hydroxy-8,14-secogammacera-7,14-dien-21-one: A new onoceranoid triterpenes from Lansium domesticum Corr. cv Kokossan. Molbank 2020; 2020(4), M1157.
S Purwani, J Nahar and T Mayanti. Molecular docking on kokosanolide A and C for anticancer activity against human breast cancer cell MCF-7. Jurnal Kimia Valensi 2021; 7(1), 52-57.
T Mayanti, Zulfikar, S Fawziah, AA Naini, R Maharani, K Farabi, Nurlelasari, M Yusuf, D Harneti, D Kurnia and U Supratman. New triterpenoids from Lansium domesticum Corr. cv kokossan and their cytotoxic activity. Molecules 2023; 28(5), 2144.
A Hardianto, SS Mardetia, W Destiarani, YP Budiman, D Kurnia and T Mayanti. Unveiling the anti-cancer potential of onoceranoid triterpenes from Lansium domesticum Corr. cv. kokosan: An in silico study against estrogen receptor alpha. International Journal of Molecular Sciences 2023; 24(19), 15033.
SE Sinaga, FF Abdullah, U Supratman, T Mayanti and R Maharani. Isolation and structure determination of stigmaterol from the stem bark of Lansium domesticum Corr. cv. Kokosan (Meliaceae). Chimica et Natura Acta 2022; 10(3), 106-111.
KL Ji, MY Dai, CF Xiao and YK Xu. Two new steroids with NO inhibitory effects from Lansium domesticum. Natural Product Research 2021; 35(7), 1147-1152.
HM Abdallah, GA Mohamed and SRM Ibrahim. Lansium domesticum - a fruit with multi-benefits: Traditional uses, phytochemicals, nutritional value, and bioactivities. Nutrients 2022; 14(7), 1531.
CY Ragasa, P Labrador and JA Rideout. Antimicrobial terpenoids from Lansium domesticum. Philippine Agricultural Scientist 2006; 89(1), 101-105.
K Fadhilah, S Wahyuono and P Astuti. Fractions and isolated compounds from Lansium domesticum fruit peel exhibited cytotoxic activity against T-47D and HepG2 cell lines. Biodiversitas Journal of Biological Diversity 2021; 22(9), 3743-3748.
T Matsumoto, T Kitagawa, T Ohta, T Yoshida, D Imahori, S Teo, HS bin Ahmad and T Watanabe. Structures of triterpenoids from the leaves of Lansium domesticum. Journal of Natural Medicines 2019; 73, 727-734.
T Potipiranun, W Worawalai and P Phuwapraisirisan. Lamesticumin G, a new α-glucosidase inhibitor from the fruit peels of Lansium parasiticum. Natural Product Research 2018; 32(16), 1881-1886.
Kanupriya, PC Kumar and A Sane. An assessment of fruiting and polyembryony in Langsat (Lansium domesticum Corr.) from Nilgiris, India. Journal of Horticultural Sciences 2019; 14(1), 79-82.
K Fadhilah, S Wahyuono and P Astuti. A sesquiterpene aldehyde isolated from ethyl acetate extract of Lansium domesticum fruit peel. Indonesian Journal of Pharmacy 2020; 32(3), 394-398.
SE Sinaga, T Mayanti, AA Naini, D Harneti, N Nurlelasari, R Maharani, K Farabi, U Supratman, S Fajriah and MN Azmi. Sesquiterpenoids from the stem bark of Lansium domesticum Corr. cv. Kokossan and their cytotoxic activity against MCF-7 breast cancer cell lines. Indonesian Journal of Chemistry 2022; 22(4), 1035-1042.
M Nishizawa, H Nishide, K Kuriyama and Y Hayashi. Regioselective reduction of α, γ-onoceradienedione: Synthesis of lansiolic acid. Chemical and Pharmaceutical Bulletin 1986; 34(10), 4443-4446.
W Safriansyah, SE Sinaga, N Rustaman, K Farabi, MN Azmi, R Maharani, N Nurlelasari, U Supratman, S Fajriah and D Harneti. The isolation of novel pregnane steroids from Aglaia pachyphylla Miq. and the cytotoxicity against breast cancer cell lines (MCF-7). RSC Advances 2024; 14, 25042-25047.
T Tanaka, M Ishibashi, H Fujimoto, E Okuyama, T Koyano, T Kowithayakorn, M Hayashi and K Komiyama. New onoceranoid triterpene constituents from Lansium domesticum. Journal of Natural Products 2002; 65(11), 1962-1964.
DF Veber, SR Johnson, HY Cheng, BR Smith, KW Ward and KD Kopple. Molecular properties that influence the oral bioavailability of drug candidates. Journal of Medicinal Chemistry 2002; 45(12), 2615-2623.
A Manzari-Tavakoli, A Babajani, M M Tavakoli, F Safaeinejad and A Jafari. Integrating natural compounds and nanoparticle-based drug delivery systems: A novel strategy for enhanced efficacy and selectivity in cancer therapy. Cancer Medicine 2024; 13(5), e7010.
CA Lipinski. Lead-and drug-like compounds: The rule-of-five revolution. Drug Discovery Today: Technologies 2004; 1(4), 337-341.
NM Krstić, IZ Matić, ZD Juranić, IT Novaković and DM Sladić. Steroid dimers - in vitro cytotoxic and antimicrobial activities. The Journal of Steroid Biochemistry and Molecular Biology 2014; 143, 365-375.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






