Limonoid Extracted from Chisocheton macrophyllus Seeds and Their Cytotoxic Activity against MCF-7 Breast Cancer Cell Lines
DOI:
https://doi.org/10.48048/tis.2025.9173Keywords:
Cytotoxic activity, Chisocheton macrophyllus, MCF-7 cell lines, LimonoidsAbstract
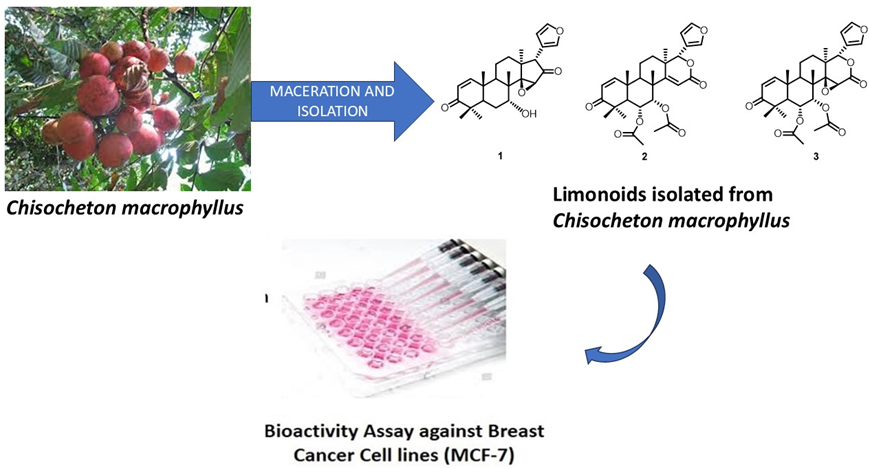
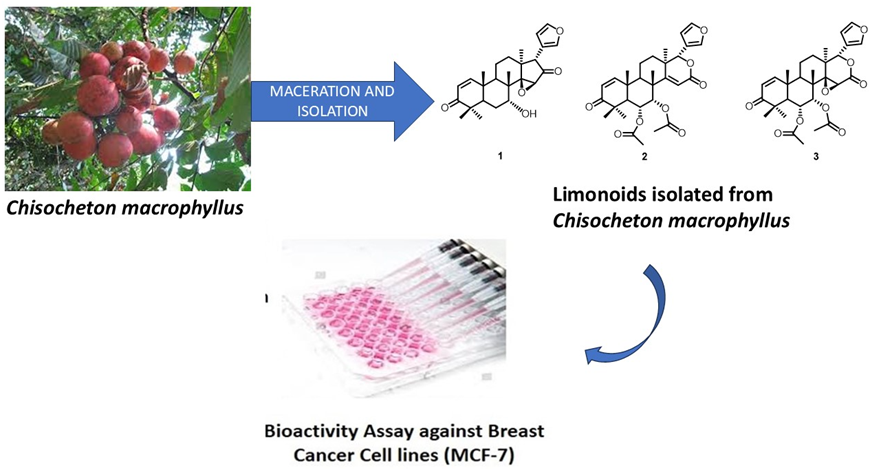
Meliaceae family consists of various plants that have been used as a source of medicinal plants and limonoid compounds. Limonoid is a compound group of terpenoid which has many bioactivities. One of the species from Meliaceae which produced limonoids is Chisocheton macrophyllus. The n-hexane fraction of the C. macrophyllus seed afforded 3 limonoids, namely 7-deacetylepoxyazadiradione (1), 6α-acetoxydeoxygedunin (2) and 6α-acetoxygedunin (3). Data from a variety of spectroscopic techniques, including as mass spectroscopy, FTIR, 1- and 2-dimensional NMR, were used to identify the secondary metabolite structures, and the PrestoBlueTM, method was used for the cytotoxic assay. Compound (2), known as 6α-acetoxydeoxygedunin, was first reported in this genus. MCF-7 breast cancer cell lines have also been used to assess limonoids compounds (1 - 3). The 2 and 3 were categorized as weak with an IC50 value of 231.26 and 475.87 µg/mL, while compound 1 showed inactive with IC50 values of > 500 µg/mL. These findings encourage further exploration of Meliaceae plants for new bioactive compounds and advance the development of novel cancer treatments.
HIGHLIGHTS
- The n-hexane fraction of the seeds of Chisocheton macrophyllus, yielded three limonoids, 7-deacetylepoxyazadiradione (1), 6α-acetoxydeoxygedunin (2), and 6α-acetoxygedunin (3).
- 6α-acetoxydeoxygedunin (2) is reported for the first time in this genus, expanding the known chemical diversity of Chisocheton
- The structures of these compounds were identified through advanced spectroscopic techniques, including mass spectrometry, FTIR, and one- and two-dimensional NMR.
- Cytotoxicity tests against MCF-7 breast cancer cells showed that compounds 2 and 3 were weakly active with IC50 values of 231.26 μg/mL and 475.87 μg/mL, respectively, while compound 1 was inactive with an IC50 value >500 μg/mL.
GRAPHICAL ABSTRACT
Downloads
References
GM Happi and R Teufel. Steroids from the Meliaceae family and their biological activities. Phytochemistry 2024; 221, 114039.
AT Hidayat, K Farabi, D Harneti, R Maharani, N Darwati, T Mayanti and Y Shiono. Cytotoxicity and structure-activity relationship of Dammarane-type triterpenoids from the bark of Aglaia elliptica against P-388 murine leukemia cells. Natural Product Sciences 2017; 23(4), 291-298.
MA Ngadni, SL Chong, H Hazni, N Asib, IH Ishak, NM Misnan and K Awang. Limonoids from the fruits of Chisocheton erythrocarpus and their mosquito larvicidal activities. Phytochemistry 2024; 222, 114092.
U Supratman, W Naibaho, S Salam, R Maharani, AT Hidayat, D Harneti and Y Shiono. Cytotoxic triterpenoids from the bark of Chisocheton patens Blume (Meliaceae). Phytochemistry Letters 2019; 30, 81-87.
T Mayanti, SE Sinaga and U Supratman. Phytochemistry and biological activity of Lansium domesticum Corr. species: A review. Journal of Pharmacy and Pharmacology 2022; 74(11), 1568-1587.
F Mulani, S Nandikol and H Thulasiram. Chemistry and biology of novel Meliaceae limonoids. Biological and Medicinal Chemistry 2022. http://dx.doi.org/10.26434/chemrxiv-2022-2bpb9-v2
S Fu and B Liu. Recent progress in the synthesis of limonoids and limonoid-like natural products. Organic Chemistry Frontiers 2020; 7(14), 1903-1947.
P Zhang, S Xue, W Huang, C Wang, Z Cui, J Luo and L Kong. Diverse prieurianin-type limonoids with oxygen-bridged caged skeletons from two Aphanamixis species: Discovery and biomimetic conversion. Organic Chemistry Frontiers 2021: 8(3), 566-571.
MJ Durán-Peña, JM Botubol-Ares, IG Collado and R Hernandez-Galán. Degraded limonoids: Biologically active limonoid fragments re-enhancing interest in Meliaceae and Rutaceae sources. Phytochemistry Reviews 2023; 22(3), 695-741.
TM Braga, L Rocha, TY Chung, RF Oliveira, C Pinho, AI Oliveira and A Cruz. Biological activities of gedunin - A limonoid from the Meliaceae family. Molecules 2020; 25(3), 493.
W Fan, L Fan, Z Wang and L Yang. Limonoids from the genus Melia (Meliaceae): Phytochemistry, synthesis, bioactivities, pharmacokinetics, and toxicology. Frontiers in Pharmacology 2022; 12, 795565.
TN Tan, HT Trung, Q Le Dang, HV Thi, HD Vu, TN Ngoc, HT Thi Do, TH Nguyen, DN Quang and TT Dinh. Characterization and antifungal activity of limonoid constituents isolated from Meliaceae plants Melia dubia, Aphanamixis polystachya, and Swietenia macrophylla against plant pathogenic fungi in vitro. Journal of Chemistry 2021; 2021, 4153790.
JA Shilpi, S Saha, SL Chong, L Nahar, SD Sarker and K Awang. Advances in chemistry and bioactivity of the genus Chisocheton Blume. Chemistry & Biodiversity 2016; 13(5), 483-503.
Supriatno, Nurlelasari, T Herlina, D Harneti, R Maharani, AT Hidayat and Y Shiono. A new limonoid from stem bark of Chisocheton pentandrus (Meliaceae). Natural Product Research 2018; 32(21), 2610-2616.
K Mohamad, Y Hirasawa, CS Lim, K Awang, AHA Hadi, K Takeya and H Morita. Ceramicine A and walsogyne A, novel limonoids from two species of Meliaceae. Tetrahedron Letters 2008; 49(27), 4276-4278.
W Maneerat, S Laphookhieo, S Koysomboon and K Chantrapromma. Antimalarial, antimycobacterial and cytotoxic limonoids from Chisocheton siamensis. Phytomedicine 2008; 15(12), 1130-1134.
K Awang, CS Lim, K Mohamad, H Morita, Y Hirasawa, K Takeya, O Thoison and AHA Hadi. Erythrocarpines A-E, new cytotoxic limonoids from Chisocheton erythrocarpus. Bioorganic & Medicinal Chemistry 2007; 15(17), 5997-6002.
MH Yang, JS Wang, JG Luo, XB Wang and LY Kong. Tetranortriterpenoids from Chisocheton paniculatus. Journal of Natural Products 2009; 72(11), 2014-2018.
IA Najmuldeen, AHA Hadi, K Awang, K Mohamad and SW Ng. 14-Deoxyxyloccensin K from Chisocheton ceramicus (Meliaceae). Acta Crystallographica Section E: Structure Reports Online 2010; 66(8), o1927-o1927.
Nurlelasari, I Rahmayanti, S Salam, A Safari, D Harneti, R Maharani and U Supratman. A new havanensin-type limonoid from Chisocheton macrophyllus. Applied Biological Chemistry 2021; 64, 35.
KC Murthy, GK Jayaprakasha, S Safe and BS Patil. Citrus limonoids induce apoptosis and inhibit the proliferation of pancreatic cancer cells. Food & Function 2021; 12(3), 1111-1120.
S Shaheen, S Khalid, K Aaliya, A Gul, A Hafeez, M Armaghan and J Sharifi-Rad. Insights into Nimbolide molecular crosstalk and its anticancer properties. Medical Oncology 2024; 41(6), 158.
F Bray, M Laversanne, H Sung, J Ferlay, RL Siegel, I Soerjomataram and A Jemal. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Elinicians 2024; 74(3), 229-263.
SE Sinaga, M Fajar, T Mayanti and U Supratman. Bioactivities screening and elucidation of terpenoid from the stem bark extracts of Lansium domesticum Corr. cv. Kokosan (Meliaceae). Sustainability 2023; 15(3), 2140.
PA Yadav, CP Kumar, B Siva, KS Babu, AD Allanki, PS Sijwali and AV Rao. Synthesis and evaluation of anti-plasmodial and cytotoxic activities of epoxyazadiradione derivatives. European Journal of Medicinal Chemistry 2017; 134, 242-257.
T Suzuki, NR Ariefta, T Koseki, H Furuno, E Kwon, H Momma, D Harneti, R Maharani, U Supratman, K Kimura and Y Shiono. New polyketides, paralactonic acids A - E produced by Paraconiothyrium sp. SW-B-1, an endophytic fungus associated with a seaweed, Chondrus ocellatus Holmes. Fitoterapia 2019; 132, 75-81.
Y Tanaka, T Yamada, Y In, O Muraoka, T Kajimoto and R Tanaka. Absolute stereostructure of Andirolides A - G from the flower of Carapa guianensis (Meliaceae). Tetrahedron 2011; 67(4), 782-792.
Y Widiyastuti, IYM Sholikhah and S Haryanti. Cytotoxic activities of ethanolic and dichloromethane extract of leaves, stems, and flowers of Jarong [Stachytarpheta jamaicensis (L.) Vahl.] on HeLa and T47D cancer cell line. AIP Conference Proceedings 2019; 2202, 020101.
RH Shoemaker. The NCI60 human tumour cell line anticancer drug screen. Nature Reviews Cancer 2006; 6(10), 813-823.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






