Ovalbumin-Induced Chronic Asthma in Female Wistar Rats: Eosinophilic Infiltration, IL-5 Profiles and Bronchial Histopathology
DOI:
https://doi.org/10.48048/tis.2025.9768Keywords:
Chronic asthma, Eosinophil, Histopathology, Interleukin-5, OvalbuminAbstract
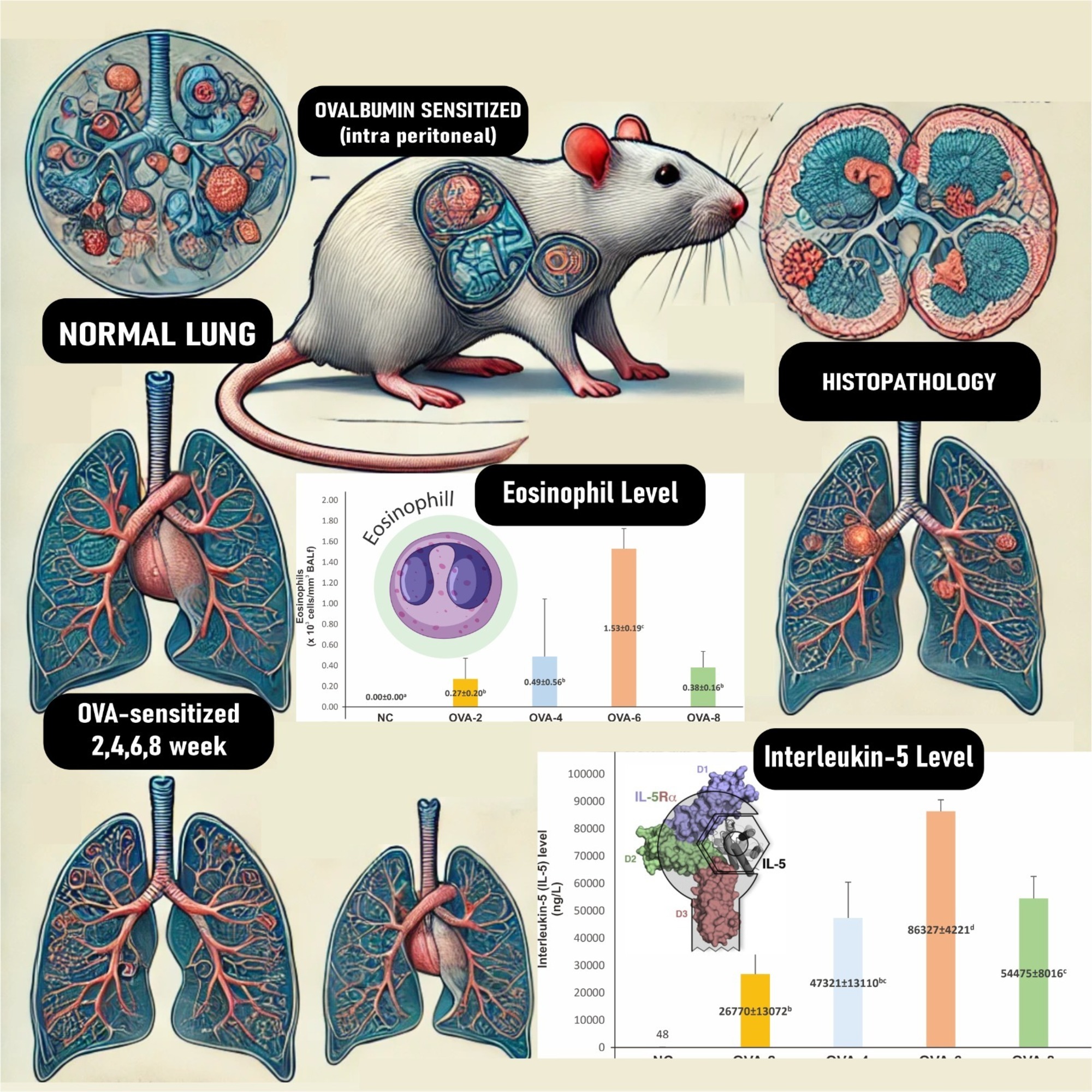
Asthma, a chronic inflammatory airway disease, is marked by bronchial hyper responsiveness, airway inflammation, and remodeling, leading to symptoms such as wheezing, chest tightness, and cough, which may result in permanent lung function impairment. This study aimed to investigate the progressive effects of ovalbumin (OVA) sensitization on eosinophil levels, interleukin-5 (IL-5) concentrations, and bronchial histopathology in a rat model of asthma. Female Wistar rats were divided into 5 groups: A control group and 4 OVA-sensitized groups (2, 4, 6, and 8 weeks) via intraperitoneal injection and inhalation at varying frequencies. Eosinophil levels, IL-5 concentrations, and bronchial histopathological changes were assessed. Eosinophil levels and IL-5 concentrations varied significantly among groups, with OVA sensitization leading to elevated levels compared to the Normal Control (NC) group. The highest eosinophil and IL-5 levels were observed in the 6 weeks (OVA-6 group), indicating pronounced airway inflammation at 6 weeks post-OVA induction (p < 0.05). Histopathological analysis indicated progressive bronchial damage in OVA-exposed groups, with severe structural changes such as septal thickening and lymphocyte infiltration peaking at 6 weeks (OVA-6) and slightly reduced at 8 weeks (OVA-8). OVA sensitization induces time-dependent increases in eosinophil levels, IL-5 concentrations, and bronchial remodeling, with peak inflammation at 6 weeks. IL-5 plays a pivotal role in regulating eosinophilic activity and airway inflammation. These findings underscore the therapeutic potential of targeting IL-5 during peak inflammation to mitigate chronic asthma symptoms and prevent long-term airway remodeling.
HIGHLIGHTS
- Time-dependent effects of ovalbumin sensitization on eosinophils, IL-5, and bronchial remodeling identified.
- Peak inflammation at 6 weeks marked by highest eosinophil levels and IL-5 concentrations.
- Severe bronchial structural changes peak at 6 weeks, slightly improving at 8 weeks.
- IL-5 identified as a key target for reducing eosinophilic inflammation in asthma.
- Emphasizes timing's importance in asthma treatment to prevent chronic disease progression.
GRAPHICAL ABSTRACT
Downloads
References
MI Asher, L Garcia-Marcos, NE Pearce and DP Strachan. Trends in worldwide asthma prevalence. European Respiratory Journal 2020; 56(6), 2002094.
Global Initiative for Asthma. Available at: https://ginasthma.org/reports/, accessed October 2023.
PJ Barnes. Pathophysiology of asthma. British Journal of Clinical Pharmacology 1996; 42(1), 3-10.
P Gao, PG Gibson, KJ Baines, IA Yang, JW Upham, PN Reynolds, S Hodge, AL James, C Jekins, MJ Peters, J Zhang and JL Simpson. Anti-inflammatory deficiencies in neutrophilic asthma: Reduced galectin-3 and IL-1RA/IL-1β. Respiratory Research 2015; 16(1), 5.
KW McGrath, N Icitovic, HA Boushey, SC Lazarus, ER Sutherland, VM Chinchilli and JV Fahy. A large subgroup of mild-to-moderate asthma is persistently noneosinophilic. American Journal of Respiratory and Critical Care Medicine 2012; 185(6), 612-619.
MG Belvisi. Regulation of inflammatory cell function by corticosteroids. Proceedings of the American Thoracic Society 2004; 1(3), 207-214.
JC Renauld. New insights into the role of cytokines in asthma. Journal of Clinical Pathology 2001; 54(8), 577-589.
M Lee, C Seo, N Lee, H Ha, J Lee, H Lee, K Lee and H Shin. Anti-asthmatic effect of schizandrin on OVA-induced airway inflammation in a murine asthma model. International Immunopharmacology 2010; 10(11), 1374-1379.
TSC Hinks, SJ Levine and GG Brusselle. Treatment options in type-2 low asthma. European Respiratory Journal 2021; 57(1), 2000528.
B Leon and A Ballesteros-Tato. Modulating Th2 cell immunity for the treatment of asthma. Frontiers in Immunology 2021; 12, 637948.
HJ Park, E Oh, YH Park, M Yang, KH Park, J Park and J Lee. Potential of serum soluble CD93 as a biomarker for asthma in an ovalbumin-induced asthma murine model. Biomarkers 2018; 23(5), 446-452.
GF Pavon-Romero, NH Serrano-Perez, L Garcia-Sanchez, F Ramirez-Jimenez and LM Teran. Neuroimmune pathophysiology in asthma. Frontiers in Cell and Developmental Biology 2021; 9, 663535.
C Pelaia, C Crimi, A Vatrella, C Tinello, R Terracciano and G Pelaia. Molecular targets for biological therapies of severe asthma. Frontiers in Immunology 2020; 11, 603312.
G Gulati, G Uppal and J Gong. unreliable automated complete blood count results: Causes, recognition, and resolution. Annals of Laboratory Medicine 2022; 42(5), 515-530.
VR Thakur, V Khuman, JV Beladiya, KK Chaudagar and AA Mehta. An experimental model of asthma in rats using ovalbumin and lipopolysaccharide allergens. Heliyon 2019; 5(11), e02864.
H Ghobadi, F Aslani, MH Boskabady, S Saadat and MR Aslani. Saffron (crocus sativus) and its constituents in ovalbumin-induced asthma model: A preclinical systematic review and meta-analysis. Frontiers in Pharmacology 2024; 15, 1436295.
B Zhang, M Zeng, Q Zhang, R Wang, J Jia, B Cao, M Liu, P Guo, Y Zhang, X Zheng and W Feng. Ephedrae herba polysaccharides inhibit the inflammation of ovalbumin induced asthma by regulating Th1/Th2 and Th17/Treg cell immune imbalance. Molecular Immunology 2022; 152, 14-26.
MF Dogan, A Parlar, SA Cam, EM Tosun, F Uysal and SO Arslan. Glabridin attenuates airway inflammation and hyperresponsiveness in a mice model of ovalbumin-induced asthma. Pulmonary Pharmacology & Therapeutics 2020; 63, 101936.
S Azman, M Sekar, SR Bonam, SH Gan, S Wahidin, PT Lum and SB Dhadde. Traditional medicinal plants conferring protection against ovalbumin-induced asthma in experimental animals: A review. Journal of Asthma and Allergy 2021; 14, 641-662.
X Han, S Hu, Q Yang, X Sang, D Tang and G Cao. Paeoniflorin ameliorates airway inflammation and immune response in ovalbumin induced asthmatic mice: From oxidative stress to autophagy. Phytomedicine 2022; 96, 153835.
C Lin, K Chuang, S Chen, Y Chao, C Yen, S Yang, W Chen, K Chang, Y Chang and C Chen. Lactoferrin ameliorates ovalbumin-induced asthma in mice through reducing dendritic-cell-derived Th2 cell responses. International Journal of Molecular Sciences 2022; 23(22), 14185.
S Maltby, HL Tay, M Yang and PS Foster. Mouse models of severe asthma: U nderstanding the mechanisms of steroid resistance, tissue remodelling and disease exacerbation. Respirology 2017; 22(5), 874-885.
M Periz, FJ Perez-Cano, MJ Rodriguez-Lagunas, T Cambras, S Pastor-Soplin, I Best, M Castell and M Massot-Cladera. Development and characterization of an allergic asthma rat model for interventional studies. International Journal of Molecular Sciences 2020; 21(11), 3841.
IP Shilovskiy, MS Sundukova, AA Babakhin, AR Gaisina, AV Maerle, IV Sergeev, A A Nikolskiy, ED Barvinckaya, VI Kovchina, DA Kudlay, AA Nikonova and MR Khaitov. Experimental protocol for development of adjuvant-free murine chronic model of allergic asthma. Journal of Immunological Methods 2019; 468, 10-19.
J Temelkovski, SP Hogan, DP Shepherd, PS Foster and RK Kumar. An improved murine model of asthma: Selective airway inflammation, epithelial lesions and increased methacholine responsiveness following chronic exposure to aerosolised allergen. Thorax 1998; 53(10), 849-856.
JS Woodrow, MK Sheats, B Cooper and R Bayless. Asthma: The use of animal models and their translational utility. Cells 2023; 12(7), 1091.
JV Beladiya, KK Chaudagar and AA Mehta. A protective role of Gαq-RGS2 loop activator on streptozotocin induced diabetic complications in rats: An independent on elevated serum glucose level modulation. European Journal of Pharmacology 2018; 818, 141-147.
RK Nelson, A Bush, J Stokes, P Nair and P Akuthota. Eosinophilic asthma. The Journal of Allergy and Clinical Immunology: In Practice 2020; 8(2), 465-473.
Gevaert, E. (2022). Eosinophils. In: L Zhang and C Bachert (Eds.). Chronic rhinosinusitis. Springer, Singapore.
L George and CE Brightling. Eosinophilic airway inflammation: Role in asthma and chronic obstructive pulmonary disease. Therapeutic Advances in Chronic Disease 2016; 7(1), 34-51.
S Makino, T Fukuda, S Motojima and T Yukawa. Eosinophils in airway hyperresponsiveness. CRC Press, Florida, 2020.
U Shapira, M Krubiner, M Ehrenwald, I Shapira, D Zeltser, S Berliner, O Rogowski, S Shenhar-Tsarfaty and A Bar-Shai. Eosinophil levels predict lung function deterioration in apparently healthy individuals. International Journal of Chronic Obstructive Pulmonary Disease 2019; 14, 597-603.
A Voskamp, TG Kormelink, RGV Wijk, P Hiemstra, C Taube, ECD Jong and HH Smits. Modulating local airway immune responses to treat allergic asthma: Lessons from experimental models and human studies. Seminars in Immunopathology 2020; 42(1), 95-110.
R AbuJabal, RK Ramakrishnan, K Bajbouj and Q Hamid. Role of IL-5 in asthma and airway remodelling. Clinical & Experimental Allergy 2024; 54(8), 538-549.
HK Kandikattu, SU Venkateshaiah and A Mishra. Synergy of interleukin (IL)-5 and IL-18 in eosinophil mediated pathogenesis of allergic diseases. Cytokine & Growth Factor Reviews 2019; 47, 83-98.
P Ilmarinen, E Moilanen and H Kankaanranta. Regulation of spontaneous eosinophil apoptosis - A neglected area of importance. Journal of Cell Death 2014; 7, 1-9.
MW Johansson. Eosinophil activation status in separate compartments and association with asthma. Frontiers in Medicine 2017; 4, 75.
GG Brusselle, T Maes and KR Bracke. Eosinophils in the spotlight: Eosinophilic airway inflammation in nonallergic asthma. Nature Medicine 2013; 19(8), 977-979.
JA Walker, JL Barlow and AN McKenzie. Innate lymphoid cells - How did we miss them? Nature Reviews Immunology 2013; 13(2), 75-87.
T Marichal, C Mesnil and F Bureau. Homeostatic eosinophils: Characteristics and functions. Frontiers in Medicine 2017; 4, 101.
SW Park, DJ Kim, HS Chang, SJ Park, YM Lee, JS Park, IY Chung, JH Lee and C Park. Association of interleukin-5 and eotaxin with acute exacerbation of asthma. International Archives of Allergy and Immunology 2003; 131(4), 283-290.
K Antosz, J Batko, M Blazejewska, A Gawor, J Sleziak and K Gomulka. Insight into IL-5 as a potential target for the treatment of allergic diseases. Biomedicines 2024; 12(7), 1531.
M Nagata and K Nakagome. Allergen immunotherapy in asthma: Current status and future perspectives. Allergology International 2010; 59(1), 15-59.
CA Akdis and M Akdis. Mechanisms of allergen-specific immunotherapy and immune tolerance to allergens. World Allergy Organization Journal 2015; 8(1), 17.
K Kusmardi, E Wiyarta, NK Rusdi, AM Maulana, A Estuningtyas and H Sunaryo. The potential of lunasin extract for the prevention of breast cancer progression by upregulating E-Cadherin and inhibiting ICAM-1. F1000Research 2021; 10, 902.
JS Woodrow, MK Sheats, B Cooper and RA Bayless. The use of animal models and their translational utility. Cells 2023; 12(7), 1091.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






