Cisplatin-Induced Oral Mucositis Prevention: Nigella sativa’s Anti-Inflammatory Role through NFκB Pathway
DOI:
https://doi.org/10.48048/tis.2024.8105Keywords:
Nigella sativa, Cisplatin, Oral mucositis, NFκB pathway, InflammationAbstract
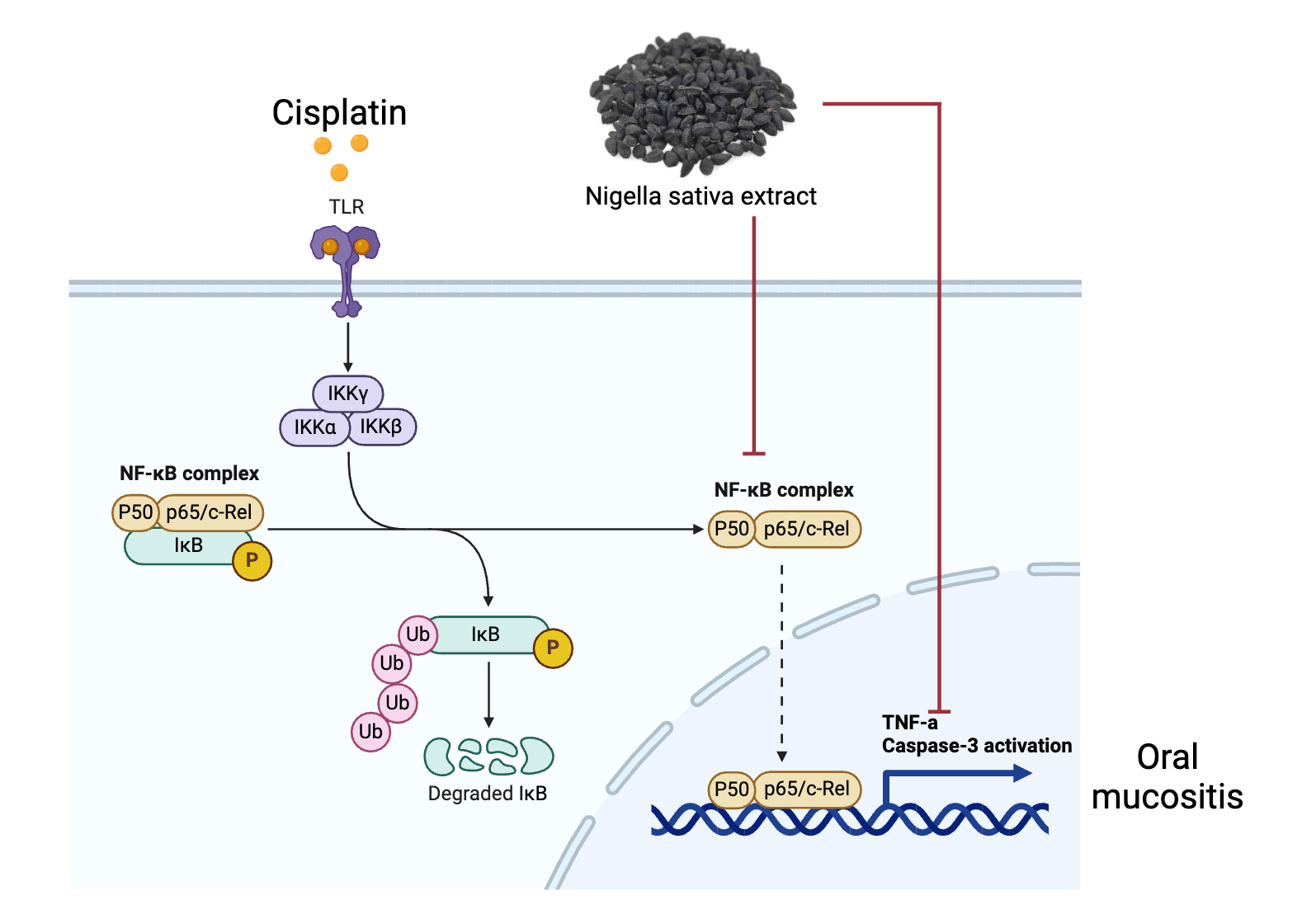
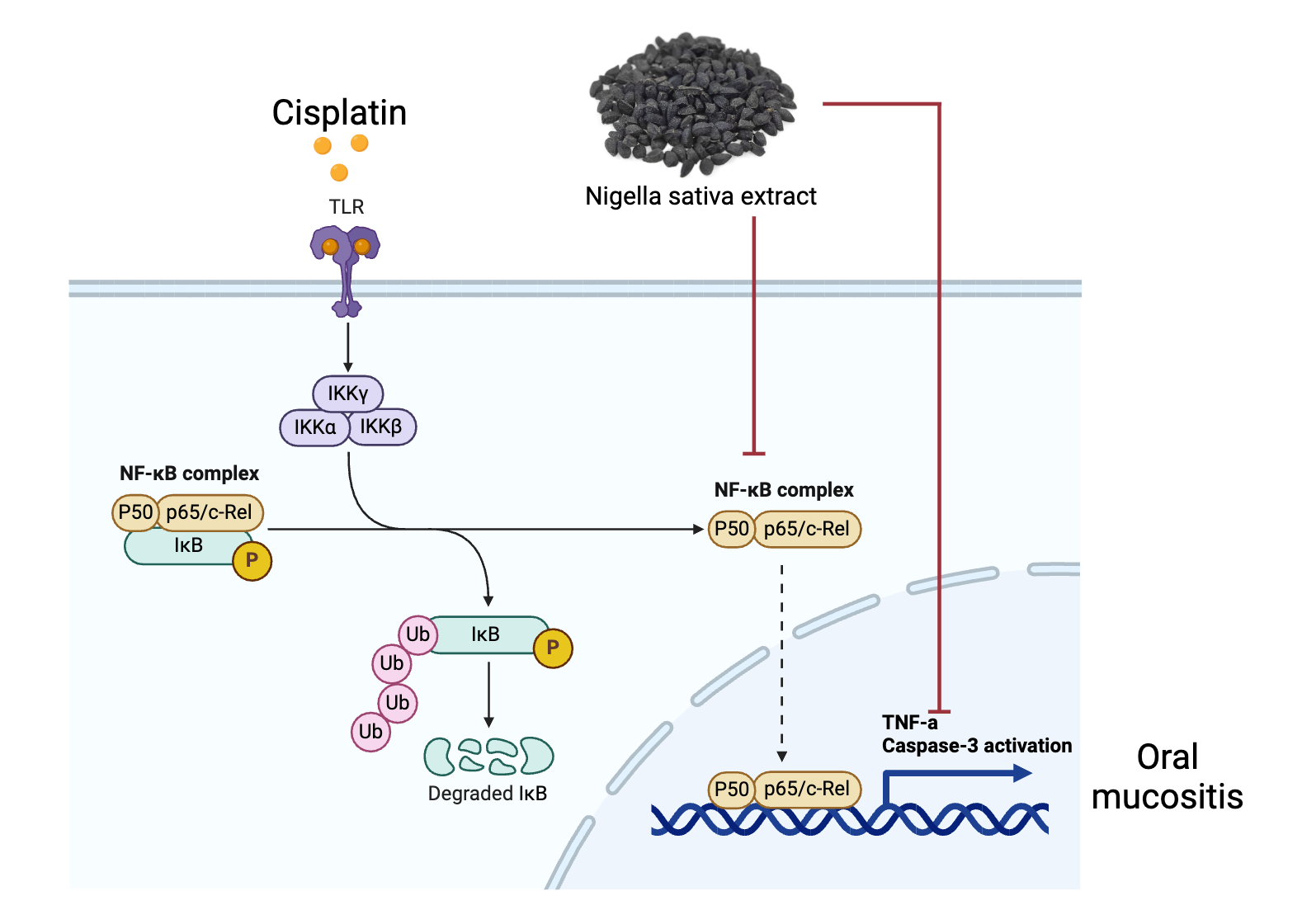
Prevention of Cisplatin-induced oral mucositis (OM) Presents substantial challenges due to its association with oxidative stress, inflammation, apoptosis and NFκB pathway activation. This study aims to evaluate the potential of Nigella sativa (NS) extract in inhibiting a cisplatin-induced oral mucositis. Cisplatin-induced oral mucositis was modeled in experimental groups treated with varying doses of NS extract (125, 250 and 500 mg/kg BW), compared to negative controls and N-acetylcysteine (NAC) as a positive control. Expression levels of tumor necrosis factor-alpha (TNF-α) were analysed under Western blot analysis, the p50 and p65 gene expression level was determined by qRT-PCR analysis. NS extract notably inhibited TNF-α expression compared to the negative control group, akin to the positive control (NAC). Additionally, NS extract exhibited a dose-dependent regulation of NFκB pathway subunits (p50 and p65) towards levels closer to the baseline, indicating its potential in modulating the inflammatory pathway. The findings suggest that NS extract possesses promising therapeutic potential in mitigating inflammation and NFκB pathway activation in cisplatin-induced oral mucositis.
HIGHLIGHTS
As far as we have seen, similar studies in experimental animals using Nigella sativa oil related to histopathological findings of oral mucositis due to cisplatin. This study demonstrated Nigella sativa (NS) extract’s potential in mitigating cisplatin-induced oral mucositis (OM) by inhibiting TNF-α and modulating NFκB pathway subunits (p50 and p65). This highlights NS extract as a promising therapeutic agent for OM associated with cisplatin treatment.
GRAPHICAL ABSTRACT
Downloads
References
C Pulito, A Cristaudo, CL Porta, S Zapperi, G Blandino, A Morrone and S Strano. Oral mucositis: The hidden side of cancer therapy. J. Exp. Clin. Canc. Res. 2020; 39, 210.
D Jicman, MI Sârbu, S Fotea, A Nechifor, G Bălan, M Anghele, CI Vasile, E Niculeț, N Sârbu, LF Rebegea and AL Tatu. Oral mucositis induced by chemoradiotherapy in head and neck cancer - a short review about the therapeutic management and the benefits of bee honey. Medicina 2022; 58, 751.
SS Shetty, M Maruthi, V Dhara, JAA de Arruda, LG Abreu, RA Mesquita, AL Teixeira, TA Silva and Y Merchant. Oral mucositis: Current knowledge and future directions. Disease-a-Month 2022; 68, 101300.
A Blakaj, M Bonomi, ME Gamez and DM Blakaj. Oral mucositis in head and neck cancer: Evidence-based management and review of clinical trial data. Oral Oncol. 2019; 95, 29-34.
SK Ps, A Balan, A Sankar and T Bose. Radiation induced oral mucositis. Indian J. Palliat. Care 2009; 15, 95-102.
VL Rajesh, TS Stephen and EP Douglas. Management of oral mucositis in patients with cancer. Dent. Clin. North Am. 2008; 52, 61-8.
EU Cidon. Chemotherapy induced oral mucositis: Prevention is possible. Chin. Clin. Oncol. 2018; 7, 6.
NT Hasanah and TS Dewi. Holistic approach of a leukemic child suffering from oral mucositis with coinfections: A case report. Int. Med. Case Rep. J. 2023; 16, 363-9.
S Elad, N Yarom, Y Zadik, M Kuten‐Shorrer and ST Sonis. The broadening scope of oral mucositis and oral ulcerative mucosal toxicities of anticancer therapies. CA: Can. J. Clin. 2022; 72, 57-77.
AP Cotrim, M Yoshikawa, AN Sunshine, C Zheng, AL Sowers, AD Thetford, JA Cook, JB Mitchell and BJ Baum. Pharmacological protection from radiation ± cisplatin-induced oral mucositis. Int. J. Radiat. Oncol. Biol. Phys. 2012; 83, 1284-90.
Z Li, C Li, D Yang, J Song, T Liu, Z Zhou, L Zhou and M Kang. Comparing the efficacy and safety of cisplatin and other platinum-based chemotherapies in locally advanced nasopharyngeal carcinoma: A systematic review and meta-analysis. BMC Cancer 2022; 22, 616.
S Mirzaei, K Hushmandi, A Zabolian, H Saleki, SMR Torabi, A Ranjbar, SH SeyedSaleh, SO Sharifzadeh, H Khan, M Ashrafizadeh, A Zarrabi and KS Ahn. Elucidating role of Reactive Oxygen Species (Ros) in cisplatin chemotherapy: A focus on molecular pathways and possible therapeutic strategies. Molecules 2021; 26, 2382.
S Giridharan and M Srinivasan. Mechanisms of NF-κB p65 and strategies for therapeutic manipulation. J. Inflamm. Res. 2018; 11, 407-19.
MJ Morgan and ZG Liu. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2011; 21, 103-15.
H Nguyen, S Sangha, M Pan, DH Shin, H Park, AI Mohammed and N Cirillo. Oxidative stress and chemoradiation-induced oral mucositis: A scoping review of in vitro, in vivo and clinical studies. Int. J. Mol. Sci. 2022; 23, 4863.
C Chen, Q Zhang, W Yu, B Chang and AD Le. Oral mucositis: An update on innate immunity and new interventional targets. J. Dent. Res. 2020; 99, 1122-30.
OK Eğilmez, N Kökten, MT Kalcıoğlu, AID Ekici, S Şerifler and E Yeşilada. Investigation of the protective effect of Nigella sativa oil in cisplatin induced oral mucositis: An experimental study. Turk. Arch. Otorhinolaryngol. 2020; 58, 10-5.
HAM Ameen, MO Mohammed, RHG Ali, KM Ahmed and SA Hussain. Nigella sativa oil for oral mucositis. Al Mustansiriyah J. Pharmaceut. Sci. 2019; 19, 54-67.
RM Pop, O Sabin, Ș Suciu, SC Vesa, SA Socaci, VS Chedea, IC Bocsan and AD Buzoianu. Nigella sativa’s anti-inflammatory and antioxidative effects in experimental inflammation. Antioxidants 2020; 9, 921.
A Ahmad, A Husain, M Mujeeb, SA Khan, AK Najmi, NA Siddique, ZA Damanhouri and F Anwar. A review on therapeutic potential of Nigella sativa: A miracle herb. Asian Pac. J. Trop. Biomed. 2013; 3, 337-52.
M Dalli, O Bekkouch, SE Azizi, A Azghar, N Gseyra and B Kim. Phytochemistry and pharmacological activities: A review (2019 - 2021). Biomolecules 2022; 12, 20.
D Kadam and SS Lele. Extraction, characterization and bioactive properties of Nigella sativa seedcake. J. Food Sci. Tech. 2017; 54, 3936-47.
N Chehl, G Chipitsyna, Q Gong, CJ Yeo and HA Arafat. Anti-inflammatory effects of the Nigella sativa seed extract, thymoquinone, in pancreatic cancer cells. HPB 2009; 11, 373-81.
ND Amalina, S Wahyuni and Harjito. Cytotoxic effects of the synthesized Citrus aurantium peels extract nanoparticles against MDA-MB-231 breast cancer cells. J. Phys. Conf. Ser. 2021; 1918, 032006.
M Suzery, B Cahyono and ND Amalina. Antiproliferative and apoptosis effect of hyptolide from Hyptis pectinata (L.) Poit on human breast cancer cells. J. Appl. Pharmaceut. Sci. 2020; 10, 001-6.
Y Shimamura, I Takeuchi, H Terada and K Makino. A mouse model for oral mucositis induced by cancer chemotherapy. Anticancer Res. 2018; 38, 307-12.
RI Jenie, ND Amalina, GPN Ilmawati, RY Utomo, M Ikawati, A Khumaira, JY Kato and E Meiyanto. Cell cycle modulation of CHO-K1 cells under genistein treatment correlates with cells senescence, apoptosis and ROS level but in a dose-dependent manner. Adv. Pharmaceut. Bull. 2019; 9, 453.
DA Paramita, D Hermansyah, DA Paramita and ND Amalina. Regulation of p53 and surviving by Curcuma longa extract to caspase-3 dependent apoptosis in triple negative breast cancer cells. Med. Glasnik 2022; 19, 189-96.
S Mursiti, ND Amalina and A Marianti. Inhibition of breast cancer cell development using Citrus maxima extract through increasing levels of Reactive Oxygen Species (ROS). J. Phys. Conf. Ser. 2021; 1918, 052005.
ND Amalina, S Wahyuni and Harjito. Cytotoxic effects of the synthesized Citrus aurantium peels extract nanoparticles against MDA-MB-231 breast cancer cells. J. Phys. Conf. Ser. 2021; 1918, 032006.
D Hermansyah, A Putra, D Munir, A Lelo, ND Amalina and I Alif. Synergistic effect of Curcuma longa extract in combination with Phyllanthus niruri extract in regulating annexin A2, epidermal growth factor receptor, matrix metalloproteinases, and pyruvate kinase M1/2 signaling pathway on breast cancer stem cell. Open Access Macedonian J. Med. Sci. 2021; 9, 271-85.
ND Amalina, IA Salsabila, UM Zulfin, RI Jenie and E Meiyanto. In vitro synergistic effect of hesperidin and doxorubicin downregulates epithelial-mesenchymal transition in highly metastatic breast cancer cells. J. Egypt. Natl. Canc. Inst. 2023; 35, 6.
A Utami, A Putra, JW Wibowo, ND Amalina and RCS Irawan. Hypoxic secretome mesenchymal stem cells inhibiting interleukin-6 expression prevent oxidative stress in type 1 diabetes mellitus. Med. Glasnik 2023; 20, 148-55.
N Ozawa, T Onda, K Hayashi, H Honda and T Shibahara. Effects of topical hangeshashinto (TJ-14) on chemotherapy-induced oral mucositis. Canc. Manag. Res. 2020; 12, 1069-78.
KF Shad, W Soubra and DJ Cordato. The role of thymoquinone, a major constituent of Nigella sativa, in the treatment of inflammatory and infectious diseases. Clin. Exp. Pharmacol. Physiol. 2021; 48, 1445-53.
L Peng, A Liu, Y Shen, HZ Xu, SZ Yang, XZ Ying, W Liao, HX Liu, ZQ Lin, QY Chen, SW Cheng and WD Shen. Antitumor and anti-angiogenesis effects of thymoquinone on osteosarcoma through the NF-κB pathway. Oncol. Rep. 2013; 29, 571-8.
MS Butt, M Imran, A Imran, MS Arshad, F Saeed, TA Gondal, MA Shariati, SA Gilani, T Tufail, I Ahmad, NA Rind, MF Mahomoodally, S Islam and Z Mehmood. Therapeutic perspective of thymoquinone: A mechanistic treatise. Food Sci. Nutr. 2021; 9, 1792-809.
G Sethi, KS Ahn and BB Aggarwal. Targeting nuclear factor-κB activation pathway by thymoquinone: Role in suppression of antiapoptotic gene products and enhancement of apoptosis. Mol. Canc. Res. 2008; 6, 1059-70.
AFC Silva, PI Haris, ML Serralheir and R Pacheco. Mechanism of action and the biological activities of Nigella sativa oil components. Food Biosci. 2020; 38, 100783.
OK Eğilmez, N Kökten, MT Kalcıoğlu, AID Ekici, S Şerifle and E Yeşilada. Investigation of the protective effect of Nigella sativa oil in cisplatin induced oral mucositis: An experimental study. Turk. Arch. Otorhinolaryngol. 2020; 58, 10-5.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






