The Potential of Chromatographic and Chemometric Profiling as Early Detection Tools for Metabolic Disturbances in Third-Hand Smoke-Exposed Rats: An Exploratory Study
DOI:
https://doi.org/10.48048/tis.2025.10129Keywords:
Chromatography, Third-hand smoke, Chemometric analysis, Metabolic biomarkers, Toxicant exposure, Early detection, Exploratory studyAbstract
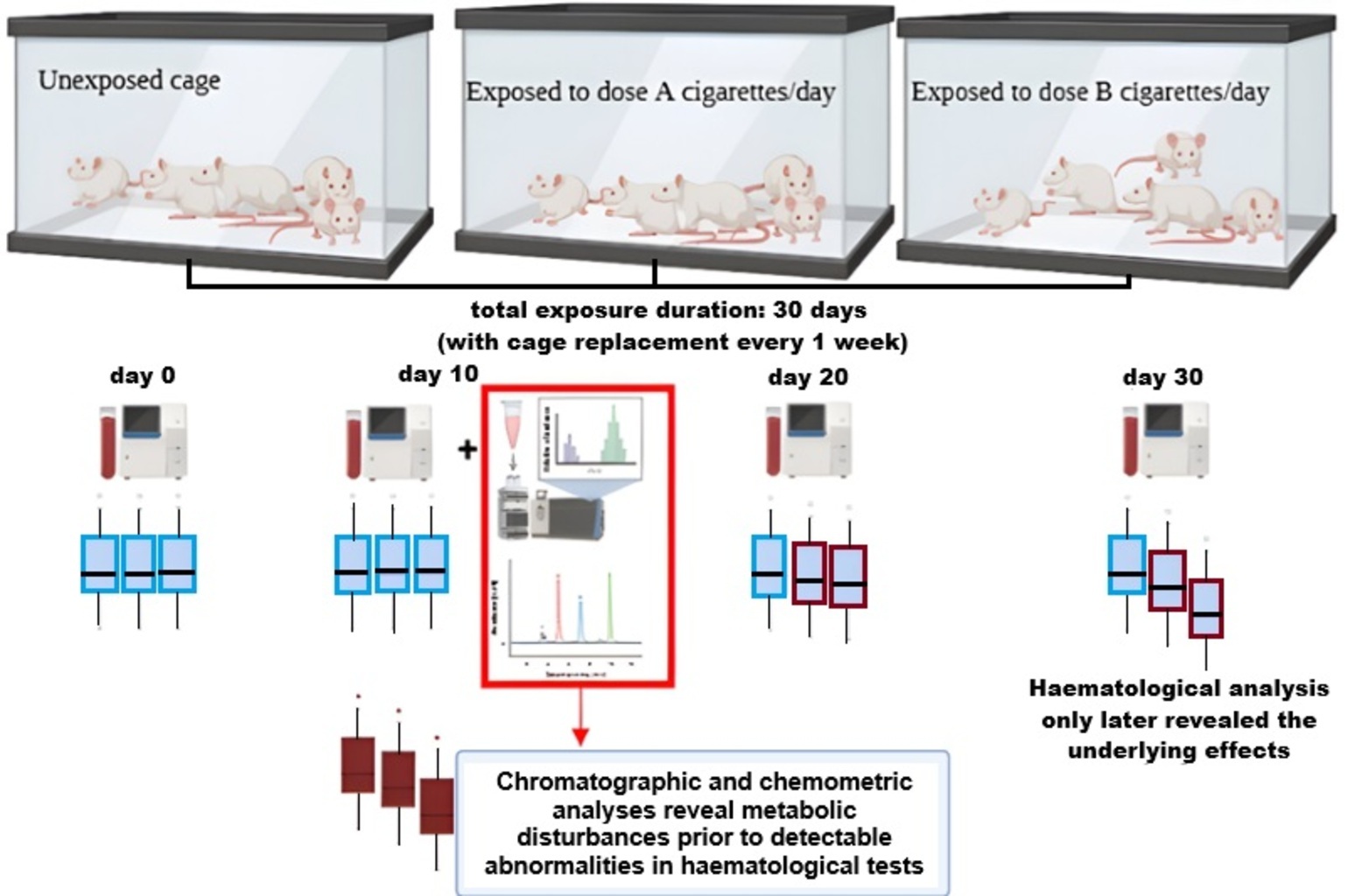
The application of chromatography in detecting toxicant-induced systemic alterations remains underexplored. Third-hand smoke (THS) exposure is increasingly recognised as a significant environmental health concern; however, its metabolic and haematological consequences are not yet fully characterised. This study investigates the potential of chromatographic profiling and chemometric analysis as tools for the early detection of metabolic disturbances induced by THS. Fifteen male Wistar rats were allocated to 3 groups: Control and THS exposure (12 or 24 cigarettes per day). Blood samples were collected at multiple time points for haematological analysis, while serum chromatography was conducted on day 10, and lung histology was assessed on day 30. Early metabolic disturbances were observed, characterised by the depletion of key amino acids (L-methionine, DL-lysine, DL-threonine, L-leucine, DL-arginine, and DL-carnitine) and elevated lipid biomarkers (palmitoyl sphingomyelin and phosphatidylcholine) in THS-exposed groups. These metabolic alterations preceded detectable haematological abnormalities. By day 30, haematological impairments and substantial pulmonary damage were evident. The depletion of essential amino acids and elevations in lipid biomarkers may serve as early indicators of toxicant-induced systemic effects. Chromatographic profiling detected metabolic perturbations prior to haematological and pulmonary impairments, highlighting its potential as an early toxicity detection tool. The findings of this exploratory study underscore the promise of chromatography for early toxicity detection, warranting further validation in larger cohorts.
HIGHLIGHTS
- Chromatographic profiling detects early metabolic disruptions before conventional haematological abnormalities manifest in THS-exposed rats.
- Depletion of key amino acids (L-methionine, DL-lysine, DL-threonine, L-leucine, DL-arginine, DL-carnitine), increased Phosphatidylcholine (significant) and Palmitoyl Sphingomyelin were observed on day 10, despite normal haematological parameters.
- Early elevation of phosphatidylcholine (significant) and palmitoyl sphingomyelin aligns with inflammasome activation (IL-18 elevation) and progressive lung damage, indicating their potential as biomarkers of third-hand smoke (THS)-induced toxicity.
- Routine haematological tests did not detect early systemic toxicity, reinforcing the need for chromatography-based biomonitoring for hidden toxicant exposure.
- Findings support the integration of chromatography into environmental toxicology frameworks for early detection of exposure-related diseases in at-risk populations.
GRAPHICAL ABSTRACT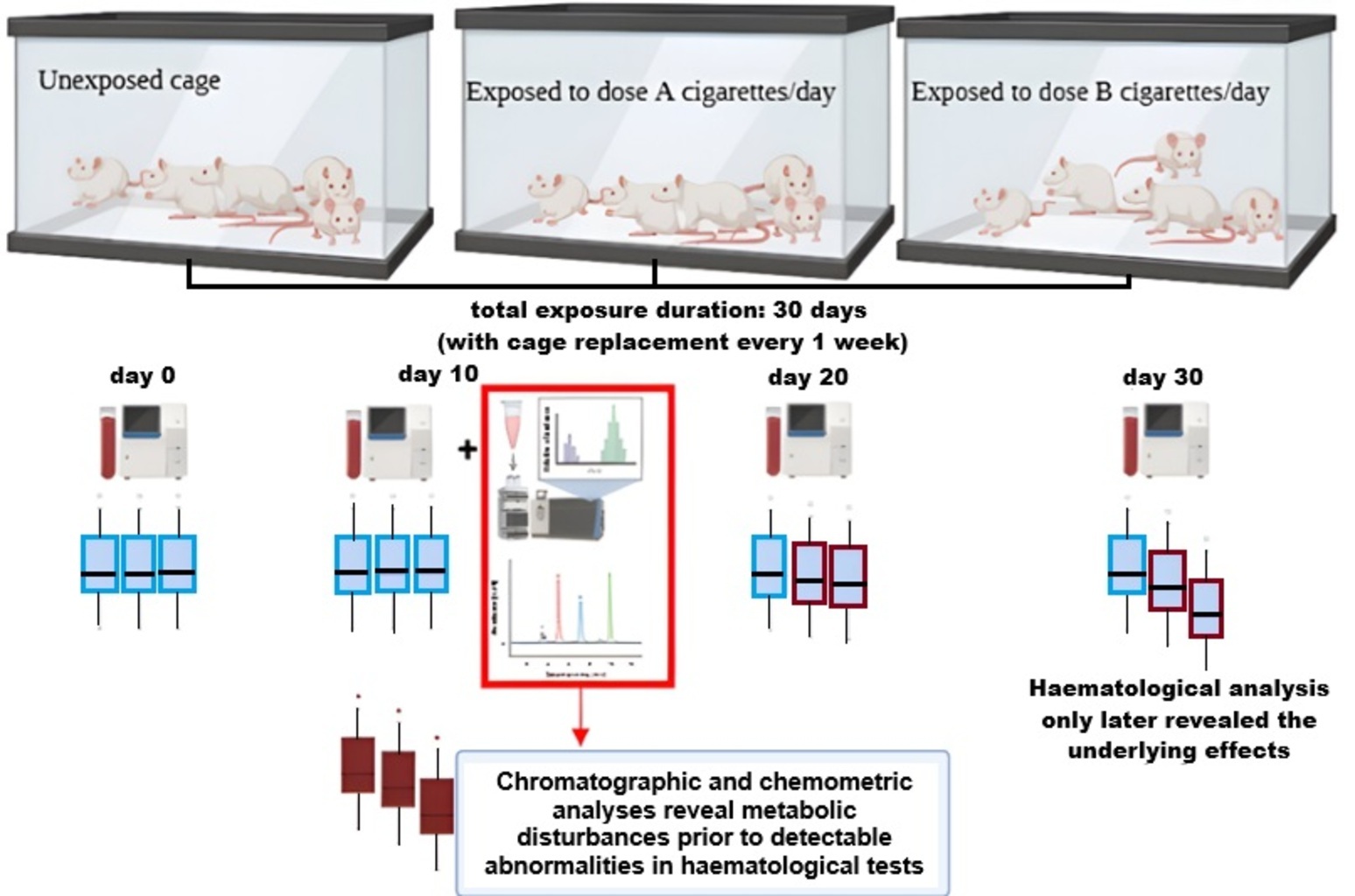
Downloads
References
P Jacob, NL Benowitz, H Destaillats, L Gundel, B Hang, M Martins-Green, GE Matt, PJE Quintana, JM Samet, SF Schick, P Talbot, N Aquilina, MF Hovell, JH Mao and TP Whitehead. Thirdhand smoke: New evidence, challenges, and future directions. Chemical Research in Toxicology 2017; 30(1), 270-294.
B Hang, AH Sarker, C Havel, S Saha, TK Hazra, S Schick, P Jacob, VK Rehan, A Chenna, D Sharan, M Sleiman, H Destaillats and LA Gundel. Thirdhand smoke causes DNA damage in human cells. Mutagenesis 2013; 28(4), 381-391.
M Martins-Green, N Adhami, M Frankos, M Valdez, B Goodwin, J Lyubovitsky, S Dhall, M Garcia, I Egiebor, B Martinez, HW Green, C Havel, L Yu, S Liles, G Matt, H Destaillats, M Sleiman, LA Gundel, N Benowitz, P Jacob, M Hovell, JP Winickoff and M Curras-Collazo. Cigarette smoke toxins deposited on surfaces: Implications for human health. PLoS One 2014; 9(1), e86391.
National Center for Biotechnology Information, Methanol. PubChem Compound Summary, Available at: https://pubchem.ncbi.nlm.nih.gov/compound/Methanol, Accessed March 2025.
M Fischer and TM Jefferies. Optimization of nicotine extraction from tobacco using supercritical fluid technology with dynamic extraction modeling. Journal of Agricultural and Food Chemistry 1996; 44(5), 1258-1264.
H Nerome, M Ito, S Machmudah, Wahyudiono, H Kanda and M Goto. Extraction of phytochemicals from saffron by supercritical carbon dioxide with water and methanol as entrainer. Journal of Supercritical Fluids 2016; 107, 377-383.
B Hang, AM Snijders, Y Huang, SF Schick, P Wang, Y Xia, C Havel, P Jacob, N Benowitz, H Destaillats, LA Gundel and JH Mao. Early exposure to third-hand cigarette smoke affects body mass and the development of immunity in mice. Scientific Reports 2017; 7, 41915.
A Windarsih, FDO Riswanto, NKA Bakar, ND Yuliana, Dachriyanus and A Rohman. Detection of pork in beef meatballs using LC-HRMS based untargeted metabolomics and chemometrics for halal authentication. Molecules 2022; 27(23), 8325.
TV Hoang, C Nardiello, DES Solaligue, JA Rodríguez-Castillo, P Rath, K Mayer, I Vadász, S Herold, K Ahlbrecht, W Seeger and RE Morty. Stereological analysis of individual lung lobes during normal and aberrant mouse lung alveolarisation. Journal of Anatomy 2018; 232(3), 472-484.
YH Chung, M Gulumian, RC Pleus and IJ Yu. Animal welfare considerations when conducting OECD test guideline inhalation and toxicokinetic studies for nanomaterials. Animals 2022; 12(23), 3305.
GE Matt, PJE Quintana and H Destaillats. Third-hand smoke: Chemical dynamics, exposure, and health effects. Environmental Research 2018. https://doi.org/10.3390/atmos16040370
JR Smith, ER Bolton and MR Dwinell. The rat: A model used in biomedical research. Methods in Molecular Biology 2019; 2018, 1-41.
EM Blais, KD Rawls, BV Dougherty, ZI Li, GL Kolling, P Ye, A Wallqvist and JA Papin. Reconciled rat and human metabolic networks for comparative toxicogenomics and biomarker predictions. Nature Communications 2017; 8, 14250.
D Villalobos-García, HEA Ali, AB Alarabi, MS El-Halawany, FZ Alshbool and FT Khasawneh. Exposure of mice to third-hand smoke modulates in vitro and in vivo platelet responses. International Journal of Molecular Sciences 2022; 23(10), 5595.
J Ho, K Koshibu, W Xia, K Luettich, A Kondylis, L Garcia, B Phillips, M Peitsch and J Hoeng. Effects of cigarette smoke exposure on a mouse model of multiple sclerosis. Toxicology Reports 2022; 9, 597-610.
M Sleiman, LA Gundel, JF Pankow, P Jacob, BC Singer and H Destaillats. Formation of carcinogens indoors by surface-mediated reactions of nicotine with nitrous acid, leading to potential third-hand smoke hazards. Proceedings of the National Academy of Sciences of the United States of America 2010; 107(15), 6576-6581.
SF Schick and SA Glantz. Thirdhand smoke: Here to stay. Tobacco Control 2011; 20(1), 1-3.
S Torres, C Merino, B Paton, X Correig and N Ramírez. Biomarkers of exposure to secondhand and thirdhand tobacco smoke: Recent advances and future perspectives. International Journal of Environmental Research and Public Health 2018; 15(12), 2693.
Y Tateishi, S Toyoda, H Murakami, R Uchida, R Ichikawa, T Kikuchi, W Sato and K Suzuki. A short-term intervention of ingesting iron along with methionine and threonine leads to a higher hemoglobin level than that with iron alone in young healthy women: A randomized, double-blind, parallel-group, comparative study. European Journal of Nutrition 2023; 62(7), 3009-3019.
JK Lugata, J Oláh, XE Ozsváth, R Knop, E Angyal and C Szabó. Effects of DL and L-methionine on growth rate, feather growth, and hematological parameters of tetra-SL layers from 1-28 days of age. Animals 2022; 12(15), 1928.
A Vlachos, E Atsidaftos, ML Lababidi, E Muir, ZR Rogers, W Alhushki, J Bernstein, B Glader, B Gruner, H Hartung, C Knoll, T Loew, G Nalepa, A Narla, AR Panigrahi, CA Sieff, K Walkovich, JE Farrar and JM Lipton. L-leucine improves anemia and growth in patients with transfusion-dependent Diamond Blackfan anemia: Results from a multicenter pilot phase I/II study from the Diamond Blackfan Anemia Registry. Pediatric Blood and Cancer 2020; 67(12), e28748.
M Malaguarnera, M Vacante, M Giordano, M Motta, G Bertino, M Pennisi, S Neri, M Malaguarnera, GL Volti and F Galvano. L-carnitine supplementation improves hematological pattern in patients affected by HCV treated with Peg interferon-α 2b plus ribavirin. World Journal of Gastroenterology 2011; 17(39), 4414-4420.
YA Hannun and LM Obeid. Sphingolipids and their metabolism in physiology and disease. Nature Reviews Molecular Cell Biology 2018; 19, 175-191.
X Rao, D Zhou, H Deng, Y Chen, J Wang, X Zhou, X Jie, Y Xu, Z Wu, G Wang, X Dong, S Zhang, R Meng, C Wu, S Xing, K Fan, G Wu and R Zhou. Activation of NLRP3 inflammasome in lung epithelial cells triggers radiation-induced lung injury. Respiratory Research 2023; 24(1), 25.
EH Koh, JE Yoon, MS Ko, J Leem, JY Yun, CH Hong, YK Cho, SE Lee, JE Jang, JY Baek, HJ Yoo, SJ Kim, CO Sung, JS Lim, WI Jeong, SH Back, IJ Baek, S Torres, E Solsona-Vilarrasa, LCDL Rosa, C Garcia-Ruiz, AE Feldstein, JC Fernandez-Checa and KU Lee. Sphingomyelin synthase 1 mediates hepatocyte pyroptosis to trigger non-alcoholic steatohepatitis. Gut 2021; 70(10), 1954-1964.
F Liu, Y Zhang, Y Shi, K Xiong, F Wang and J Yang. Ceramide induces pyroptosis through TXNIP/NLRP3/GSDMD pathway in HUVECs. BMC Molecular and Cell Biology 2022; 23, 54.
JNVD Veen, JP Kennelly, S Wan, JE Vance, DE Vance and RL Jacobs. The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochimica et Biophysica Acta - Biomembranes 2017; 1859(9), 1558-1572.
A Javdani-Mallak and I Salahshoori. Environmental pollutants and exosomes: A new paradigm in environmental health and disease. Science of the Total Environment 2024; 925, 171774.
K Weigand, G Peschel, J Grimm, M Höring, S Krautbauer, G Liebisch, M Müller and C Buechler. Serum phosphatidylcholine species 32:0 as a biomarker for liver cirrhosis pre- and post-hepatitis C virus clearance. International Journal of Molecular Sciences 2024; 25(15), 8161.
R Muraki, Y Morita, S Ida, R Kitajima, S Furuhashi, M Takeda, H Kikuchi, Y Hiramatsu, Y Takanashi, Y Hamaya, K Sugimoto, J Ito, K Kawata, H Kawasaki, T Sato, T Kahyo, M Setou and H Takeuchi. Phosphatidylcholine in bile-derived small extracellular vesicles as a novel biomarker of cholangiocarcinoma. Cancer Medicine 2023; 12(12), 13007-13018.
K Muta, K Saito, Y Kemmochi, T Masuyama, A Kobayashi, Y Saito and S Sugai. Phosphatidylcholine (18:0/20:4), a potential biomarker to predict ethionamide-induced hepatic steatosis in rats. Journal of Applied Toxicology 2022; 42(9), 1533-1547.
SH Yeon, G Yang, HE Lee and JY Lee. Oxidized phosphatidylcholine induces the activation of NLRP3 inflammasome in macrophages. Journal of Leukocyte Biology 2017; 101(1), 205-215.
E Vianello, F Ambrogi, M Kalousová, J Badalyan, E Dozio, L Tacchini, G Schmitz, T Zima, GJ Tsongalis and MM Corsi-Romanelli. Circulating perturbation of phosphatidylcholine (PC) and phosphatidylethanolamine (PE) is associated to cardiac remodeling and NLRP3 inflammasome in cardiovascular patients with insulin resistance risk. Experimental and Molecular Pathology 2024; 137, 104895.
R Corrêa, LFF Silva, DJS Ribeiro, RDN Almeida, IDO Santos, LH Corrêa, LPD Sant’Ana, LS Assunção, PT Bozza and KG Magalhães. Lysophosphatidylcholine induces NLRP3 inflammasome-mediated foam cell formation and pyroptosis in human monocytes and endothelial cells. Frontiers in Immunology 2020; 10, 2927.
L Zhang, R Lukowski, F Gaertner, M Lorenz, KR Legate, K Domes and S Offermanns. Thrombocytosis as a response to high interleukin-6 levels in cGMP-dependent protein kinase I mutant mice. Arteriosclerosis, Thrombosis, and Vascular Biology 2013; 33(8), 1820-1828.
A Kaser, G Brandacher, W Steurer, S Kaser, FA Offner, H Zoller, I Theurl, W Widder, C Molnar, O Ludwiczek, MB Atkins, JW Mier and H Tilg. Interleukin-6 stimulates thrombopoiesis through thrombopoietin: Role in inflammatory thrombocytosis. Blood 2001; 98(9), 2720-2725.
T Ganz. Anaemia of inflammation. New England Journal of Medicine 2019; 381(12), 1149-1157.
L Lanser, D Fuchs, K Kurz and G Weiss. Physiology and inflammation-driven pathophysiology of iron homeostasis - Mechanistic insights into anaemia of inflammation and its treatment. Nutrients 2021; 13(11), 3732.
COS Sorzano, D Tabas-Madrid, F Núñez, C Fernández-Criado and A Naranjo. Sample size for pilot studies and precision-driven experiments. arXiv 2017. https://doi.org/10.48550/arXiv.1707.00222
AK Pham, CW Wu, X Qiu, J Xu, S Smiley-Jewell, D Uyeminami, P Upadhyay, D Zhao and KE Pinkerton. Differential lung inflammation and injury with tobacco smoke exposure in Wistar Kyoto and spontaneously hypertensive rats. Inhalation Toxicology 2020; 32(8), 328-341.
F Vivarelli, C Morosini, L Rullo, LM Losapio, A Lacorte, S Sangiorgi, S Ghini, I Fagiolino, P Franchi, M Lucarini, S Candeletti, D Canistro, P Romualdi and M Paolini. Effects of unburned tobacco smoke on inflammatory and oxidative mediators in the rat prefrontal cortex. Frontiers in Pharmacology 2024; 15, 1328917.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






