Development and Validation of A C57BL/6 Mouse Model of Androgenetic Alopecia Through Testosterone Propionate Induction
DOI:
https://doi.org/10.48048/tis.2026.12711Keywords:
Androgenetic alopecia, Testosterone propionate, C57BL/6 mice, Dermal thickness, Hair follicle density, Wnt/β-catenin signaling, Animal modelAbstract
Androgenetic alopecia (AGA) is the most prevalent form of progressive hair loss, primarily influenced by genetic and androgenic factors, particularly dihydrotestosterone. Existing therapeutic options show limited efficacy, emphasizing the need for reliable and representative animal models. This study established a testosterone propionate (TP)-induced C57BL/6 mouse model of AGA and evaluated the associated clinical, histopathological, and molecular alterations within the Wnt/β-catenin pathway. Twenty-one male C57BL/6 mice were randomized into seven groups (n = 3 per group): One control and six groups receiving subcutaneous testosterone propionate at 0.5, 1.0, or 1.5 mg/day for 7 or 14 days consecutive days. Hair regrowth, dermal thickness, follicle density, and β-catenin expression were analyzed using standardized morphometric and immunohistochemical analyses. Data with normal distribution were assessed using one-way ANOVA, while non-parametric data were evaluated with the Kruskal-Wallis test, followed by LSD or Dunn’s post hoc analysis. Statistical significance was set at p < 0.05. Low-dose, short-term treatment (0.5 mg/day for 7 days) produced only mild dermal thinning and partial follicular reduction, whereas higher doses and prolonged exposure induced progressive inhibition of hair regrowth. The regimen of 1.5 mg/day for 14 days resulted in complete growth arrest, pronounced dermal thinning, follicular depletion, and marked β-catenin suppression (p < 0.001, 0.028, and 0.017, respectively). Post hoc analysis confirmed significant reductions in dermal thickness (groups receiving 1.0 - 1.5 mg/day for 7 - 14 days), follicle density (1.5 mg/day for 7 days and 1.0 - 1.5 mg/day for 14 days), and β-catenin expression <1.5 mg/day for 7 and 14 days>. Despite the small sample size, this study demonstrates that subcutaneous TP administration at 1.5 mg/day for 14 days successfully induces androgenetic alopecia in C57BL/6. This model provides a robust, reproducible, and physiologically relevant platform for mechanistic exploration and preclinical testing of therapeutic agents tergeting Wnt/β-catenin signaling.
HIGHLIGHTS
- A reproducible testosterone propionate–induced androgenetic alopecia model was established in C57BL/6 mice.
- High-dose and prolonged androgen exposure caused dose- and duration-dependent hair growth inhibition.
- Dermal thinning, follicular miniaturization, and β-catenin suppression mirrored human AGA pathology.
- This model provides a robust platform for mechanistic studies and preclinical therapeutic evaluation.
GRAPHICAL ABSTRACT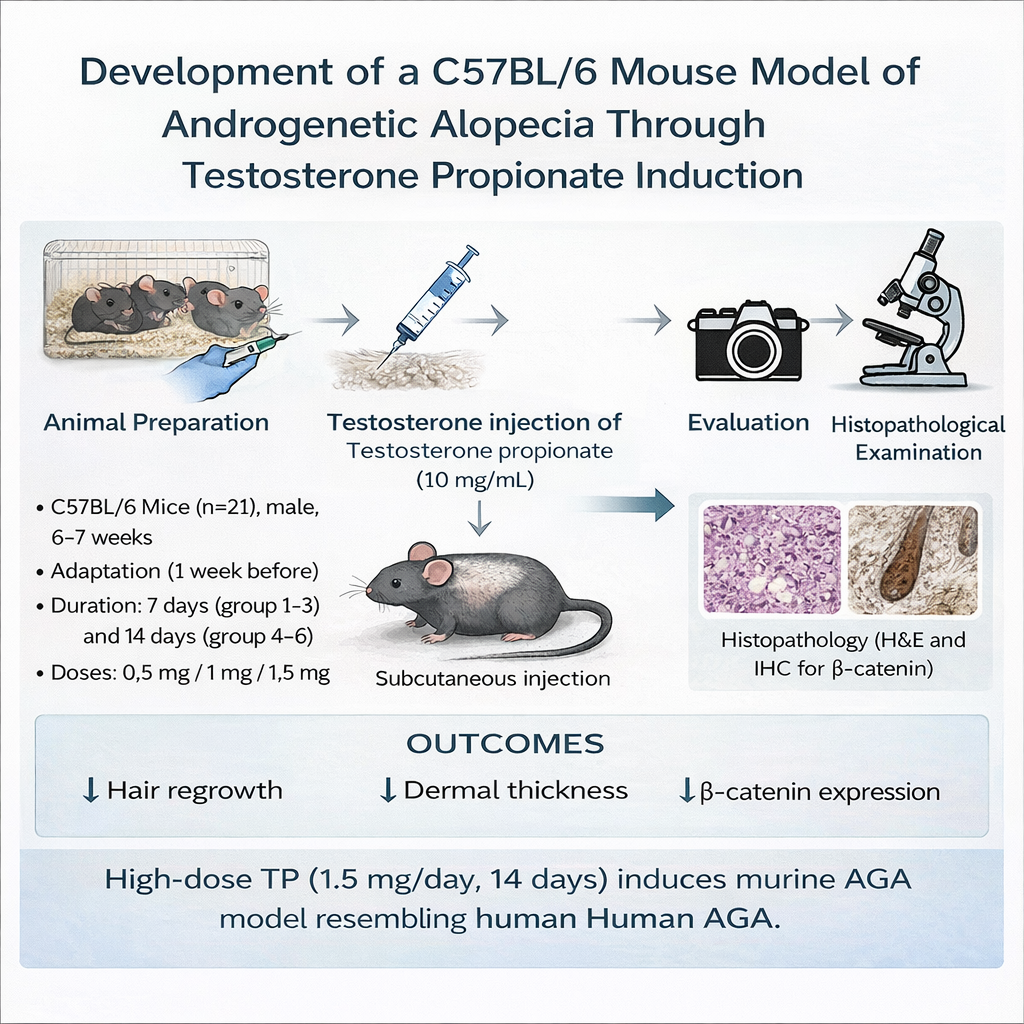
Downloads
References
OAA Ahmed and WY Rizg. Finasteride nano-transferosomal gel formula for management of androgenetic alopecia: Ex vivo investigational approach. Drug Design, Development and Therapy 2018; 12, 2259-2265.
EL Aukerman and M Jafferany. The psychological consequences of androgenetic alopecia: A systematic review. Journal of Cosmetic Dermatology 2023; 22(1), 89-95.
S Ntshingila, NP Khumalo, M Engel and AT Arowolo. An appraisal of laboratory models of androgenetic alopecia: A systematic review. Skin Health and Disease 2021; 1(2), 15.
N Choi, S Shin, SU Song and JH Sung. Minoxidil promotes hair growth through stimulation of growth factor release from adipose-derived stem cells. International Journal of Molecular Sciences 2018; 19(3), 691.
AJ Smith, RE Clutton, E Lilley, KEA Hansen and T Brattelid. PREPARE: Guidelines for planning animal research and testing. Laboratory Animals 2018; 52(2), 135-141.
HR Kim, JU Park, SH Lee, JY Park, W Lee, KM Choi, SY Kim and MH Park. Hair growth effect and the mechanisms of Rosa rugosa extract in DHT-induced alopecia mice model. International Journal of Molecular Sciences 2024; 25(21), 11362.
D Bellani, R Patil, A Prabhughate, R Shahare, M Gold, R Kapoor and D Shome. Pathophysiological mechanisms of hair follicle regeneration and potential therapeutic strategies. Stem Cell Research & Therapy 2025; 16, 302.
CC Akoh and K Sukhdeo. Finasteride counseling for male androgenetic alopecia should reflect updated findings on prostate cancer risk. Skin Appendage Disorders 2020; 6, 130-131.
Y Li, T Dong, S Wan, R Xiong, S Jin, Y Dai and C Guan. Application of multi-omics techniques to androgenetic alopecia: Current status and perspectives. Computational and Structural Biotechnology Journal 2024; 23, 2623-2636.
ND Sert, V Hurst, A Ahluwalia, S Alam, MT Avey, M Baker, WJ Browne, A Clark, IC Cuthill, U Dirnagl, M Emerson, P Garner, ST Holgate, DW Howells, NA Karp, SE Lazic, K Lidster, CJ MacCallum, M Macleod, EJ Pearl, ..., H Würbel. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biology 2020; 18(7), 3000410.
MFW Festing and DG Altman. Guidelines for the design and statistical analysis of experiments using laboratory animals. ILAR Journal 2002; 43(4), 244-258.
GJ Leirós, AI Attorresi and ME Balañá. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. British Journal of Dermatology 2012; 166(5), 1035-1042.
D Fu, J Huang, K Li, Y Chen, Y He, Y Sun, Y Guo, L Du, Q Qu, Y Miao and Z Hu. Dihydrotestosterone-induced hair regrowth inhibition by activating androgen receptor in C57BL/6 mice simulates androgenetic alopecia. Biomedicine & Pharmacotherapy 2021; 137, 111247.
X Yan, M Cheng, X Shen, S Zhao, F Tang, J Tang, C Ao, L Xue, S Zhu, F Wang and WQ Yang. Development and validation of androgen-induced hair loss and anagen induction mouse models for pharmacological evaluation of anti-androgenic agents. Journal of Pharmacology and Pharmacological Sciences 2024; 6, 192.
A Kusumawardani, N Mulianto, A Nanareswari, P Monica and T Oktaviani. Testosterone-induced androgenetic alopecia mice model: A preliminary study. International Journal of Applied Pharmaceutics 2025; 17(2), 78-82.
F Kaliyadan, A Nambiar and S Vijayaraghavan. Androgenetic alopecia: An update. Indian Journal of Dermatology, Venereology and Leprology 2013; 79(5), 613-625.
CH Ho, T Sood and PM Zito. Androgenetic alopecia. StatPearls Publishing, American, 2025.
GL Hong, HJ Lee, YJ Kim, KH Kim and JY Jung. Stauntonia hexaphylla extract ameliorates androgenic alopecia by inhibiting androgen signaling in testosterone-induced alopecia mice. Iranian Journal of Pharmaceutical Research 2023; 21(1), 133333.
KB Chung, YI Lee, YJ Kim, HA Do, J Suk, I Jung, DY Kim and JH Lee. Quantitative analysis of hair luster in a novel ultraviolet-irradiated mouse model. International Journal of Molecular Sciences 2024; 25(3), 1885.
SH Park, E Lee, D Kim, JW Yoon, SM Jeon, YH Song, J Bang and CH Jung. 2′-Fucosyllactose alleviates hair loss in testosterone-induced androgenic alopecia mice. Journal of Food Biochemistry 2025; 2025, 5625208.
MJ Woo, HY Kang, SJ Paik, HJ Choi, S Uddin, S Lee, SY Kim, S Choi and SK Jung. The in vivo and in vitro effects of Terminalia bellirica (Gaertn.) Roxb. fruit extract on testosterone-induced hair loss. Journal of Microbiology and Biotechnology 2023; 33(11), 1467-1474.
A Mehta, M Motavaf, D Raza, AJ McLure, KD Osei-Opare, LA Bordone and AA Gru. Revolutionary approaches to hair regrowth: Follicle neogenesis, Wnt/β-catenin signaling, and emerging therapies. Cells 2025; 14, 779.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






