Antioxidant, Cytotoxic Activities and Metabolite Profile of Flavonoids from Erythrina crista-galli L. Twigs by Ultrasonic Assisted Extraction Method
DOI:
https://doi.org/10.48048/tis.2025.11209Keywords:
Flavonoids; Erythrina crista-galli, Ultrasound-assisted extraction, Antioxidant, Cytotoxic, Metabolite profilingAbstract
Erythrina crista-galli L., a member of the Erythrina genus, is recognized for its high flavonoid content and promising pharmacological potential. This study investigated the impact of different ultrasound-assisted extraction (UAE) condition on the total flavonoid content (TFC) of E. crista-galli twig extract, and evaluated its antioxidant, cytotoxic, and metabolite profiles. Antioxidant activity was determined using the DPPH free radical scavenging assay, while cytotoxicity was assessed against MCF-7, HeLa, and A549 cancer cell lines using the MTT assay. Extraction using UAE with ethanol as a solvent at 40° for 35 min and a mass-to-solvent ratio of 1:25 yielded the highest TFC at 37.69 mg QE/g extract. The extract demonstrated strong antioxidant activity with an IC50 of 54.80 ± 0.26 μg/mL, and moderate cytotoxicity against MCF-7 breast cancer cells (IC50 = 194.6 μg/mL), but showed weak cytotoxicity against HeLa (IC50 = 235.0 μg/mL) and A549 cells (IC50 = 410.3 μg/mL). Comprehensive metabolite profiling using LC-MS/MS identified 34 flavonoid compounds in both positive and negative ion modes, representing the first comprehensive metabolite profile of E. crista-galli twigs. These findings highlight the potential of E. crista-galli as a valuable natural source of bioactive flavonoids for further therapeutic development, including in vivo testing and molecular target identification to validate the efficacy and mechanism of action of these compounds.
HIGHLIGHTS
- This study evaluated the effect of different ultrasound assisted extraction (UAE) condition on total flavonoids content of crista-galli twig extract
- The crista-galli twig extract exhibited strong antioxidant activity against DPPH free radicals with IC50 of 54.80 ± 0.26 μg/mL
- The crista-galli twig extract exhibited moderate cytotoxic activity against MCF-7 breast cancer cell line with IC50 of 194.6 μg/mL, while weak activity against HeLa cervical cancer cell line and A-549 lung cancer cell line with IC50 of 235.0 μg/mL and 410.3 μg/mL respectively.
- To the best of our knowledge, this is the 1st study to report a comprehensive metabolite profiling of flavonoids from Erythrina crista-galli twigs using LC-MS/MS, identifying 34 compounds across both positive and negative ion modes.
- These findings indicate the potential of crista-galli as a natural source of bioactive flavonoid compounds for further therapeutic development.
GRAPHICAL ABSTRACT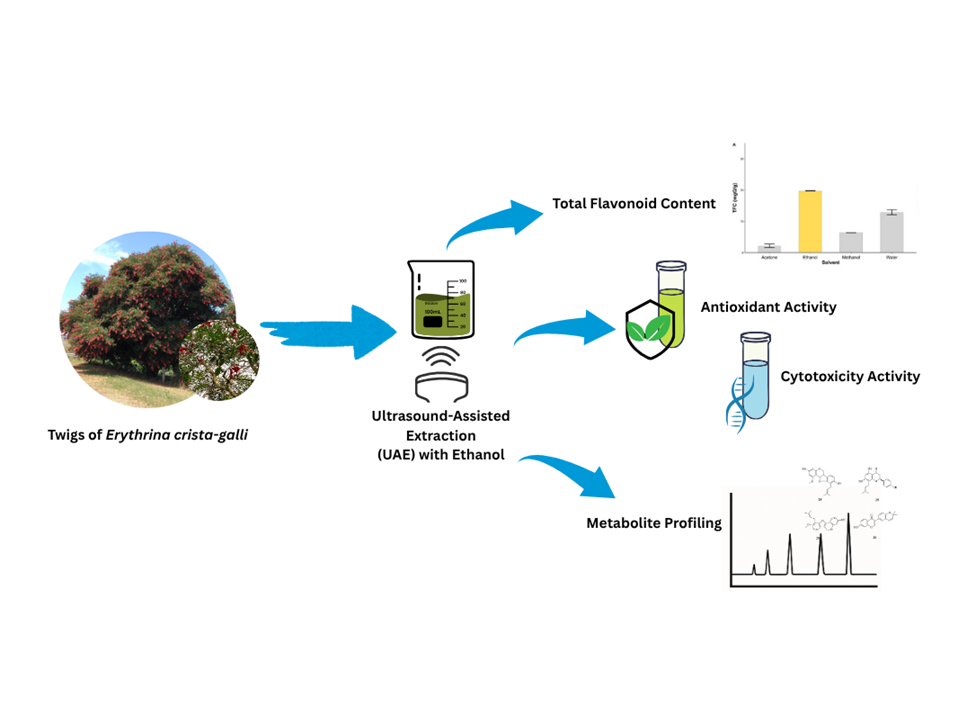
Downloads
References
SL Badshah, S Faisal, A Muhammad, BG Poulson, AH Emwas and M Jaremko. Antiviral activities of flavonoids. Biomedicine & Pharmacotherapy 2021; 140(6), 111596.
S Chen, X Wang, Y Cheng, H Gao and X Chen. A review of classification, biosynthesis, biological activities and potential applications of flavonoids. Molecules 2023; 28(13), 4982.
K Jomova, SY Alomar, R Valko, J Liska, E Nepovimova, K Kuca and M Valko. Flavonoids and their role in oxidative stress, inflammation, and human diseases. Chemico-Biological Interactions 2025; 413, 111489.
V Pereira, O Figueira and PC Castilho. Flavonoids as insecticides in crop protection-a review of current research and future prospects. Plants 2024; 13(6), 776.
P Chand, H Kumar, R Jain, A Jain and V Jain. Flavonoids as omnipotent candidates for cancer management. South African Journal of Botany 2023; 158, 334-346.
A Ullah, S Munir, SL Badshah, N Khan, L Ghani, BG Poulson, AH Emwas and M Jaremko. Important flavonoids and their role as a therapeutic agent. Molecules 2020; 25(22), 5243.
T Herlina, AWR Akili, V Nishinarizki, A Hardianto and JB Latip. Review on antibacterial flavonoids from genus Erythrina: structure-activity relationship and mode of action. Heliyon 2025; 11(1), e41395.
AWR Akili, A Hardianto, J Latip, M Ismiyati and T Herlina. Characterization of botanical parts of Erythrina crista-galli using pyrolysis-gas chromatography/mass spectrometry and multivariate analysis. Indonesian Journal of Chemistry 2023; 23(4), 899-912.
NT Son and AI Elshamy. Flavonoids and other non-alkaloidal constituents of genus Erythrina: phytochemical review. Combinatorial Chemistry & High Throughput Screening 2021; 24(1), 20-58.
CH Liao, HX Fu, KH Tu, JH Zheng, CH Shih and YS Lin. Impacts of extraction solvents and processing techniques on phytochemical composition and antioxidant property of mulberry (Morus spp.) leaves. Cogent Food & Agriculture 2025; 11(1), 2451731.
K Kumar, S Srivastav and VS Sharanagat. Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: A review. Ultrasonics Sonochemistry 2021; 70, 105325.
L Shen, S Pang, M Zhong, Y Sun, A Qayum, Y Liu, A Rashid, B Xu, Q Liang, H Ma and X Ren. A comprehensive review of ultrasonic assisted extraction (UAE) for bioactive components: Principles, advantages, equipment, and combined technologies. Ultrasonics Sonochemistry 2023; 101, 106646.
C Liu, F Xu, Q Zhang, N Xu, J Zhang, Y Shi and K Qin. Ultrasound-assisted enzyme extraction of total flavonoids from lotus leaf (Nelumbo nucifera Gaertn.) and its antioxidant activity. LWT 2025; 215(7), 117224.
YY Ling, PS Fun, A Yeop, MM Yusoff and J Gimbun. Assessment of maceration, ultrasonic and microwave assisted extraction for total phenolic content, total flavonoid content and kaempferol yield from Cassia alata via microstructures analysis. Materials Today: Proceedings 2019; 19(4), 1273-1279.
AR Ahmad and A Malik. Antioxidant activity of Passiflora edulis (passion fruit) seed extracts obtained from maceration and ultrasonic assisted extraction method. Fitofarmaka: Jurnal Ilmiah Farmasi 2023; 13(1), 77-81.
J Zhishen, T Mengcheng and W Jianming. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chemistry 1999; 64(4), 555-559.
DAI Pramitha, T Herlina, IP Maksum, A Hardianto, AWR Akili and J Latip. Metabolite profile and antioxidant activities of Trikatu, black pepper, Javanese long pepper, and red ginger essential oils. Phytomedicine Plus 2025; 5(1), 100702.
N Lall, CJH Smith, MND Canha, CB Oosthuizen and D Berrington. Viability reagent, PrestoBlue, in comparison with other available reagents, utilized in cytotoxicity and antimicrobial assays. International Journal of Microbiology 2013; 2013(9), 420601.
T Mosmann. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Journal of Immunological Methods 1983; 65(1-2), 55-63.
OA Olalere, NH Abdurahmanm RBM Yunus, OR Alara and MM Ahmad. Mineral element determination and phenolic compounds profiling of oleoresin extract using an accurate mass LC-MS-QTOF and ICP-MS. Journal of King Saud University-Science 2018; 31(4), 859-863.
AW Bouhalla, D Benabdelmoumene, S Dahmouni, Z Bengharbi, K Hellal, WSM Qadi, E A-Olayan, A Moreno, A Bekada, N Buzgaia, H Aziz and A Mediani. Comparative LC-MS-based metabolite profiling, antioxidant, and antibacterial properties of Bunium bulbocastanum tubers from two regions in Algeria. Scientific Reports 2024; 14(1), 21719.
ZL Sheng, PF Wan, CL Dong and YH Li. Optimization of total flavonoids content extracted from Flos Populi using response surface methodology. Industrial Crops and Products 2013; 43(1), 778-786.
CC Chang, MH Yang, HM Wen and JC Chern. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. Journal of Food and Drug Analysis 2002; 10(3), 178-182.
A Wojdyło, J Oszmiański and R Czemerys. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chemistry 2007; 105(3), 940-949.
M Shahriar, NZ Khair, Z Sheikh, SF Chowdhury, M Kamruzzaman, MSI Bakhtiar, SJ Chisty, SN Narjish, R Akhter and N Akter. Phytochemical analysis, cytotoxic and in vitro antioxidant activity of Erythrina variegate bark. European Journal of Medicinal Plants 2016; 11(3), 1-5.
I Fidrianny, Annisa and K Ruslan. Antioxidant activities of Arabica green coffee from three regions using ABTS and DPPH assays. Asian Journal of Pharmaceutical and Clinical Research 2016; 9(2), 189-193.
V Deviani, A Hardianto, K Farabi and T Herlina. Flavonones from Erythrina crista-galli twigs and their antioxidant properties determined through in silico and in vitro studies. Molecules 2022; 27(18), 6018.
F Chemat, MA Vian and G Cravotto. Green extraction of natural products: concept and principles. International Journal of Molecular Sciences 2012; 13(7), 8615-8627.
N Chandimali, SG Bak, EH Park, HJ Lim, YS Won, EK Kim, SI Park and SJ Lee. Free radicals and their impact on health and antioxidant defenses: A review. Cell Death Discovery 2025; 11(1), 19.
A Torgovnick and B Schumacher. DNA repair mechanisms in cancer development and therapy. Front Genet 2015; 6, 157.
M Rafi, AH Karomah, DA Septaningsih, Trivadila, M Rahminiwati, SP Putri and D Iswantini. LC-MS/MS based metabolite profiling and lipase enzyme inhibitory activity of Kaempferia angustifolia Rosc. With different extracting solvents. Arabian Journal of Chemistry 2022; 15(11), 104232.
AJ Pérez, EM Hassan, L Pecio, EA Omer, M Kucinska, M Murias and A Srochmal. Triterpenoid saponins and C-glycosyl flavones from stem bark of Erythrina abyssinica Lam and their cytotoxic effects. Phytochemistry Letters 2015; 13, 59-67.
S El-Masry, HM Hammoda, MM Radwan, SA Ross and HH Zaatout. C-flavonoidal glycosides from Erythrina caffra flowers. Natural Product Sciences 2010; 16(4), 217-222.
DRD Oliveira, CR Zamberlam, RB Gaiardo, GM Rêgo, JM Cerutti, AJ Cavalheiro and SM Cerutti. Flavones from Erythrina falcata are modulators of fear memory. BMC Complementary and Alternative Medicine 2014; 14(1), 288.
NS Ashmawy, ML Ashour, M Wink, M El-Shazly, FR Chang, N Swilam, AB Abdel-Naim and N Ayoub. Polyphenols from Erythrina crista-galli: Structures, molecular docking and phytoestrogenic activity. Molecules 2016; 21(6), 726.
KG Bedane, S Kusari, IB Masesane, M Spiteller and RRT Majinda. Flavanones of Erythrina livingstoniana with antioxidant properties. Fitoterapia 2016; 108, 48-54.
W Watjen, AK Suckow-Schnitker, R Rohrig, A Kulawik, J Addae-Kyereme, CW Wright and CM Passreiter. Prenylated flavonoid derivates from the bark of Erythrina Addisoniae. Journal of Natural Products 2008; 71(4), 735-738.
Y Zarev, K Foubert, VLD Almeida, R Anthonissen, E Elgorashi, S Apers, I Ionkova, L Verschaeve and L Pieters. Antigenotoxic prenylated flavonoids from stem bark of Erythrina latissima. Phytochemistry 2017; 141, 140-146.
F Redko, ML Clavin, D Weber, F Ranea, T Anke and V Martino. Antimicrobial isoflavonoids from Erythrina crista-galli infected with Phomopsis sp. Zeitschrift für Naturforschung C 2007; 62(3-4), 164-168.
M Na, J Jang, D Njamen, JT Mbafor, ZT Fomum, BY Kim, WK Oh and JS Ahn. Protein tyrosine phosphatase-1B inhibitory activity of isoprenylated flavonoids isolated from Erythrina mildbraedii. Journal of Natural Products 2006; 69(11), 1572-1576.
L Cui, DT Ndinteh, MK Na, PT Thuong, J Silike-Muruumu, D Njamen, JT Mbafor, ZT Fomum, JS Ahn and WK Oh. Isoprenylated flavonoids from the stem bark of Erythrina abyssinica. Journal of Natural Products 2007; 70(6), 1039-1042.
P Innok, T Rukachaisirikul, S Phongpaichit and A Suksamrarn. Fuscacarpans A-C, new pterocarpans from the stems of Erythrina fusca. Fitoterapia 2010; 81(6), 518-523.
M Sato, H Tanaka, S Fujiwara, M Hirata, R Yamaguchi, H Etoh and C Tokuda. Antibacterial property of isoflavonoids isolated from Erythrina variegata against cariogenic oral bacteria. Phytomedicine 2003; 10(5), 427-433.
ANJ Imanuddin, I Wiani, A Hardianto and T Herlina. Flavonoids from extract butanol of twigs Erythrina crista-galli against the breast cancer cell line within in silico method. Trends in Sciences 2023; 20(7), 5350.
H Tanaka, T Tanaka and H Etoh. Three pterocarpans from Erythrina crista-galli. Phytochemistry 1997; 45(4), 835-838.
S Djiogue, M Halabalaki, X Alexi, D Njamen, ZT Fomum, MN Alexis and AL Skaltsoounis. Isoflavonoids from Erythrina poeppigiana: Evaluation of their binding affinity for the estrogen receptor. Journal of Natural Products 2009; 72(9), 1603-1607.
P Innok, T Rukachaisirikul and A Suksamrarn. Flavonoids and pterocarpans from the bark of Erythrina fusca. Chemical and Pharmaceutical Bulletin 2009; 57(9), 993-996.
T Herlina, AWR Akili, V Nishinarizki, A Hardianto, AP Sulaeman, S Gaffar, E Julaeha, T Mayanti, U Supratman, MA Nafiah and JB Latip. Cytotoxic evaluation, molecular docking, molecular dynamics, and ADMET prediction of isolupalbigenin isolated from Erythrina subumbrans (Hassk). Merr. (Fabaceae) stem bark: Unveiling its anticancer efficacy. Onco Targets and Therapy 2024; 2024(17), 829-840.
KK Atindehou, EF Queiroz, C Terreaux, D Traore and K Hostettmann. Three new prenylated isoflavonoids from the root bark of Erythrina cogelii. Planta Medica 2002; 68(2), 181-182.
H Tanaka, H Etoh, N Watanabe, H Shimizu, M Ahmad and GH Rizwani. Erysubins C-F, four isoflavonoids from Erythrina suberosa var. glabrescences. Phytochemisty 2001; 56(7), 769-773.
H Tanaka, M Hirata, H Etoh, M Sako, M Sato, J Murata, H Murata, D Darnaedi and T Fukai. Six new constituents from the roots of Erythrina variegata. Chemistry & Biodiversity 2004; 1(7), 1101-1108.
PH Nguyen, TNA Nguyen, KW Kang, DT Ndinteh, JT Mbafor, YR Kim and WK Oh. Prenylated pterocarpans as bacterial neuraminidase inhibitors. Bioorganic & Medicinal Chemistry 2010; 18(9), 3335-3344.
AW Andayi, A Yenesew, S Derese, JO Midiwo, PM Gitu, OJI Jondiko, H Akala, P Liyala, J Wangui, NC Waters, M Heydenreich and MG Peter. Antiplasmodial flavonoids from Erythrina sacleuxii. Planta Med 2006; 72(2), 187-189.
T Rukachaisirikul, P Innok, N Aroonrerk, W Boonamnuaylap, S Limrangsun, C Boonyon, U Woonjina and A Suksamrarn. Antibacterial pterocarpans from Erythrina subumbrans. Journal of Ethnopharmacology 2007; 110(1), 171-175.
H Tanaka, M Hirata, H Etoh, H Shimizu, M Sako, J Murata, H Murata, D Darnaedi and T Fukai. Eryvarins F and G, two 3-phenoxychromones from the roots of Erythrina variegate. Phytochemistry 2003; 62(8), 1243-1246.
T Herlina, U Supratman, MS Soedjanaatmadja, A Subarnas, S Sutardjo, NR Abdullah and H Hayashi. Anti-malarial compound from the stem bark of Erythrina variegate. Indonesian Journal of Chemistry 2010; 9(2), 308-311.
M Satyamitra, S Mantena, CKK Nair, S Chandna, BS Dwarakanath and DP Uma. The antioxidant flavonoids, orientin and vicenin enhance repair of radiation-induced damage. Scholarena Journal of Pharmacy and Pharmacology 2014; 1(1), 105.
MS Abdelrahim, AM Abdel-Baky, SAL Bayoumi, SM Mohamed, WM Abdel-Mageed and EY Backheet. Cytotoxic flavone-C-glycosides from the leaves of Dypsis pembana (H.E.Moore) Beentje & J.Dransf., Arecaceae: In vitro and molecular docking studies. BMC Complementary Medicine and Therapies 2023; 23(1), 214.
NI Kashchenko, DN Olennikov and NK Chirikova. Phytohormones and elicitors enhanced the ecdysteroid and glycosylflavone content and antioxidant activity of Silene repens. Applied Sciences 2021; 11(23), 11099.
B Pandey, S Thapa, MS Biradar, B Singh, JB Ghale, P Kharel, PK Jha, RK Yadav, S Dawadi and V Poojashree. LC-MS profiling and cytotoxic activity of Angiopteris helferiana against HepG2 cell line: Molecular insight to investigate anticancer agent. Plos One 2024; 19(12), e0309797.
F Xu, Y Liu, Z Zhang, C Yang and Y Tian. Quansi-MSn identification of flavonone 7-glycoside isomers in Da Chengqi Tang by high performance liquid chromatography-tandem mass spectrometry. Chinese Medicine 2009; 4, 15.
NH Zaidun, ZC Thent and AA Latiff. Combating oxidative stress disorders with citrus flavonoid: Naringenin. Life Sciences 2018; 208(2), 111-122.
R Rashmi, SB Magesh, KM Ramkumar, S Suryanarayananl and MV SubbaRao. Antioxidant potential of naringenin helps to protect liver tissue from Streptozotocin-induced damage. Reports of Biochemistry and Molecular Biology 2018; 7(1), 76-84.
AW Harmon and YM Patel. Naringenin inhibits glucose uptake in MCF-7 breast cancer cells: A mechanism for impaired cellular proliferation. Breast Cancer Research and Treatment 2004; 85(2), 103-110.
I Kruk, HY Aboul-Enein, T Michalska, K Lichszteld and A Kladna. Scavenging of reactive oxygen species by the plant phenols genistein and oleuropein. Luminescence 2005; 20(2), 81-89.
JM Pavese, RL Farmer and RC Bergan. Inhibition of cancer cell invasion and metastasis by genistein. Cancer and Metastasis Reviews 2010; 29(3), 465-482.
W Zhao, Z Shang, Q Li, M Huang, W He, Z Wang and J Zhang. Rapid screening and identification of daidzein metabolites in rats based on UHPLC-LTQ-Orbitrap mass spectrometry coupled with data-mining technologies. Molecules 2018; 23(1), 151.
C Jiang and PK Gates. Systematic characterization of the fragmentation of flavonoids using high-resolution accurate mass electrospray tandem mass spectrometry. Molecules 2024; 29(22), 5246.
MY Sun, Y Ye, L Xiao, K Rahman, W Xia and H Zhang. Daidzein: A review of pharmacological effects. African Journal of Traditional, Complementary and Alternative Medicines 2016; 13(3), 117-132.
KE Heim, AR Tagliaferro and DJ Bobilya. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. The Journal of Nutritional Biochemistry 2002; 13(10), 572-584.
B Botta, A Vitali, P Menendez, D Misiti and GD Monache. Prenylated flavonoids: Pharmacology and biotechnology. Current Medicinal Chemistry 2005; 12(6), 713-739.
CA Williams and RJ Grayer. Antocyanins and other flavonoids. Natural Product Reports 2004; 21(4), 539-573.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






