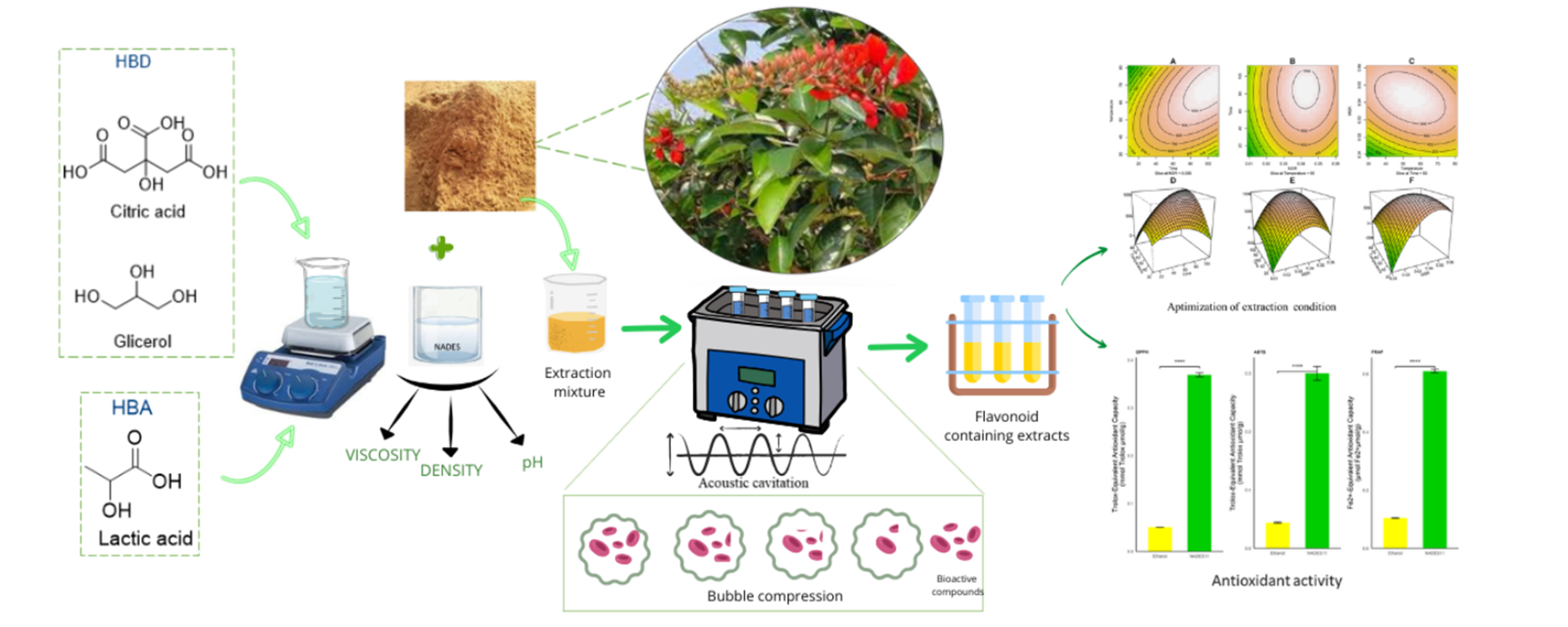
Ultrasound-Assisted Extraction of Flavonoids from Erythrina crista-galli Twigs Using Natural Deep Eutectic Solvents: Process Optimization and Antioxidant Evaluation
DOI:
https://doi.org/10.48048/tis.2025.11099Keywords:
Flavonoids, Erythrina crista-galli, Natural deep eutectic solvent (NADES), Ultrasound-assisted extraction, Response surface methodologyAbstract
Natural Deep Eutectic Solvents (NADES) have emerged as promising, environmentally friendly alternatives to conventional organic solvents for the extraction of bioactive compounds. However, their application to Erythrina crista-galli, a species known for its rich flavonoid content and therapeutic potential, has not yet been systematically investigated. This study addresses this research gap by developing an ultrasound-assisted extraction (UAE) process using NADES, statistically optimized through Central Composite Design (CCD). Among thirteen NADES formulations tested, the NADES11 formulation, composed of lactic acid, citric acid, and glycerol in a molar ratio of 4.5:4.5:1, demonstrated the highest total flavonoid content (TFC) and was selected for further optimization using Response Surface Methodology (RSM). The optimal extraction conditions were established at 97 min, 64 °C, and a solvent-to-material ratio of 1:25. Under these optimized parameters, the antioxidant activities of NADES11 and ethanol extracts were evaluated using DPPH, ABTS, and FRAP assays. The NADES11 extract exhibited significantly higher antioxidant activities, with values of 0.3690 ± 0.0039, 0.2965 ± 0.0118 mmol Trolox/g, and 0.6095 ± 0.0059 mmol Fe²⁺/g, respectively, compared to the ethanol extract, which yielded 0.0532 ± 0.0022, 0.0439 ± 0.0009 mmol Trolox/g, and 0.1043 ± 0.0018 mmol Fe²⁺/g. These results confirm the superior capability of NADES11 not only in efficiently extracting flavonoid compounds but also in preserving their antioxidant properties.
HIGHLIGHTS
- This study explores natural deep eutectic solvents (NADES) as eco-friendly alternatives to conventional solvents for extracting flavonoids from crista-galli twigs.
- Thirteen NADES formulations were tested, with NADES-11 comprising od lactic acid, citric acid, and glycerol with a ratio of 4.5:4.5:1 showing the highest flavonoid yield.
- NADES-based extraction was combined with Ultrasound-Assisted Extraction (UAE) to improve efficiency.
- Extraction parameters were optimized using Response Surface Methodology (RSM) and Central Composite Design (CCD), yielding optimal conditions at 97 min, 64 °C, and a 1:25 solvent-to-material ratio.
- NADES-11 extract exhibited significantly higher antioxidant activity (DPPH, ABTS, and FRAP assays) compared to the ethanol extract.
- This work represents the first systematic and statistically optimized NADES-UAE protocol for flavonoid extraction from crista-galli, highlighting its potential as a sustainable green extraction strategy.
GRAPHICAL ABSTRACT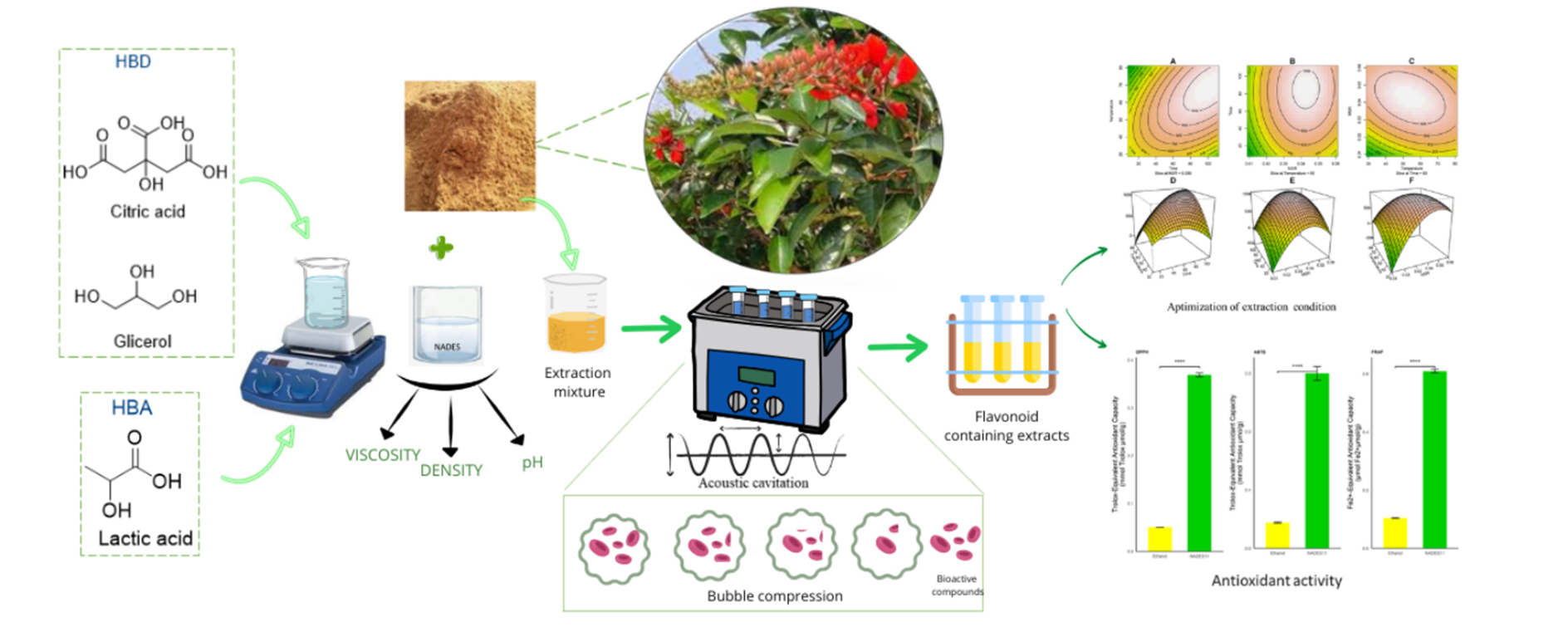
Downloads
References
V Deviani, A Hardianto, K Farabi and T Herlina. Flavanones from Erythrina crista-galli twigs and their antioxidant properties determined through in silico and in vitro studies. Molecules 2022; 27(18), 6018.
ANJ Imanuddin, I Wiani, A Hardianto and T Herlina. Flavonoids from extract butanol of twigs erythrina crista-galli against the breast cancer cell line within in silico method. Trends in Sciences 2023; 20(7), 5350.
NM Fahmy, E Al-Sayed, M El-Shazly and AN Singab. Comprehensive review on flavonoids biological activities of Erythrina plant species. Industrial Crops and Products 2018; 123, 500-538.
NS Ashmawy, ML Ashour, M Wink, M El-Shazly, FR Chang, N Swilam, AB Abdel-Naim and N Ayoub. Polyphenols from Erythrina crista-galli: Structures, molecular docking and phytoestrogenic activity. Molecules 2016; 21(6), 726.
S Liga, C Paul and F Peter. Flavonoids: Overview of biosynthesis, biological activity, and current extraction techniques. Plants 2023; 12(14), 2732.
KM Brodowska. Natural flavonoids: Classification, potential role, and application of flavonoid analogues. European Journal of Biological Research 2017; 7(20), 108-123.
A Hosseinzade, O Sadeghi, AN Biregani, S Soukhtehzari, GS Brandt and A Esmaillzadeh. Immunomodulatory effects of flavonoids: Possible induction of T CD4+ regulatory cells through suppression of mTOR pathway signaling activity. Frontiers in Immunology 2019; 10(51), 00051.
P Pandey, F Khan, HA Qari and M Oves. Rutin (Bioflavonoid) as cell signaling pathway modulator: Prospects in treatment and chemoprevention. Pharmaceuticals (Basel) 2021; 14(11), 1069.
Y Pyo, KH Kwon and YJ Jung. Anticancer potential of flavonoids: Their role in cancer prevention and health benefits. Foods 2024; 13(14), 2253.
P Jauregi, L Esnal-Yeregi and J Labidi. Natural deep eutectic solvents (NADES) for the extraction of bioactives: Emerging opportunities in biorefinery applications. PeerJ Analytical Chemistry 2024; 6, 32.
A Sato and T Nakajima. Pharmacokinetics of organic solvent vapors in relation to their toxicity. Scandinavian Journal of Work, Environment & Health 1987; 13(2), 81-93.
Q Zhao, Y Li, X Chai, L Xu, L Zhang, P Ning, J Huang and S Tian. Interaction of inhalable volatile organic compounds and pulmonary surfactant: Potential hazards of VOCs exposure to lung. Journal of Hazardous Materials 2019; 369, 512-520.
S Mehra, M Singh and P Chadha. View of adverse impact of textile dyes on the aquatic environment as well as on human beings. Toxicology International 2021; 28(2), 165-176.
K Hackl and W Kunz. Some aspects of green solvents. Comptes Rendus Chimie 2018; 21(6), 572-580.
AJ Li, VK Pal and K Kannan. A review of environmental occurrence, toxicity, biotransformation and biomonitoring of volatile organic compounds. Environmental Chemistry and Ecotoxicology 2021; 3, 91-116.
FD Dick. Solvent neurotoxicity. Occupational and Environmental Medicine 2006; 63(3), 221-226.
Y Ma, X He, K Qi, T Wang, Y Qi, L Cui, F Wang and M Song. Effects of environmental contaminants on fertility and reproductive health. Journal of Environmental Sciences 2019; 77, 210-217.
CW Cho, TPT Pham, Y Zhao, S Stolte and YS Yun. Review of the toxic effects of ionic liquids. Science of the Total Environment 2021; 786, 147309.
A Prabhune and R Dey. Green and sustainable solvents of the future: Deep eutectic solvents. Journal of Molecular Liquids 2023; 379, 121676.
A Varfalvyova, A Kalyniukova, I Tomaskova, V Peskova, F Pastierovic, A Jirosova, K Resnerova, D Popelkova and V Andruch. Sugar-based natural deep eutectic solvent ultrasound-assisted extraction for the determination of polyphenolic compounds from various botanical sources. Microchemical Journal 2023; 194, 109249.
Z Xu, X Da, J Qu and S Xiao. Natural deep eutectic solvent-based ultrasound-assisted extraction of flavonoids from fagopyrum tataricum bran. Separations 2024; 11(5), 145.
L Shen, S Pang, M Zhong, Y Sun, A Qayum, Y Liu, A Rashid, B Xu, Q Liang, H Ma and X Ren. A comprehensive review of ultrasonic assisted extraction (UAE) for bioactive components: Principles, advantages, equipment, and combined technologies. Ultrasonics Sonochemistry 2023; 101, 106646.
LM Anaya-Esparza, EF Aurora-Vigo, Z Villagran, E Rodriguez-Lafitte, JM Ruvalcaba-Gomez, MA Solano-Cornejo, VM Zamora-Gasga, E Montalvo-Gonzalez, H Gomez-Rodriguez, CE Aceves-Aldrete and N Gonzalez-Silva. Design of experiments for optimizing ultrasound-assisted extraction of bioactive compounds from plant-based sources. Molecules 2023; 28(23), 7752.
S Kumar, F Abbas, I Ali, MK Gupta, S Kumar, M Garg and D Kumar. Integrated network pharmacology and in-silico approaches to decipher the pharmacological mechanism of Selaginella tamariscina in the treatment of non-small cell lung cancer. Phytomedicine Plus 2023; 3(2), 100419.
CH Tran, MT Nghiem, AMT Dinh, TTN Dang, TTV Do, TN Chu, TH Mai and VM Phan. Optimization conditions of ultrasound-assisted extraction for phenolic compounds and antioxidant activity from Rubus alceifolius poir leaves. International Journal of Food Science 2023; 2023, 7576179.
TP Vo, NTU Nguyen, VH Le, TH Phan, THY Nguyen and DQ Nguyen. Optimizing ultrasonic-assisted and microwave-assisted extraction processes to recover phenolics and flavonoids from passion fruit peels. ACS Omega 2023; 8(37), 33870-33882.
H Koraqi, AY Aydar, W Khalid, S Ercisli, S Rustagi, S Ramniwas and R Pandiselvam. Ultrasound-assisted extraction with natural deep eutectic solvent for phenolic compounds recovery from Rosa damascene Mill.: Experimental design optimization using central composite design. Microchemical Journal 2024; 196, 109585.
NPE Hikmawanti, D Ramadon, I Jantan and A Mun’im. Natural deep eutectic solvents (Nades): Phytochemical extraction performance enhancer for pharmaceutical and nutraceutical product development. Plants 2021; 10(10), 2091.
O Djaoudene, M Bachir-Bey, C Schisano, S Djebari, GC Tenore and A Romano. A sustainable extraction approach of phytochemicals from date (Phoenix dactylifera L.) fruit cultivars using ultrasound-assisted deep eutectic solvent: A comprehensive study on bioactivity and phenolic variability. Antioxidants 2024; 13(2), 181.
Z Wang, Y Tian, M Yang, J Yang, Y Wang, L Tao, J Sheng and C Shi. Extraction of phenolic compounds from Moringa oleifera Lam. leaves with ultrasonic-assisted deep eutectic solvents. Frontiers in Nutrition 2024; 11, 1405128.
K Mulia, F Fauzia and EA Krisanti. Polyalcohols as hydrogen-bonding donors in choline chloride-based deep eutectic solvents for extraction of xanthones from the pericarp of Garcinia mangostana L. Molecules 2019; 24(3), 636.
O Zannou, H Pashazadeh, M Ghellam, AA Redha and I Koca. Enhanced ultrasonically assisted extraction of bitter melon (Momordica charantia) leaf phenolic compounds using choline chloride-acetic acid–based natural deep eutectic solvent: An optimization approach and in vitro digestion. Biomass Conversion and Biorefinery 2024; 14, 11491-11503.
P Diaz, SC Jeong, S Lee, C Khoo and SR Koyyalamudi. Antioxidant and anti-inflammatory activities of selected medicinal plants and fungi containing phenolic and flavonoid compounds. Chinese Medicine 2012; 7(1), 26.
N Phuyal, PK Jha, PP Raturi and S Rajbhandary. Total phenolic, flavonoid contents, and antioxidant activities of fruit, seed, and bark extracts of Zanthoxylum armatum DC. Scientific World Journal 2020; 2020, 8780704.
F Nazir, A Nazir, S Javed and HA Abid. Synthesis and characterization of natural deep eutectic solvents as green extractants for isolation of bioactive flavonoids from Amaranthus viridis. Sustainable Chemistry and Pharmacy 2023; 33, 101058.
A Garcia, MG Alriols, G Spigno and J Labidi. Lignin as natural radical scavenger. Effect of the obtaining and purification processes on the antioxidant behaviour of lignin. Biochemical Engineering Journal 2012; 67, 173-185.
L Yang, D Wang, D Zhou and Y Zhang. Effect of different isolation methods on structure and properties of lignin from valonea of Quercus variabilis. International Journal of Biological Macromolecules 2016; 85, 417-424.
G Grillo, EC Gaudino, R Rosa, C Leonelli, A Timonina, S Grygiškis, S Tabasso and G Cravotto. Green deep eutectic solvents for microwave-assisted biomass delignification and valorisation. Molecules 2021; 26(4), 798.
A NN. A Review on the extraction methods use in medicinal plants, principle, strength and limitation. Medicinal & Aromatic Plants 2015; 4(3), 1000196.
A Khoddami, MA Wilkes and TH Roberts. Techniques for analysis of plant phenolic compounds. Molecules 2013; 18(2), 2328-2375.
XJ Zhang, ZT Liu, XQ Chen, TT Zhang and Y Zhang. Deep eutectic solvent combined with ultrasound technology: A promising integrated extraction strategy for anthocyanins and polyphenols from blueberry pomace. Food Chemistry 2023; 422, 136244.
M Hayyan, A Abo-Hamad, MAH Alsaadi and MA Hashim. Functionalization of graphene using deep eutectic solvents. Nanoscale Research Letters 2015; 10, 324.
W Wang, Y Pan, J Zhao, Y Wang, Q Yao and S Li. Development and optimization of green extraction of polyphenols in Michelia alba using natural deep eutectic solvents (NADES) and evaluation of bioactivity. Sustainable Chemistry and Pharmacy 2024; 37, 101425.
J Rumpf, R Burger and M Schulze. Statistical evaluation of DPPH, ABTS, FRAP, and Folin-Ciocalteu assays to assess the antioxidant capacity of lignins. International Journal of Biological Macromolecules 2023; 233, 123470.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






