The Implementation of Response Surface Methodology in the Optimization of Lipid Nanoparticle Preparation for Vaccine Development
DOI:
https://doi.org/10.48048/tis.2024.7142Keywords:
Aqueous flow rate, Lipid nanoparticles, Particle size, Response surface methodology, N/P ratioAbstract
Lipid nanoparticles (LNPs) are the delivery system behind the success story of mRNA vaccines and the development of DNA and peptide vaccines. LNPs should have particle sizes ranging from 60 to 150 nm for such applications. Achieving such particle size criteria can be challenging in the LNP preparation using a micromixer system with different parameters: i.e., the N/P ratio, aqueous flow rate (AFR) and total flow rate (TFR). Such an issue can be addressed by applying response surface methodology (RSM). Therefore, in this study, we implemented RSM to optimize LNP preparation parameters, including the N/P ratio and AFR. TFR remained constant at 1 mL.min–1 since, from a previous study, it is not a critical parameter for achieving the desired LNP size. Here, we used the pDNA of ACE-2 as a dummy nucleotide. Particle sizes of the resulting LNPs were determined using a dynamic light scattering (DLS) method. Our results indicate that AFR is a significant factor in determining LNP size, whereas the N/P ratio does not have a significant impact on LNP size. We achieved an LNP size of 104.00 nm by applying the N/P ratio of 32.0000 and AFR of 0.9621 mL.min–1. Our results showed that RSM could ease the optimization process in the preparation of LNP, which may support the acceleration of vaccine development.
HIGHLIGHTS
- The significance of lipid nanoparticles (LNPs) as critical delivery systems for mRNA vaccines, DNA vaccines, and peptide vaccines is highlighted
- The study focuses on addressing the challenge of achieving the desired particle size range during LNP preparation
- Response surface methodology (RSM) was employed to optimize the N/P ratio and aqueous flow rate for efficient LNP production
- The study identifies the aqueous flow rate (AFR) as a key factor influencing the size of LNPs
- Overall, the findings of this study contribute to accelerating vaccine development by streamlining the preparation of LNPs
GRAPHICAL ABSTRACT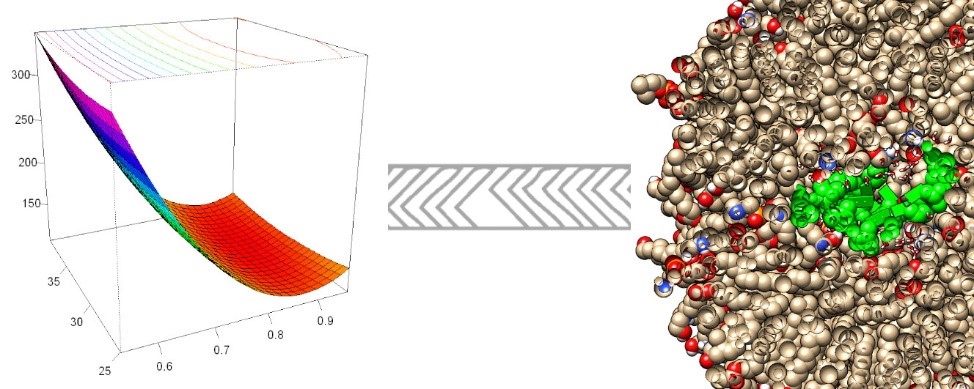
Downloads
References
M Danaei, M Dehghankhold, S Ataei, FH Davarani, R Javanmard, A Dokhani, S Khorasani and MR Mozafari. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 2018; 10, 57.
FP Polack, SJ Thomas, N Kitchin, J Absalon, A Gurtman, S Lockhart, JL Perez, GP Marc, ED Moreira, C Zerbini, R Bailey, KA Swanson, S Roychoudhury, K Koury, P Li, WV Kalina, D Cooper, RWJ Frenck, LL Hammitt, Ö Türeci, H Nell, A Schaefer, S Ünal, DB Tresnan, S Mather, PR Dormitzer, U Şahin, KU Jansen and WC Gruber. Safety and efficacy of the BNT162b2 MRNA Covid-19 vaccine. New Engl. J. Med. 2020; 383, 2603-15.
LR Baden, HME Sahly, B Essink, K Kotloff, S Frey, R Novak, D Diemert, SA Spector, N Rouphael, CB Creech, J McGettigan, S Khetan, N Segall, J Solis, A Brosz, C Fierro, H Schwartz, K Neuzil, L Corey, P Gilbert, …, T Zaks. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. New Engl. J. Med. 2021; 384, 403-16.
Y Fujita and H Taguchi. Nanoparticle-based peptide vaccines. Micro and nanotechnology in vaccine development. Elsevier Inc., Amsterdam, Netherlands, 2017, p. 149-70.
X Han, H Zhang, K Butowska, KL Swingle, MG Alameh, D Weissman and MJ Mitchell. An ionizable lipid toolbox for RNA delivery. Nat. Comm. 2021; 12, 7233.
V Francia, RM Schiffelers, PR Cullis and D Witzigmann. The biomolecular corona of lipid nanoparticles for gene therapy. Bioconjugate Chem. 2020; 31, 2046-59.
MI Henderson, Y Eygeris, A Jozic, M Herrera and G Sahay. Leveraging biological buffers for efficient messenger RNA delivery via lipid nanoparticles. Mol. Pharm. 2022; 19, 4275-85.
S Feng, Z Wang, A Li, X Xie, J Liu, S Li, Y Li, B Wang, L Hu, L Yang and T Guo. Strategies for high-efficiency mutation using the CRISPR/Cas system. Front. Cell Dev. Biol. 2022; 9, 803252.
SA Dilliard and DJ Siegwart. Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs. Nat. Rev. Mater. 2023; 8, 282-300.
M Maeki, Y Fujishima, Y Sato, T Yasui, N Kaji, A Ishida, H Tani, Y Baba, H Harashima and M Tokeshi. Understanding the formation mechanism of lipid nanoparticles in microfluidic devices with chaotic micromixers. PLos One 2017; 12, e0187962.
KJ Hassett, J Higgins, A Woods, B Levy, Y Xia, CJ Hsiao, E Acosta, MJ Moore and LA Brito. Impact of lipid nanoparticle size on MRNA vaccine immunogenicity. J. Contr. Release 2021; 335, 237-46.
A Moreno, J Liu, R Gueret, SE Hadi, L Bergström, A Slabon and MH Sipponen. Unravelling the hydration barrier of lignin oleate nanoparticles for acid‐ and base‐catalyzed functionalization in dispersion state. Angewandte Chemie 2021; 60, 20897-905.
SM Moghimi, AC Hunter and JC Murray. Long-circulating and target-specific nanoparticles: Theory to practice. Pharmacol. Rev. 2001; 53, 283-318.
W Asad, T Kiran, F Saleem, S Siddiqui and SA Rasool. Co-optimization of B. licheniformis 208 biomass and alpha amylase synthesis using response surface methodology. Pakistan J. Bot. 2021; 53, 2287-97.
DC Montgomery. Design and analysis of experiments. 8th eds. John Wiley & Sons, New Jersey, 2013.
J Lawson. Design and analysis of experiments with R. Chapman and Hall/CRC, New York, 2015.
A Permana, HH Purba and S Hasibuan. Design of experiment (DOE) analysis with response surface method (RSM) to optimize the electroplating parameter. ComTech 2021; 12, 99-109.
A Doniavi, A Hosseini and G Ranjbary. Prediction and optimization of mechanical properties of St52 in gas metal arc weld using response surface methodology and ANOVA. Int. J. Eng. 2016; 29, 1307-13.
B Subramaniam, ZH Siddik and NH Nagoor. Optimization of nanostructured lipid carriers: Understanding the types, designs, and parameters in the process of formulations. J. Nanoparticle Res. 2020; 22, 141.
YW Hartati, DR Komala, D Hendrati, S Gaffar, A Hardianto, Y Sofiatin and HH Bahti. An aptasensor using ceria electrodeposited-screen-printed carbon electrode for detection of epithelial sodium channel protein as a hypertension biomarker. Roy. Soc. Open Sci. 2021; 8, 202040.
RH Myers, DC Montgomery and CM Anderson-Cook. Response surface methodology: Process and product optimization using designed experiments. John Wiley & Sons Inc., New Jersey, 2016.
CE Mendoza-Chávez, A Carabin, A Dirany, P Drogui, G Buelna, MM Meza-Montenegro, RG Ulloa-Mercado, LM Díaz-Tenorio, LA Leyva-Soto and P Gortáres-Moroyoqui. Statistical optimization of arsenic removal from synthetic water by electrocoagulation system and its application with real arsenic-polluted groundwater. Environ. Tech. 2020; 42, 3463-74.
C Demirel, A Kabutey, D Herak, A Sedlaček, Č Mizera and O Dajbych. Using box-behnken design coupled with response surface methodology for optimizing rapeseed oil expression parameters under heating and freezing conditions. Processes 2022; 10, 490.
P Tebas, KA Kraynyak, A Patel, JN Maslow, MP Morrow, AJ Sylvester, D Knoblock, E Gillespie, D Amante, T Racine, T McMullan, M Jeong, CC Roberts, YK Park, J Boyer, KE Broderick, GP Kobinger, M Bagarazzi, DB Weiner, NY Sardesai and SM White. Intradermal SynCon® Ebola GP DNA vaccine is temperature stable and safely demonstrates cellular and humoral immunogenicity advantages in healthy volunteers. J. Infect. Dis. 2019; 220, 400-10.
S Stenler, P Blomberg and CIE Smith. Safety and efficacy of DNA vaccines: Plasmids vs. minicircles. Hum. Vaccine. Immunotherapeutics 2014; 10, 1306-8.
C Cayabyab, A Brown, G Tharmarajah and A Thomas. mRNA lipid nanoparticles: Robust low-volume production for screening high-value nanoparticle materials. Precision NanoSystems Inc., British Columbia, Canada, 2019.
CM Bailey-Hytholt, P Ghosh, J Dugas, IE Zarraga and A Bandekar. Formulating and characterizing lipid nanoparticles for gene delivery using a microfluidic mixing platform. J. Visual. Exp. 2021; 168, e62226.
KH Moss, P Popova, SR Hadrup, K Astakhova and M Taskova. Lipid nanoparticles for delivery of therapeutic RNA oligonucleotides. Mol. Pharm. 2019; 16, 2265-77.
F Yanar, A Mosayyebi, C Nastruzzi, D Carugo and X Zhang. Continuous‐flow production of liposomes with a Millireactor under varying fluidic conditions. Pharmaceutics 2020; 12, 1001.
T Terada, JA Kulkarni, A Huynh, S Chen, RVD Meel, YYC Tam and PR Cullis. Characterization of lipid nanoparticles containing ionizable cationic lipids using design-of-experiments approach. Langmuir 2021; 37, 1120-8.
Sigma-Aldrich. 10X Phosphate-Buffered Saline (PBS) for Western blotting, Available at: https:// www.sigmaaldrich.com/ID/en/support/calculators-and-apps/10x-phosphate-buffered-saline, accessed March 2023.
A Behle. Recipe for 50x TAE buffer, Available at: https://www.protocols.io/view/recipe-for-50x-tae-buffer-ewov1d47vr24/v1, accessed March 2023.
AM García-Alegría, I Anduro-Corona, CJ Pérez-Martínez, MAG Corella-Madueño, ML Rascón-Durán and H Astiazaran-Garcia. Quantification of DNA through the nanodrop spectrophotometer: Methodological validation using standard reference material and Sprague Dawley rat and human DNA. Int. J. Anal. Chem. 2020, https://doi:10.1155/2020/8896738.
M Qiu, Z Glass, J Chen, M Haas, X Jin, X Zhao, X Rui, Z Ye, Y Li, F Zhang and Q Xu. Lipid nanoparticle-mediated codelivery of Cas9 MRNA and single-guide RNA achieves liver-specific in vivo genome editing of Angptl3. Proc. Natl. Acad. Sci. Unit. States Am. 2021; 118, e2018362118.
R Core Team. R: A language and environment for statistical computing. R Core Team, Vienna, Austria, 2016.
T Kluyver, B Ragan-Kelley, F Pérez, B Granger, M Bussonnier, J Frederic, K Kelley, J Hamrick, J Grout, S Corlay, P Ivanov, D Avila, S Abdalla and C Willing. Jupyter notebooks - a publishing format for reproducible computational workflows. In: Proceedings of the 20th International Conference on Electronic Publishing, Göttingen, Germany. 2016, p. 87-90
UR Grömping. Package FrF2 for creating and analyzing fractional factorial 2-level designs. J. Stat. Software 2014; 56, 1-56.
R Lenth. V response-surface methods in R, using rsm. J Stat Software 2009; 32, 1-17.
S Bhattacharya. Central composite design for response surface methodology and its application in pharmacy. Response surface methodology in engineering science. IntechOpen, London, 2021.
J Hao, F Wang, X Wang, D Zhang, Y Bi, Y Gao, X Zhao and Q Zhang. Development and optimization of baicalin-loaded solid lipid nanoparticles prepared by coacervation method using central composite design. Eur. J. Pharmaceut. Sci. 2012; 47, 497-505.
P Balagurumoorthy, SJ Adelstein and AI Kassis. Method to eliminate linear DNA from mixture containing nicked circular, supercoiled, and linear plasmid DNA. Anal. Biochem. 2008; 381, 172-4.
JA Glasel. Validity of nucleic acid purities monitored by 260nm/280nm absorbance ratios. Biotechniques 1995; 18, 62-3.
R Hassan, A Husin, S Sulong, S Yusoff, MF Johan, BH Yahaya, C Ang, S Ghazali and SK Cheong. Guidelines for nucleic acid detection and analysis in hematological disorders. Malays. J. Pathol. 2015; 37, 165-73.
Sigma-Aldrich. GenEluteTM plasmid miniprep kit. Sigma-Aldrich, Missouri, 2013.
G Lucena-Aguilar, AM Sánchez-López, C Barberán-Aceituno, JA Carrillo-Ávila, JA López-Guerrero and R Aguilar-Quesada. DNA source selection for downstream applications based on DNA quality indicators analysis. Biopreservation Biobanking 2016; 14, 264-70.
EM Mucker, PP Karmali, J Vega, SA Kwilas, H Wu, M Joselyn, J Ballantyne, D Sampey, R Mukthavaram, E Sullivan, P Chivukula and JW Hooper. Lipid nanoparticle formulation increases efficiency of DNA-vectored vaccines/immunoprophylaxis in animals including transchromosomic bovines. Sci. Rep. 2020; 10, 8764.
M Maugeri, M Nawaz, A Papadimitriou, A Angerfors, A Camponeschi, M Na, M Hölttä, P Skantze, S Johansson, M Sundqvist, J Lindquist, T Kjellman, IL Mårtensson, T Jin, P Sunnerhagen, S Östman, L Lindfors and H Valadi. Linkage between endosomal escape of LNP-mRNA and loading into EVs for transport to other cells. Nat. Comm. 2019; 10, 4333.
C Rae and NJ Thorwirth. Systems and methods for decentralizing commerce and rights management for digital assets using a blockchain rights ledger. Verimatrix Inc, California, 2017.
H Steinle, A Behring, C Schlensak, HP Wendel, M Avci-Adali, SF Ahmed, AA Quadeer, MR McKay, NN Zhang, XF Li, YQ Deng, H Zhao, YJ Huang, G Yang, WJ Huang, P Gao, C Zhou, RR Zhang, Y Guo, SH Sun, …, M Jäättelä. International council for harmonisation (ICH) technical requirements for registration of pharmaceuticals for human use, E6(R2) Good Clinical Practice (GCP) guidance. Viruses 2020; 12, 133-43.
MJ Carrasco, S Alishetty, MG Alameh, H Said, L Wright, M Paige, O Soliman, D Weissman, TE Cleveland, A Grishaev and MD Buchmsnn. Ionization and structural properties of MRNA lipid nanoparticles influence expression in intramuscular and intravascular administration. Comm. Biol. 2021; 4, 956.
X Cheng and RJ Lee. The role of helper lipids in lipid nanoparticles (LNPs) designed for oligonucleotide delivery. Adv. Drug Deliv. Rev. 2016; 99, 129-37.
Y Eygeris, S Patel, A Jozic, G Sahay and G Sahay. Deconvoluting lipid nanoparticle structure for messenger RNA delivery. Nano Lett. 2020; 20, 4543-9.
MY Arteta, T Kjellman, S Bartesaghi, S Wallin, X Wu, AJ Kvist, A Dabkowska, N Székely, A Radulescu, J Bergenholtz and L Lindfors. Successful reprogramming of cellular protein production through MRNA delivered by functionalized lipid nanoparticles. Proc. Natl. Acad. Sci. Unit. States Am. 2018; 115, E3351-E3360.
BL Mui, YK Tam, M Jayaraman, SM Ansell, X Du, YYC Tam, PJ Lin, S Chen, JK Narayanannair, KG Rajeev, M Manoharan, A Akinc, MA Maier, P Cullis, TD Madden and MJ Hope. Influence of polyethylene glycol lipid desorption rates on pharmacokinetics and pharmacodynamics of siRNA lipid nanoparticles. Mol. Ther. Nucleic Acids 2013; 2, e139.
L Xu, X Wang, Y Liu, G Yang, RJ Falconer and CX Zhao. Lipid nanoparticles for drug delivery. Adv. NanoBiomed Res. 2021; 2, 2100109.
AK Blakney, PF McKay, BI Yus, Y Aldon and RJ Shattock. Inside out: Optimization of lipid nanoparticle formulations for exterior complexation and in vivo delivery of saRNA. Gene Ther. 2019; 26, 363-72.
J Wysocki, M Ye, E Rodriguez, FR González-Pacheco, C Barrios, K Evora, M Schuster, H Loibner, KB Brosnihan, CM Ferrario, JM Penninger and D Batlle. Targeting the degradation of angiotensin II with recombinant angiotensin-converting enzyme 2: Prevention of angiotensin II-dependent hypertension. Hypertension 2010; 55, 90-8.
MP Lokugamage, D Vanover, J Beyersdorf, MZC Hatit, L Rotolo, ES Echeverri, HE Peck, H Ni, JK Yoon, YT Kim, PJ Santangelo and JE Dahlman. Optimization of lipid nanoparticles for the delivery of nebulized therapeutic mRNA to the lungs. Nat. Biomed. Eng. 2021; 5, 1059-68.
MJ Anderson and PJ Whitcomb. RSM Simplified: Optimizing processes using response surface methods for design of experiments. 2nd ed. Productivity Press, New York, 2016.
MW Mumtaz, A Adnan, H Mukhtar, U Rashid and M Danish. Biodiesel production through chemical and biochemical transesterification: Trends, technicalities, and future perspectives. Clean energy for sustainable development. Academic Press, London, 2017.
AI Khuri. Response surface methodology and its applications in agricultural and food sciences. Biometrics Biostatistics Int. J. 2017; 5, 155-63.
AM García-Alegría, I Anduro-Corona, CJ Pérez-Martínez, MAG Corella-Madueño, ML Rascón-Durán and H Astiazaran-Garcia. Quantification of DNA through the nanodrop spectrophotometer: Methodological validation using standard reference material and Sprague Dawley rat and human DNA. Int. J. Anal. Chem. 2020; 2020, 8896738.
S Addelman. Statistics for experimenters. Technometrics 1979; 21, 387-8.
FL Vigario, NA Nagy, MH The, R Sparrius, JA Bouwstra, A Kros, W Jiskoot, ECD Jong and B Slütter. The use of a staggered herringbone micromixer for the preparation of rigid liposomal formulations allows efficient encapsulation of antigen and adjuvant. J. Pharmaceut. Sci. 2022; 111, 1050-7.
M Maeki, Y Fujishima, Y Sato, T Yasui, N Kaji, A Ishida, H Tani, Y Baba, H Harashima and M Tokeshi. Understanding the formation mechanism of lipid nanoparticles in microfluidic devices with chaotic micromixers. Plos One 2017; 12, e0187962.
K Kubota, K Onishi, K Sawaki, T Li, K Mitsuoka, T Sato and S Takeoka. Effect of the nanoformulation of siRrNAa-lipid assemblies on their cellular uptake and immune stimulation. Int. J. Nanomedicine 2017; 12, 5121-33.
Y Zhang, H Li, J Sun, J Gao, W Liu, B Li, Y Guo and J Chen. DC-Chol/DOPE cationic liposomes: A comparative study of the influence factors on plasmid pDNA and siRNA gene delivery. Int. J. Pharm. 2010; 390, 198-207.
N Kimura, M Maeki, Y Sato, Y Note, A Ishida, H Tani, H Harashima and M Tokeshi. Development of the ILiNP device: Fine tuning the lipid nanoparticle size within 10 nm for drug delivery. ACS Omega 2018; 3, 5044-51.
NM Belliveau, J Huft, PJ Lin, S Chen, AK Leung, TJ Leaver, AW Wild, JB Lee, RJ Taylor, YK Tam, CL Hansen and PR Cullis. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of siRNA. Mol. Ther. Nucleic Acids 2012; 1, e37.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






