β-Sitosterol from Piper crocatum: A Dual-Action Antifungal and Antibacterial Agent for Oral Infections
DOI:
https://doi.org/10.48048/tis.2025.10420Keywords:
β-sitosterol, Phytosterol, Piper crocatum, Bioactivity-guided, Antimicrobial, β-sitosterol, Phytosterol, Piper crocatum, Bioactivity-guided, Antibacterial, AntifungalAbstract
The high prevalence of antibiotic resistance and oral health problems has sparked research into the development of new antimicrobial medications. The habit of chewing Piper crocatum leaves among Asians has driven this research, leading to the isolation of a bioactive compound. From the methanol extract, the compound -sitosterol, a phytosterol, was isolated for the 1st time from this leaf, this compound has broad medicinal properties, including antifungal and antibacterial effects. The structure of the -sitosterol compound was validated by 1H-NMR, 13C-NMR, IR and MS spectroscopy. Using the broth dilution method, we determined the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of β-sitosterol against the oral pathogens Streptococcus mutans, Streptococcus sanguinis, and Candida albicans. The MIC values were 312.5 ± 0.16 µg/mL for S. mutans, 625 ± 0.11 µg/mL for S. sanguinis, and 625 ± 0.15 µg/mL for C. albicans, respectively. To support these data, we also predicted the potential of the compounds as specific enzyme inhibitors and their absorption, distribution, metabolism, excretion and toxicity (ADMET) properties of the compound and their derivatives by in silico. This study revealed that the derivative β-sitosterol-3-O- -d-glucoside is the most potent as inhibitor of GbpC and SrtC, as antibacterial properties, and is an antifungal agent against Sap5 and CYP51. By preventing the formation of harmful oral bacteria and fungi, the -sitosterol found in P. crocatum leaves and their derivatives can therefore potentially function as an antibacterial agent.The high prevalence of antibiotic resistance and oral health problems has sparked research into the development of new antimicrobial medications. The habit of chewing Piper crocatum leaves among Asians has driven this research, leading to the isolation of a bioactive compound. From the methanol extract, the compound -sitosterol, a phytosterol, was isolated for the 1st time from this leaf, this compound has broad medicinal properties, including antifungal and antibacterial effects. The structure of the -sitosterol compound was validated by 1H-NMR, 13C-NMR, IR and MS spectroscopy. Using the broth dilution method, we determined the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of β-sitosterol against the oral pathogens Streptococcus mutans, Streptococcus sanguinis, and Candida albicans. The MIC values were 312.5 ± 0.16 µg/mL for S. mutans, 625 ± 0.11 µg/mL for S. sanguinis, and 625 ± 0.15 µg/mL for C. albicans, respectively. To support these data, we also predicted the potential of the compounds as specific enzyme inhibitors and their absorption, distribution, metabolism, excretion and toxicity (ADMET) properties of the compound and their derivatives by in silico. This study revealed that the derivative β-sitosterol-3-O- -d-glucoside is the most potent as inhibitor of GbpC and SrtC, as antibacterial properties, and is an antifungal agent against Sap5 and CYP51. By preventing the formation of harmful oral bacteria and fungi, the -sitosterol found in P. crocatum leaves and their derivatives can therefore potentially function as an antibacterial agent.
HIGHLIGHTS
- β-sitosterol, a bioactive phytosterol with therapeutic potential, was successfully isolated from the methanol extract of P. crocatum leaves for the 1st time.
- β-sitosterol has significant activity against Streptococcus mutans, Streptococcus sanguinis, and Candida albicans, with low MIC values indicating strong inhibitory potential.
- Molecular docking revealed that β-sitosterol targets key bacterial and fungal enzymes (GbpC, SrtC, Sap5, and CYP51), supporting its role as a dual-action antimicrobial agent.
- β-Sitosterol-3-O-β-D-glucoside exhibits strong and stable binding to CYP51, as evidenced by consistently low RMSD values, favorable MM/GBSA binding energies, and minimal structural fluctuations observed via molecular dynamics (MD) simulations.
GRAPHICAL ABSTRACT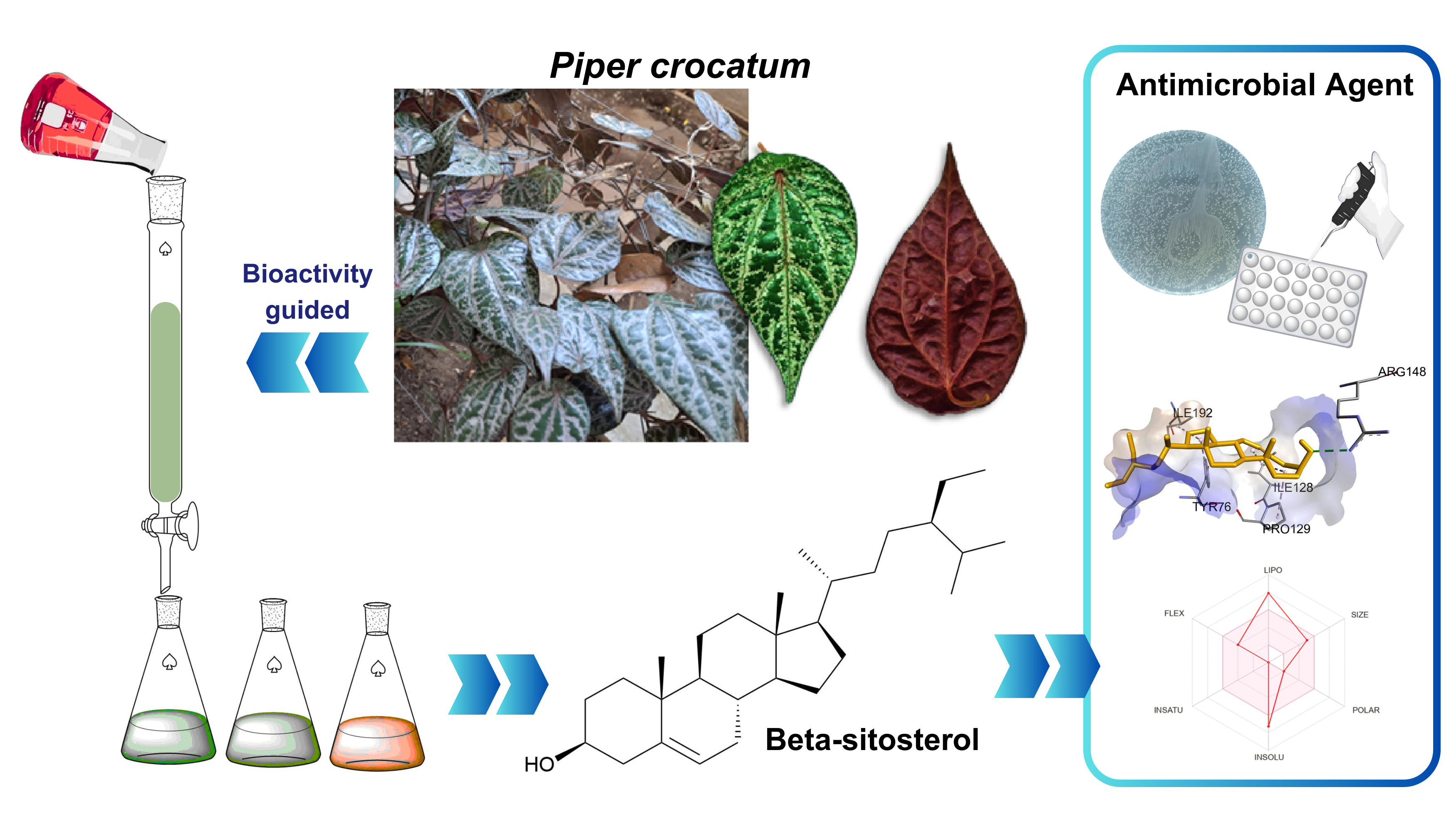
Downloads
References
P Natarajan, S Madanian and S Marshall. Investigating the link between oral health conditions and systemic diseases: A cross-sectional analysis. Scientific Reports 2025; 15, 10476.
B Zhu, LC Macleod, T Kitten and P Xu. Streptococcus sanguinis biofilm formation & interaction with oral pathogens. Future Microbiology 2018; 13(8), 915-932.
SF Kane. The effects of oral health on systemic health. General Dentistry 2017; 65(6), 30-34.
MS Tonetti, P Bottenberg, G Conrads, P Eickholz, P Heasman, MC Huysmans, R López, P Madianos, F Müller, I Needleman, B Nyvad, PM Preshaw, I Pretty, S Renvert, F Schwendicke, L Trombelli, GJVD Putten, J Vanobbergen, N West, A Young and S Paris. Dental caries and periodontal diseases in the ageing population: Call to action to protect and enhance oral health and well‐being as an essential component of healthy ageing. Journal of Clinical Periodontology 2017; 44(S18), S135-S144.
JH Meurman and A Bascones-Martinez. Oral infections and systemic health - More than just links to cardiovascular diseases. Oral Health and Preventive Dentistry 2021; 19, 441-448.
D Shetty, M Dua, K Kumar, R Dhanapal, M Astekar and DC Shetty. Oral hygiene status of individuals with cardiovascular diseases and associated risk factors. Clinics and Practice 2012; 2(4), e86.
WJ Loesche. Role of Streptococcus mutans in human dental decay. Microbiology Reviews 1986; 50(4), 353-380.
C Nabert‐Georgi, AC Rodloff, H Jentsch, DR Reissmann, R Schaumann and CS Stingu. Influence of oral bacteria on adhesion of Streptococcus mutans and Streptococcus sanguinis to dental materials. Clinical and Experimental Dental Research 2018; 4(3), 72-77.
CO Truss. The role of Candida albicans in human illness. Journal of Orthomolecular Psychiatry 1981; 10, 228-238.
M Patel. Oral cavity and Candida albicans: Colonisation to the development of infection. Pathogens 2022; 11(3), 335.
S Patil, RS Rao, B Majumdar and S Anil. Clinical appearance of oral Candida infection and therapeutic strategies. Frontiers in Microbiology 2015; 6, 1391.
W Qiu, Y Zhou, Z Li, T Huang, Y Xiao, L Cheng, X Peng, L Zhang and B Ren. Application of antibiotics/antimicrobial agents on dental caries. BioMed Research International 2020; 2020, 5658212.
R Rautemaa and G Ramage. Oral candidosis-clinical challenges of a biofilm disease. Critical Reviews in Microbiology 2011; 37(4), 328-336.
MC Fisher, A Alastruey-Izquierdo, J Berman, T Bicanic, EM Bignell, P Bowyer, M Bromley, R Brüggemann, G Garber, OA Cornely, SJ Gurr, TS Harrison, E Kuijper, J Rhodes, DC Sheppard, A Warris, PL White, J Xu, B Zwaan and PE Verweij. Tackling the emerging threat of antifungal resistance to human health. Nature Reviews Microbiology 2022; 20, 557-571.
A Arastehfar, T Gabaldón, R Garcia-Rubio, JD Jenks, M Hoenigl, HJF Salzer, M Ilkit, C Lass-Flörl and DS Perlin. Drug-resistant fungi: An emerging challenge threatening our limited antifungal armamentarium. Antibiotics 2020; 9(12), 877.
MC Fisher, NJ Hawkins, D Sanglard and SJ Gurr. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018; 360(6390), 739-742.
A Budimir. Fighting antimicrobial resistance. IAPC Publishing, Zagreb, Croatia, 2018.
NA Church and JL McKillip. Antibiotic resistance crisis: Challenges and imperatives. Biologia 2021; 76, 1535-1550.
AN Olaimat, MA Al‐Holy, HM Shahbaz, AA Al‐Nabulsi, MHA Ghoush, TM Osaili, MM Ayyash and RA Holley. Emergence of antibiotic resistance in Listeria monocytogenes isolated from food products: A comprehensive review. Comprehensive Reviews in Food Science and Food Safety 2018; 17, 1277-1292.
AT Nishimoto, C Sharma and PD Rogers. Molecular and genetic basis of azole antifungal resistance in the opportunistic pathogenic fungus Candida albicans. Journal of Antimicrobial Chemotherapy 2020; 75(2), 257-270.
S Silva, CF Rodrigues, D Araújo, ME Rodrigues and M Henriques. Candida species biofilms’ antifungal resistance. Journal of Fungi 2017; 3(1), 8.
SK Ahmed, S Hussein, K Qurbani, RH Ibrahim, A Fareeq, KA Mahmood and MG Mohamed. Antimicrobial resistance: Impacts, challenges, and future prospects. Journal of Medicine, Surgery, and Public Health 2024; 2, 100081.
Z Khan, N Nath, A Rauf, TB Emran, S Mitra, F Islam, D Chandran, J Barua, MU Khandaker, AM Idris, P Wilairatana and M Thiruvengadam. Multifunctional roles and pharmacological potential of β-sitosterol: Emerging evidence toward clinical applications. Chemico-biological Interactions 2022; 365, 110117.
R Barbieri, E Coppo, A Marchese, M Daglia, E Sobarzo-Sánchez, SF Nabavi and SM Nabavi. Phytochemicals for human disease: An update on plant-derived compounds antibacterial activity. Microbiological Research 2017; 196, 44-68.
D Ghosh, N Datta, D Banerjee, S Adhikary, S Banerjee and A De. Current trends on phytochemicals toward herbal medicine development. In: SC Izah, MC Ogwu and M Akram (Eds.). Herbal medicine phytochemistry: Applications and trends. Springer, Cham, Switzerlands, 2024.
SA Zacchino, E Butassi, E Cordisco and LA Svetaz. Hybrid combinations containing natural products and antimicrobial drugs that interfere with bacterial and fungal biofilms. Phytomedicine 2017; 37, 14-26.
M Matsumoto-Nakano. Role of Streptococcus mutans surface proteins for biofilm formation. Japanese Dental Science Review 2018; 54(1), 22-29.
T Rezaei, B Mehramouz, P Gholizadeh, L Yousefi, K Ganbarov, R Ghotaslou, S Taghizadeh and HS Kafil. Factors associated with Streptococcus mutans pathogenicity in the oral cavity. Biointerface Research in Applied Chemistry 2023; 13(4), 368.
MB Winter, EC Salcedo, MB Lohse, N Hartooni, M Gulati, H Sanchez, J Takagi, B Hube, DR Andes, AD Johnson, CS Craik and CJ Nobile. Global identification of biofilm-specific proteolysis in Candida albicans. mBio 2016; 7(5), e01514-16.
A Nobbs and J Kreth. Genetics of sanguinis - Group streptococci in health and disease. Microbiology Spectrum 2019. https://doi.org/10.1128/microbiolspec.GPP3-0052-2018.
D Kurnia, E Apriyanti, C Soraya and MH Satari. Antibacterial flavonoids against oral bacteria of enterococcus faecalis ATCC 29212 from Sarang Semut (Myrmecodia pendans) and its inhibitor activity against enzyme MurA. Current Drug Discovery Technologies 2019; 16, 290-296.
SA Putri, AAN Shadrina, E Julaeha and D Kurnia. Potential nevadensin from ocimum basilicum as antibacterial agent against Streptococcus mutans: In vitro and in silico studies. Combinatorial Chemistry and High Throughput Screening 2023; 26(9), 1746-1754.
SA Putri, E Julaeha, N Kagawa and D Kurnia. Phenolic compounds from Ocimum basilicum revealed as antibacterial by experimental and computational screening‐based studies against oral infections. Journal of Chemistry 2024; 2024, 3696250.
D Kurnia, GS Hutabarat, D Windaryanti, T Herlina, Y Herdiyati and MH Satari. Potential allylpyrocatechol derivatives as antibacterial agent against oral pathogen of S. sanguinis ATCC 10556 and as inhibitor of MurA enzymes: In vitro and in silico study. Drug Design, Development and Therapy 2020; 14, 2977-2985.
DO Oladejo, GO Oduselu, TM Dokunmu, I Isewon, J Oyelade, E Okafor, EE Iweala and E Adebiyi. In silico structure prediction, molecular docking, and dynamic simulation of Plasmodium falciparum AP2-I transcription factor. Bioinformatics and Biology Insights 2023; 17, 11779322221149616.
S Shaweta, S Akhil and G Utsav. Molecular docking studies on the Anti-fungal activity of Allium sativum (Garlic) against Mucormycosis (black fungus) by BIOVIA discovery studio visualizer 21.1.0.0. Annals of Antivirals and Antiretrovirals 2021; 2021, 28-32.
WP Feinstein and M Brylinski. Calculating an optimal box size for ligand docking and virtual screening against experimental and predicted binding pockets. Journal of Cheminformatics 2015; 7, 18.
E Mateev, I Valkova, B Angelov, M Georgieva and A Zlatkov. Validation through re-docking, cross-docking and ligand enrichment in various well-resoluted MAO-B receptors. International Journal of Pharmaceutical Sciences and Research 2022; 13(8), 1099-1107.
R Amini, S Moradi, R Najafi, M Mazdeh and A Taherkhani. BACE1 inhibition utilizing organic compounds holds promise as a potential treatment for Alzheimer’s and Parkinson’s diseases. Oxidative Medicine and Cellular Longevity 2024; 2024, 6654606.
FX Domínguez-Villa, NA Durán-Iturbide and JG Ávila-Zárraga. Synthesis, molecular docking, and in silico ADME/Tox profiling studies of new 1-aryl-5-(3-azidopropyl)indol-4-ones: Potential inhibitors of SARS CoV-2 main protease. Bioorganic Chemistry 2021; 106, 104497.
PV Zadorozhnii, VV Kiselev and AV Kharchenko. In slico ADME profiling of salubrinal and its analogues. Future Pharmacology 2022; 2(2), 160-197.
U Das, T Chanda, J Kumar and A Peter. Discovery of natural MCL1 inhibitors using pharmacophore modelling, QSAR, docking, ADMET, molecular dynamics, and DFT analysis. Computational Biology and Chemistry 2025; 117, 108427.
L Fu, S Shi, J Yi, N Wang, Y He, Z Wu, J Peng, Y Deng, W Wang, C Wu, A Lyu, X Zeng, W Zhao, T Hou and D Cao. ADMETlab 3.0: An updated comprehensive online ADMET prediction platform enhanced with broader coverage, improved performance, API functionality and decision support. Nucleic Acids Research 2024; 52(W1), W422-W431.
AJ Uttu, MS Sallau, H Ibrahim and ORA Iyun. Isolation, characterization, and docking studies of campesterol and β-sitosterol from Strychnos innocua (Delile) root bark. Journal of Taibah University Medical Sciences 2023; 18(3), 566-578.
L Narayanan, T Maruthavanan, Vaithiyanathan R, G Venkatesan, KE Vivekanandan, G Kaliyannan, PSM Kumar, MSAJ Bosco, G Murugadoss and SR Suseem. Comprehensive investigation of antibacterial, cytotoxic, and antioxidant activities of Ipomoea staphylina flower extract with isolation and characterization of beta-sitosterol. Journal of Molecular Structure 2025; 1336, 142045.
L Cartuche, C Vallejo, E Castillo, N Cumbicus and V Morocho. Chemical profiling of Drimys granadensis (Winteraceae) essential oil, and their antimicrobial, antioxidant, and anticholinesterase properties. Plants 2024; 13(13), 1806.
D Somda, JL Bargul, JM Wesonga and SW Wachira. Green synthesis of Brassica carinata microgreen silver nanoparticles, characterization, safety assessment, and antimicrobial activities. Scientific Reports 2024; 14, 29273.
K Rainey, SM Michalek, ZT Wen and H Wu. Glycosyltransferase-mediated biofilm matrix dynamics and virulence of Streptococcus mutans. Applied and Environmental Microbiology 2019; 85, e02247-18.
AM Martini, BS Moricz, AK Ripperger, PM Tran, ME Sharp, AN Forsythe, K Kulhankova, W Salgado-Pabón and BD Jones. Association of novel Streptococcus sanguinis virulence factors with pathogenesis in a native valve infective endocarditis model. Frontiers in Microbiology 2020; 11, 10.
B Nijampatnam, P Ahirwar, P Pukkanasut, H Womack, L Casals, H Zhang, X Cai, SM Michalek, H Wu and SE Velu. Discovery of potent inhibitors of Streptococcus mutans biofilm with antivirulence activity. ACS Medicinal Chemistry Letters 2021; 12(1), 48-55.
TM Vieira, JG Barco, SLD Souza, ALO Santos, I Daoud, S Rahali, N Amdouni, JK Bastos, CHG Martins, RB Said and AEM Crotti. In vitro and in silico studies of the antimicrobial activity of prenylated phenylpropanoids of green propolis and their derivatives against oral bacteria. Antibiotics 2024; 13(8), 787.
B Udosen, O Soremekun, C Ekenna, OI Omotuyi, T Chikowore, O Nashiru and S Fatumo. In-silico analysis reveals druggable single nucleotide polymorphisms in angiotensin 1 converting enzyme involved in the onset of blood pressure. BMC Research Notes 2021; 14, 457.
SC Lovell, IW Davis, WB Arendall, PIWD Bakker, JM Word, MG Prisant, JS Richardson and DC Richardson. Structure validation by Cα geometry: ϕ, ψ and Cβ deviation. Proteins: Structure, Function, and Bioinformatics 2003; 50(3), 437-450.
GN Ramachandran, C Ramakrishnan and V Sasisekharan. Stereochemistry of polypeptide chain configurations. Journal of Molecular Biology. 1963; 7, 95-99.
A Abdullaev, I Abdullaev, A Bogbekov, U Gayibov, S Omonturdiev, S Gayibova, M Turahodjayev, K Ruziboev and T Aripov. Antioxidant potential of Rhodiola heterodonta extract: Activation of Nrf2 pathway via integrative in vivo and in silico studies. Trends in Sciences 2025; 22(5), 9521.
OJ Juvik, B Holmelid, GW Francis, HL Andersen, APD Oliveira, RDGO Júnior, JGDS Almeida and T Fossen. Non-polar natural products from Bromelia laciniosa, Neoglaziovia variegata and Encholirium spectabile (Bromeliaceae). Molecules 2017; 22, 1478.
NS Njinga, MI Sule, UU Pateh, HS Hassan, ST Abdullahi and RN Ache. Isolation and antimicrobial activity of β-sitosterol-3-O-glucoside from Lannea Kerstingii Engl. & K. Krause (Anacardiacea). Nitte University Journal of Health Sciences 2016; 6(1), 4-8.
T Peshin and HK Kar. Isolation and characterization of β-Sitosterol-3-O-β-D-glucoside from the extract of the flowers of Viola odorata. British Journal of Pharmaceutical Research. 2017; 16(4), 33160.
N Tripathi, S Kumar and R Singh. Isolation of 7-β-Hydroxy-β-Sitosterol from roots of Girardinia heterophylla (Decne). Asian Journal of Research in Chemistry 2013; 6, 1107-1109.
D Labaree, RM Hoyte and RB Hochberg. A direct stereoselective synthesis of 7β-hydroxytestosterone. Steroids 1997; 62(6), 482-486.
P Mohana, A Singh, F Rashid, S Singh, K Kaur, R Rana, PMS Bedi, N Bedi, R Kaur and S Arora. Inhibition of virulence associated traits by β-Sitosterol isolated from Hibiscus rosa-sinensis flowers against Candida albicans: Mechanistic insight and molecular docking studies. Journal of Microbiology 2024; 62(12), 1165-1175.
J Wu, Y Zhou, J Zhang, HX Zhang and R Jia. Molecular dynamics simulation investigation of the binding and interaction of the EphA6-Odin protein complex. The Journal of Physical Chemistry B 2022; 126, 4914-4924.
PG Pappas, CA Kauffman, DR Andes, CJ Clancy, KA Marr, L Ostrosky-Zeichner, AC Reboli, MG Schuster, JA Vazquez, TJ Walsh, TE Zaoutis and JD Sobel. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clinical Infectious Diseases 2016; 62(4), e1-e50.
Y Feng, H Lu, M Whiteway and Y Jiang. Understanding fluconazole tolerance in Candida albicans: Implications for effective treatment of candidiasis and combating invasive fungal infections. Journal of Global Antimicrobial Resistance 2023; 35, 314-321.
T Herlina, V Nishinarizki, AWR Akili, A Hardianto, S Gaffar, M Muchtaridi and J Latip. Exploring Erythrina flavonoids as potential SARS-CoV-2 RdRp inhibitors through virtual screening, in silico ADMET evaluation, and molecular dynamics simulation studies. Scientific Reports 2025; 15, 14259.
JF Fatriansyah, AG Boanerges, SR Kurnianto, AF Pradana, Fadilah and SN Surip. Molecular dynamics simulation of ligands from Anredera cordifolia (Binahong) to the main protease (Mpro) of SARS-CoV-2. Journal of Tropical Medicine 2022; 2022, 1178228.
S Lee, AR Wong, AWH Yang and A Hung. Interaction of compounds derived from the Chinese medicinal formula Huangqi Guizhi Wuwu Tang with stroke-related numbness and weakness targets: An in-silico docking and molecular dynamics study. Computers in Biology and Medicine 2022; 146, 105568.
F Talab, A Alam, Zainab, S Ullah, AA Elhenawy, SAA Shah, M Ali, SA Halim, A Khan, A Latif, A Al-Harrasi and M Ahmad. Novel hydrazone schiff’s base derivatives of polyhydroquinoline: Synthesis, in vitro prolyl oligopeptidase inhibitory activity and their Molecular docking study. Journal of Biomolecular Structure and Dynamics 2024. https://doi.org/10.1080/07391102.2024.2319677.
I Sama-ae, NC Pattaranggoon and A Tedasen. In silico prediction of antifungal compounds from natural sources towards Lanosterol 14-alpha demethylase (CYP51) using molecular docking and molecular dynamic simulation. Journal of Molecular Graphics and Modelling 2023; 121, 108435.
MA Hosen, YE Bakri, HM Rehman, HE Hashem, M Saki and SMA Kawsar. A computational investigation of galactopyranoside esters as antimicrobial agents through antiviral, molecular docking, molecular dynamics, pharmacokinetics, and bioactivity prediction. Journal of Biomolecular Structure and Dynamics 2024; 42(2), 1015-1030.
SQ Pantaleão, PO Fernandes, JE Gonçalves, VG Maltarollo and KM Honorio. Recent advances in the prediction of pharmacokinetics properties in drug design studies: A review. ChemMedChem 2022; 17(1), e202100542.
L Kumari, Y Choudhari, P Patel, GD Gupta, D Singh, JM Rosenholm, KK Bansal and BD Kurmi. Advancement in solubilization approaches: A step towards bioavailability enhancement of poorly soluble drugs. Life 2023; 13, 1099.
DV Bhalani, B Nutan, A Kumar and AKS Chandel. Bioavailability enhancement techniques for poorly aqueous soluble drugs and therapeutics. Biomedicines 2022; 10(9), 2055.
K Arumugam, K Chandran, A Zochedh, S Ansar, AB Sultan, YA Kumar and T Kathiresan. Pharmacoinformatics and quantum chemicals‐based analysis of aromatic molecule decanal as a potent drug against breast cancer. International Journal of Quantum Chemistry 2024; 124(15), e27451.
M Zhao, J Ma, M Li, Y Zhang, B Jiang, X Zhao, C Huai, L Shen, N Zhang, L He and S Qin. Cytochrome P450 enzymes and drug metabolism in humans. International Journal of Molecular Sciences 2021; 22, 12808.
YY Zhang, YF Huang, J Liang and H Zhou. Improved up-and-down procedure for acute toxicity measurement with reliable LD50 verified by typical toxic alkaloids and modified Karber method. BMC Pharmacology and Toxicology 2022; 23(1), 3.
M Stielow, A Witczyńska, N Kubryń, Ł Fijałkowski, J Nowaczyk and A Nowaczyk. The bioavailability of drugs—The current state of knowledge. Molecules 2023; 28(24), 8038.
ASA Wasidi, AS Hassan and AM Naglah. In vitro cytotoxicity and druglikeness of pyrazolines and pyridines bearing benzofuran moiety. Journal of Applied Pharmaceutical Science 2020; 10, 142-148.
MO Aliba, IG Ndukwe and H Ibrahim. Isolation and characterization of Β-sitosterol from methanol extracts of the stem bark of large-leaved rock fig (Ficus abutilifolia Miq.). Journal of Applied Sciences and Environmental Management 2018; 22, 1639.
Erwin, WR Pusparohmana, RD Safitry, E Marliana, Usman and IW Kusuma. Isolation and characterization of stigmasterol and beta-sitosterol from wood bark extract of Baccaurea macrocarpa Miq. Mull. Arg. Rasayan Journal of Chemistry 2020; 13, 2552-2558.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






