Exploring Antibacterial Potential of Anhuienoside E from Nigella sativa Linn: A Promising Candidate Against Dental Caries In Vitro and In Silico Studies
DOI:
https://doi.org/10.48048/tis.2025.10615Keywords:
Nigella sativa L., Dental caries, Antibacterial, Anhuienoside EAbstract
Dental caries is a chronic disease suffered by almost the entire population in the world. The main bacterium that causes dental caries is Streptococcus mutans, which has the enzyme glucosyltransferase as a virulence factor. Other fatogenic bacteria that play a role in exacerbating the biofilm form of dental caries such as Streptococccus sanguinis and Enterococcus faecalis have cell wall defenses catalyzed by the MurA enzyme. Chlorhexidine has been reported as a treatment for dental caries but has developed resistance over time. Nigella sativa L. seeds have been widely recognized to have many health benefits such as antibacterial, antioxidant and antifungal. The caries-causing antibacterial activity of N. sativa seeds has not been widely reported. The aim of this study was to isolate antibacterial compounds from N. sativa against S. mutans, S. sanguinis, E. faecalis, predict the mechanism of compounds and their derivatives by in silico molecular docking and pharmacokinetic analysis by ADMET and drug-likeness methods. Isolation of compounds was carried out by column chromatography with bioassay guidance, inhibitory mechanism and sugar substituent effects were predicted by in silico molecular docking, pharmacokinetics and drug-likeness analysis were predicted by ADMET and Lipinski rules. Anhuienoside E was successfully isolated from N. sativa extract and demonstrated antibacterial activity, which had moderate MIC and MBC values of 625 and 1000 μg/mL against S. mutans, S. sanguinis, E. faecalis. Anhuienoside E had moderate binding affinity value compared to its derivatives with ∆G values of −6.84 and −8.67 Kcal/mol against MurA and Gtf. ADMET pharmacokinetic analysis and drug-likeness evaluation suggest that Anhuienoside E and its derivatives may serve as non-toxic, non-oral drug candidates. In conclusion, Anhuienoside E has potential antibacterial activity to be developed as an alternative to chlorhexidine. The amount and type of sugar substituents greatly affect the antibacterial activity of Anhuienoside E.
HIGHLIGHTS
- Anhuienoside E was successfully isolated from the methanol extract of sativa seeds.
- Anhuienoside E has antibacterial activity against mutans, S. sanguinis, E. faecalis based on MIC and MBC tests.
- Anhuienoside E has the potential to inhibit Gtf and MurA enzymes based on In silico molecular docking study.
GRAPHICAL ABSTRACT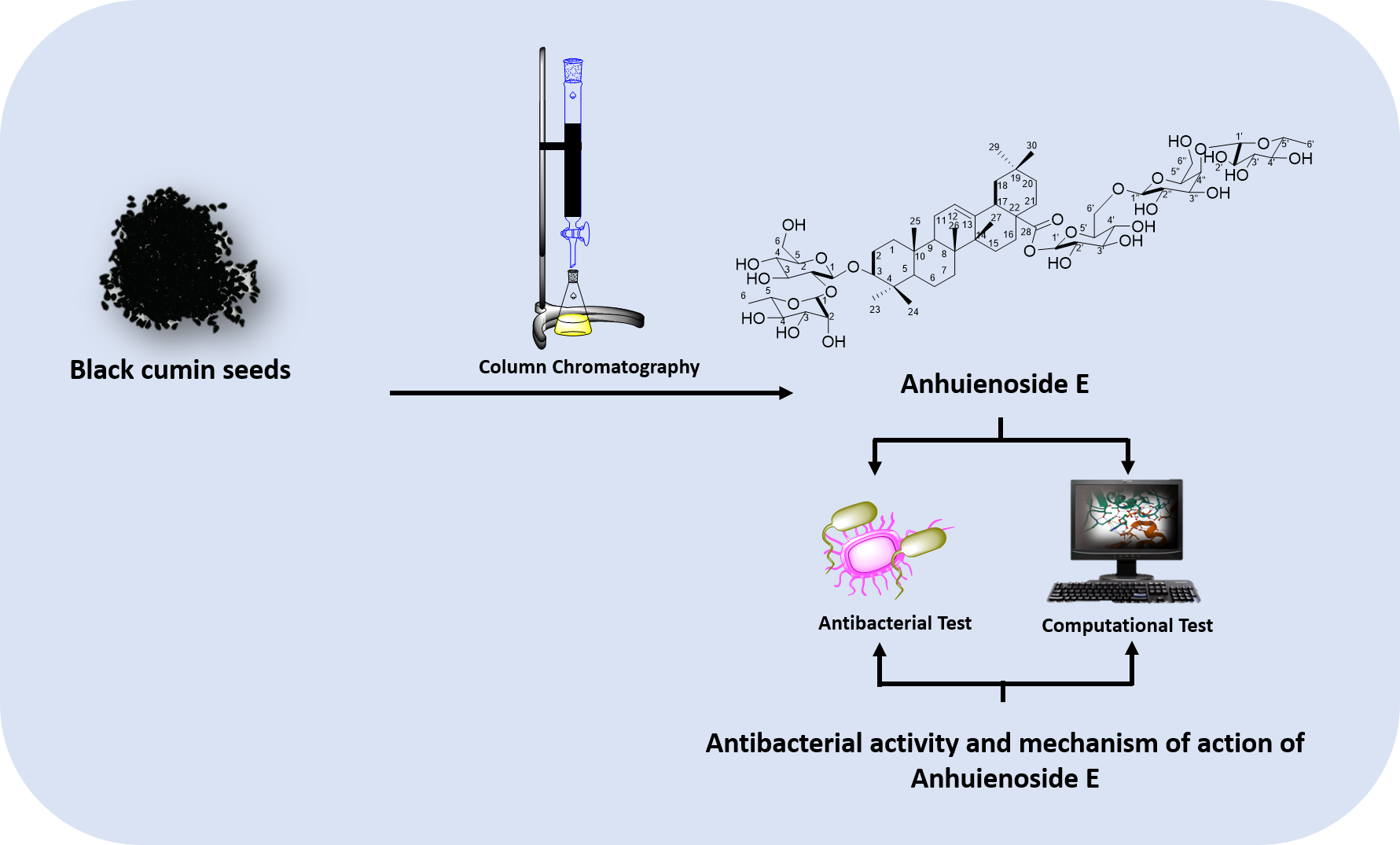
Downloads
References
R Amalia, F Chairunisa, MF Alfian and A Supartinah. Indonesia: Epidemiological profiles of early childhood caries. Frontiers in Public Health 2019; 7, 210.
JA Lemos, SR Palmer, L Zeng, ZT Wen, JK Kajfasz, IA Freires, J Abranches and LJ Brady. The Biology of Streptococcus mutans. Microbiology Spectrum 2019; 7(1), 1-18.
S Javed, M Zakirulla, RU Baig, SM Asif and AB Meer. Development of artificial neural network model for prediction of post-streptococcus mutans in dental caries. Computer Methods and Programs in Biomedicine 2020; 186, 105198.
B Zhu, LC Macleod, T Kitten and P Xu. Streptococcus sanguinis biofilm formation & interaction with oral pathogens. Future Microbiology 2018; 13(8), 915-932.
L Xin, Z Hu, R Han, X Xu, C Wang, D Li, Y Guo and F Hu. Asp50Glu mutation in MurA results in fosfomycin resistance in Enterococcus faecium. Journal of Global Antimicrobial Resistance 2022; 30, 50-55.
S Huang, M Wu, Y Li, J Du, S Chen, S Jiang, X Huang and L Zhan. The dlt operon contributes to the resistance to chlorhexidine in streptococcus mutans. International Journal of Antimicrobial Agents 2022; 59(3), 106540.
Y Lin, J Chen, X Zhou and Y Li. Inhibition of Streptococcus mutans biofilm formation by strategies targeting the metabolism of exopolysaccharides. Critical Reviews in Microbiology 2021; 47(5), 667-677.
MM Miyachiro, D Granato, DM Trindade, C Ebel, AFP Leme and A Dessen. Complex formation between mur enzymes from streptococcus pneumoniae. Biochemistry 2019; 58(30), 3314-3324.
LD Garbinski, BP Rosen and M Yoshinaga. Organoarsenicals inhibit bacterial peptidoglycan biosynthesis by targeting the essential enzyme MurA. Chemosphere 2020; 254, 126911.
E Sutrisna, S Wahyuni and A Fitriani. Antibacterial effect of nigella sativa L. seed from Indonesia. Pharmacognosy Journal 2022; 14(6), 1029-1032.
M Ikhsan, N Hiedayati, K Maeyama and F Nurwidya. Nigella sativa as an anti-inflammatory agent in asthma. BMC Research Notes 2018; 11, 744.
D Kurnia, R Padilah, E Apriyanti and HDA Dharsono. Phytochemical analysis and anti-biofilm potential that cause dental caries from black cumin seeds (nigella sativa linn.). Drug Design, Development and Therapy 2024; 18, 1917-1932.
A Shah, S Akhtar, F Mahmood, S Urooj, AB Siddique, MI Irfan, M Naeem-Ul-Hassan, M Sher, A Alhoshani, A Rauf, HMA Amin and A Abbas. Fagonia arabica extract-stabilized gold nanoparticles as a highly selective colorimetric nanoprobe for Cd2+ detection and as a potential photocatalytic and antibacterial agent. Surfaces and Interfaces 2024; 51, 104556.
S Ullah, R Khalid, MF Rehman, MI Irfan, A Abbas, A Alhoshani, F Anwar and HMA Amin. Biosynthesis of phyto-functionalized silver nanoparticles using olive fruit extract and evaluation of their antibacterial and antioxidant properties. Frontiers in Chemistry 2023; 11, 1202252.
A Jabbar, A Abbas, N Assad, M Naeem-ul-Hassan, HA Alhazmi, A Najmi, K Zoghebi, MA Bratty, A Hanbashi and HMA Amin. A highly selective Hg2+ colorimetric sensor and antimicrobial agent based on green synthesized silver nanoparticles using Equisetum diffusum extract. RSC Advances 2023; 13(41), 28666-28675.
AB Siddique, MA Shaheen, A Abbas, Y Zaman, MA Bratty, A Najmi, A Hanbashi, M Mustaqeem, HA Alhazmi, Z ur Rehman, K Zoghebi and HMA Amin. Thermodynamic and kinetic insights into azo dyes photocatalytic degradation on biogenically synthesized ZnO nanoparticles and their antibacterial potential. Heliyon 2024; 10(23), e40679.
AF Majdalawieh, SM Yousef and IA Abu-Yousef. Thymoquinone, a major constituent in Nigella sativa seeds, is a potential preventative and treatment option for atherosclerosis. European Journal of Pharmacology 2021; 909, 174420.
MF Mahomoodally, MZ Aumeeruddy, LJ Legoabe, D Montesano and G Zengin. Nigella sativa L. and its active compound thymoquinone in the clinical management of diabetes: A systematic review. International Journal of Molecular Sciences 2022; 23(20), 12111.
P Gnanasekaran, A Roy, NS Natesh, V Raman, P Ganapathy and MK Arumugam. Removal of microbial pathogens and anticancer activity of synthesized nano-thymoquinone from Nigella sativa seeds. Environmental Technology & Innovation 2021; 24, 102068.
W Huang. Open tubular ion chromatography: A state-of-the-Art review. Analytica Chimica Acta 2021; 1143, 210-224.
S Segan, D Opsenica and D Milojkovic-Opsenica. Thin-layer chromatography in medicinal chemistry. Journal of Liquid Chromatography and Related Technologies 2019; 42(9-10), 238-248.
IA Evangelina, Y Herdiyati, A Laviana, R Rikmasari, C Zubaedah, Anisah and D Kurnia. Bio-mechanism inhibitory prediction of β-sitosterol from kemangi (ocimum basilicum L.) as an inhibitor of murA enzyme of oral bacteria: In vitro and in silico Study. Advances and Applications in Bioinformatics and Chemistry 2021; 14, 103-115.
JR Hanson. A hundred years in the elucidation of the structures of natural products. Science Progress 2017; 100(1), 63-79.
P Parvekar, J Palaskar, S Metgud, R Maria and S Dutta. The minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of silver nanoparticles against Staphylococcus aureus. Biomaterial Investigations in Dentistry 2020; 7(1), 105-109.
N Schormann, M Patel, L Thannickal, S Purushotham, R Wu, JL Mieher, H Wu and C Deivanayagam. The catalytic domains of streptococcus mutans glucosyltransferases: A structural analysis. Acta Crystallographica Section F: Structural Biology Communications 2023; 79, 119-127.
D Kurnia, GS Hutabarat, D Windaryanti, T Herlina, Y Herdiyati and MH Satari. Potential allylpyrocatechol derivatives as antibacterial agent against oral pathogen of S. Sanguinis ATCC 10,556 and as inhibitor of MurA Enzymes: In vitro and in silico study. Drug Design, Development and Therapy 2020; 14, 2977-2985.
OV Sobolev, PV Afonine, NW Moriarty, ML Hekkelman, RP Joosten, A Perrakis and PD Adams. A global ramachandran score identifies protein structures with unlikely stereochemistry. Structure 2020; 28(11), 1249-1258.
W Tian, C Chen, X Lei, J Zhao and J Liang. CASTp 3.0: Computed atlas of surface topography of proteins. Nucleic Acids Research 2018; 46(W1), W363-W367.
MS Valdes-Tresanco, ME Valdes-Tresanco, PA Valiente and E Moreno. AMDock: A versatile graphical tool for assisting molecular docking with autodock vina and autodock4. Biology Direct 2020; 15, 12.
LLG Ferreira and AD Andricopulo. ADMET modeling approaches in drug discovery. Drug Discovery Today 2019; 24(5), 1157-1165.
A Daina, O Michielin and V Zoete. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Scientific Reports 2017; 7, 42717.
C Jia, J Li, G Hao and G Yang. A drug-likeness toolbox facilitates ADMET study in drug discovery. Drug Discovery Today 2020; 25(1), 248-258.
Y Bibi, S Nisa, FM Chaudhary and M Zia. Antibacterial activity of some selected medicinal plants of Pakistan. BMC Complementary and Alternative Medicine 2011; 11, 52.
MD Awouafack, LJ McGaw, S Gottfried, R Mbouangouere, P Tane, M Spiteller and JN Eloff. Antimicrobial activity and cytotoxicity of the ethanol extract, fractions and eight compounds isolated from Eriosema robustum (Fabaceae). BMC Complementary and Alternative Medicine 2013; 13, 289.
T Scrob, A Hosu and C Cimpoiu. Trends in analysis of vegetables by high performance TLC. Journal of Liquid Chromatography and Related Technologies 2019; 42(9-10), 249-257.
A Pyka. Detection progress of selected drugs in TLC. BioMed Research International 2014; 2014(1), 732078.
M Asemani and AR Rabbani. Detailed FTIR spectroscopy characterization of crude oil extracted asphaltenes: Curve resolve of overlapping bands. Journal of Petroleum Science and Engineering 2020; 185, 106618.
Y Huang, X Li, X Peng, AT Adegoke, J Chen, H Su, G Hu, G Wei and M Qiu. NMR-based structural classification, identification, and quantification of triterpenoids from edible mushroom ganoderma resinaceum. Journal of Agricultural and Food Chemistry 2020; 68(9), 2816-2825.
Q Shi, J Yan, B Jiang, X Chi, J Wang, X Liang and X Ai. A general strategy for the structural determination of carbohydrates by multi-dimensional NMR spectroscopies. Carbohydrate Polymers 2021; 267, 118218.
S Qiu, N Van Hung, LT Xuan, J Gu, E Lobkovsky, TC Khanh, DD Soejarto, J Clardy, JM Pezzuto, Y Dong, MV Tri, LM Huong and HHS Fong. A pregnane steroid from Aglaia lawii and structure confirmation of cabraleadiol monoacetate by X-ray crystallography. 2001; 56(7), 775-780.
A Furevi, A Ruda, TA D’Ortoli, H Mobarak, J Stahle, C Hamark, C Fontana, O Engstrom, P Apostolica and G Widmalm. Complete 1H and 13C NMR chemical shift assignments of mono-to tetrasaccharides as basis for NMR chemical shift predictions of oligo- and polysaccharides using the computer program CASPER. Carbohydrate Research 2022; 513, 108528.
S Belarbi, M Vivier, W Zaghouani, AD Sloovere, V Agasse-Peulon and P Cardinael. Comparison of new approach of GC-HRMS (Q-Orbitrap) to GC–MS/MS (triple-quadrupole) in analyzing the pesticide residues and contaminants in complex food matrices. Food Chemistry 2021; 359, 129932.
WC Ye, QW Zhang, SX Zhao and CT Che. Four new oleanane saponins from anemone anhuiensis. Chemical and Pharmaceutical Bulletin 2001; 49(5), 632-634.
SB Tankeo, F Damen, MD Awouafack, J Mpetga, P Tane, JN Eloff and V Kuete. Antibacterial activities of the methanol extracts, fractions and compounds from Fagara tessmannii. Journal of Ethnopharmacology 2015; 169, 275-279.
PY Chung. Novel targets of pentacyclic triterpenoids in staphylococcus aureus: A systematic review. Phytomedicine 2020; 73, 152933.
M Zhao, N Zhang, T Gao, J Jin, T Jing, J Wang, Y Wu, X Wan, W Schwab and C Song. Sesquiterpene glucosylation mediated by glucosyltransferase UGT91Q2 is involved in the modulation of cold stress tolerance in tea plants. New Phytologist 2020; 226(2), 362-372.
SW Park, BH Lee, SH Song, and MK Kim. Revisiting the Ramachandran plot based on statistical analysis of static and dynamic characteristics of protein structures. Journal of Structural Biology 2023; 215, 107939.
P Chakrabarti and D Pal. The interrelationships of side-chain and main-chain conformations in proteins 2001; 76(1-2), 1-102.
GD Rose. Ramachandran maps for side chains in globular proteins. Proteins: Structure, Function and Bioinformatics 2019; 87(5), 357-364.
KD Wild, F Porrecaf, HI Yamamurat and RB Raffa. Differentiation of receptor subtypes by thermodynamic analysis: Application to opioid 6 receptors 1994; 91(25), 12018-12021.
F Azimi, H Azizian, M Najafi, F Hassanzadeh, H Sadeghi-aliabadi, JB Ghasemi, M Ali Faramarzi, S Mojtabavi, B Larijani, L Saghaei and M Mahdavi. Design and synthesis of novel quinazolinone-pyrazole derivatives as potential α-glucosidase inhibitors: Structure-activity relationship, molecular modeling and kinetic study. Bioorganic Chemistry 2021; 114, 105127.
J Song, X Jin, XC Wang and P Jin. Preferential binding properties of carboxyl and hydroxyl groups with aluminium salts for humic acid removal. Chemosphere 2019; 234, 478-487.
F Ntie-Kang. An in silico evaluation of the ADMET profile of the StreptomeDB database. SpringerPlus 2013; 2, 353.
TJ Hou and XJ Xu. ADME evaluation in drug discovery. 3. modeling blood-brain barrier partitioning using simple molecular descriptors. Journal of Chemical Information and Computer Sciences 2003; 43(6), 2137-2152.
L Zhu, J Zhao, Y Zhang, W Zhou, L Yin, Y Wang, Y Fan, Y Chen and H Liu. ADME properties evaluation in drug discovery: In silico prediction of blood–brain partitioning. Molecular Diversity 2018; 22, 979-990.
T Siswina, MM Rustama, D Sumiarsa, E Apriyanti, H Dohi and D Kurnia. Antifungal constituents of piper crocatum and their activities as ergosterol biosynthesis inhibitors discovered via in silico study using ADMET and drug-likeness analysis. Molecules 2023; 28(23), 7705.
CA Lipinski. Lead- and drug-like compounds: The rule-of-five revolution. Drug Discovery Today: Technologies 2004; 1(4), 337-341.
MP Pollastri. Overview on the rule of five. Current Protocols in Pharmacology 2010; 49(1), 9.12.1-9.12.8.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






