Analyzing the Antibacterial, Anticancer, and Antioxidant Qualities of the Stink Bean (Parkia Speciosa) by Various Extraction Techniques
DOI:
https://doi.org/10.48048/tis.2024.7994Keywords:
Stink bean, Parkia speciosa, Microwave-assisted water extraction, Boiling water extraction, Biochemical propertiesAbstract
In various Southeast Asian countries, people have long incorporated the young green seeds of Parkia speciosa, commonly known as stink beans, into their culinary traditions. These seeds can be prepared using various cooking methods, including boiling, microwaving, stir-frying, or even consuming them raw. While several studies have explored the pharmaceutical properties of raw stink beans, this study seeks to expand our knowledge of how different extraction methods can influence the release of bioactive compounds from these young stink bean seeds. Three distinct extraction methods, namely (1) microwave-assisted water extraction, (2) boiling water extraction and (3) 50 % ethanol extraction were employed in this research. The study focused on total phenolic content and antioxidant activities, with the 50 % ethanol extraction method yielding the most promising results. Moreover, only the extract obtained from the 50 % ethanol method exhibited antimicrobial activity against 4 gram-positive bacteria, including Methicillin-Resistant Staphylococcus aureus, Staphylococcus aureus, Bacillus cereus, Micrococcus luteus and 1 gram-negative bacterium, Salmonella typhi. Notably, extracts obtained from the 3 extraction methods of young stink bean seeds exhibit varying degrees of inhibition of cancer cell growth with low cell toxicity. Taken together, our findings suggest that distinct extraction methods play a key role in obtaining different sets of bioactive compounds from the young seeds of stink beans. Therefore, various cooking methods, including stir-frying, microwaving and pickling in alcohol, may offer diverse sources of bioactive compounds for promoting consumer health.
HIGHLIGHTS
- A 50 % ethanol extraction of young stink bean seeds yields the highest concentrations of phenolic compounds and exhibits the highest antioxidant capacity
- The sole extract derived from the 50 % ethanol extraction of young stink bean seeds demonstrated notable antimicrobial efficacy against both selected gram-positive and gram-negative bacteria
- Three distinct extraction methodologies—microwave-assisted water extraction, boiling water extraction and 50 % ethanol extraction—exhibited varying degrees of cytotoxicity against normal cell lines and cancer cells
GRAPHICAL ABSTRACT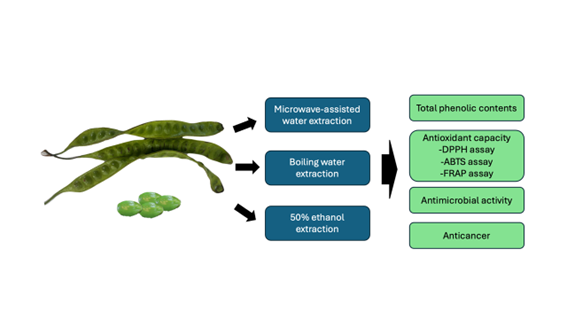
Downloads
References
TK Lim. Edible medicinal and non-medicinal plants. Vol. 1. Springer, Dordrecht, Netherlands, 2012, p. 835.
HCF Hopkins. The indo-pacific species of Parkia (Leguminosae: Mimosoideae). Kew Bulletin, 1994; 49, 181-234.
M Abdullah and T Lim. Some physical properties of Parkia speciosa seeds. In: Proceedings of the 2011 International Conference on Food Engineering and Biotechnology, Singapore. 2011, p. 1-5.
Y Kamisah, F Othman, HMS Qodriyah and K Jaarin. Parkia speciosa hassk.: A potential phytomedicine. Evid. base Compl. Alternative Med. 2013; 2013, 709028.
P Maisuthisakula, S Pasukb and P Ritthiruangdej. Relationship between antioxidant properties and chemical composition of some Thai plants. J. Food Compos. Anal. 2008; 21, 229-40.
KH Miean and S Mohamed. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. J. Agr. Food Chem. 2001; 49, 3106-12.
P Tangkanakul, G Trakoontivakorn, J Saengprakai, P Auttaviboonkul, B Niyomwit, N Lowvitoon and K Nakahara. Antioxidant capacity and antimutagenicity of thermal processed Thai foods. Jpn. Agric. Res. Q. 2011; 45, 211-8.
SFS Reihani and ME Azhar. Antioxidant activity and total phenolic content in aqueous extracts of selected traditional Malay salads (Ulam). Int. Food Res. J. 2012; 19, 1439-44.
A Murakami, AM Ali, K Mat-Salleh, K Koshimizu and H Ohigashi. Screening for the in vitro anti-tumor-promoting activities of edible plants from Malaysia. Biosci. Biotechnol. Biochem. 2000; 64, 9-16.
AFA Aisha, KM Abu-Salah, Y Darwis and AMSA Majid. Screening of antiangiogenic activity of some tropical plants by rat aorta ring assay. Int. J. Pharmacol. Res. 2009; 5, 370-6.
AFA Aisha, KM Abu-Salah, SA Alrokayan, Z Ismail and AMS Abdulmajid. Evaluation of antiangiogenic and antoxidant properties of Parkia speciosa Hassk extracts. Pakistan journal of pharmaceutical sciences. Pak. J. Pharm. Sci. 2012; 25, 7-14.
AM Uyub, IN Nwachukwu, ARA Azlan and SS Fariza. In-vitro antibacterial activity and cytotoxicity of selected medicinal plant extracts from Penang Island Malaysia on metronidazole-resistant-Helicobacter pylori and some pathogenic bacteria. Ethnobot. Res. Appl. 2010; 8, 95-106.
N Musa, LS Wei, TS Chuah, W Wee and LK Leong. Potential of edible plants as remedies of systemic bacterial disease infection in cultured fish. Global J. Pharmacol. 2008; 2, 31-6.
HL Siow and CY Gan. Extraction of antioxidative and antihypertensive bioactive peptides from Parkia speciosa seeds. Food Chem. 2013; 141, 3435-42.
Y Asikin, Kusumiyati, T Shikanai and K Wada. Volatile aroma components and MS-based electronic nose profiles of dogfruit (Pithecellobium jiringa) and stink bean (Parkia speciosa). J. Adv. Res. 2018; 9, 79-85.
HJ Ko, LH Ang and LT Ng. Antioxidant activities and polyphenolic constituents of bitter bean Parkia speciosa. Int. J. Food Prop. 2014; 17, 1977-86.
M Takó, EB Kerekes, C Zambrano, A Kotogán, T Papp, J Krisch and C Vágvölgyi. Plant phenolics and phenolic-enriched extracts as antimicrobial agents against food-contaminating microorganisms. Antioxidants 2020; 9, 165.
MC Lima, CPD Sousa, C Fernandez-Prada, J Harel, JD Dubreuil and ELD Souza. A review of the current evidence of fruit phenolic compounds as potential antimicrobials against pathogenic bacteria. Microb. Pathog. 2019; 130, 259-70.
MM Vuolo, VS Lima and MRM Junior. Phenolic compounds: Structure, classification, and antioxidant power. In: MRS Campos (Ed.). Bioactive compounds. Woodhead Publishing, Sawton, 2019, p. 33-50.
S Mathew, TE Abraham and ZA Zakaria. Reactivity of phenolic compounds towards free radicals under in vitro conditions. J. Food Sci. Technol. 2015; 52, 5790-8.
C Rice-Evans, N Miller and G Paganga. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997; 2, 152-9.
J Azmir, ISM Zaidul, MM Rahman, KM Sharif, A Mohamed, F Sahena, MHA Jahurul, K Ghafoor, NAN Norulaini and AKM Omar. Techniques for extraction of bioactive compounds from plant materials: A review. Int. J. Food Eng. 2013; 117, 426-36.
T Lefebvre, E Destandau and E Lesellier. Selective extraction of bioactive compounds from plants using recent extraction techniques: A review. J. Chrom. 2021; 1635, 461770.
I Usman, M Hussain, A Imran, M Afzaal, F Saeed, M Javed, A Afzal, I Ashfaq, EA Jbawi and SA Saewan. Traditional and innovative approaches for the extraction of bioactive compounds. Int. J. Food Prop. 2022; 25, 1215-33.
MY Kim, GY Jang, Y Lee, M Li, YM Ji, N Yoon, SH Lee, KM Kim, J Lee and HS Jeong. Free and bound form bioactive compound profiles in germinated black soybean (Glycine max L.). Food Sci. Biotechnol. 2016; 25, 1551-9.
PP McCue and K Shetty. A role for amylase and peroxidase-linked polymerization in phenolic antioxidant mobilization in dark-germinated soybean and implications for health. Process Biochem. 2004; 39, 1785-91.
P Saharan, PK Sadh and JS Duhan. Assessment of fermentation based enrichment of bioactive compounds and antioxidant activity of commonly used cereals. Int. J. Food. Ferment. Technol. 2018; 8, 175-84.
BB Li, B Smith and MM Hossain. Extraction of phenolics from citrus peels: II. Enzyme-assisted extraction method. Separ. Purif. Tech. 2006. 48(2): p. 189-196.
MR Meini, I Cabezudo, CE Boschetti and D Romanini. Recovery of phenolic antioxidants from Syrah grape pomace through the optimization of an enzymatic extraction process. Food Chem. 2019; 283, 257-64.
Y Zhu, T Li, X Fu, M Brennan, AM Abbasi, B Zheng and RH Liu. The use of an enzymatic extraction procedure for the enhancement of highland barley (Hordeum vulgare L.) phenolic and antioxidant compounds. Int. J. Agr. Food Sci. Tech. 2016; 51, 1916-24.
Y Aguilera, M Rebollo-Hernanz, S Cañas, D Taladrid and MA Martín-Cabrejas. Heat assisted extraction of phenolic compounds from Eleutherine bulbosa (Mill.) bulb and its bioactive profiles using response surface methodology. Ind. Crop. Prod. 2020; 144, 112064.
Y Aguilera, M Rebollo-Hernanz, S Cañas, D Taladrid and MA Martín-Cabrejas. Response surface methodology to optimise the heat-assisted aqueous extraction of phenolic compounds from coffee parchment and their comprehensive analysis. Food Funct. 2019; 10, 4739-50.
Z Rafiee, SM Jafari, M Alami and M Khomeiri. Microwave-assisted extraction of phenolic compounds from olive leaves; a comparison with maceration. J. Anim. Plant Sci. 2011; 21, 738-45.
H Li, Z Deng, T Wu, R Liu, S Loewen and R Tsao. Microwave-assisted extraction of phenolics with maximal antioxidant activities in tomatoes. Food Chem. 2012; 130, 928-36.
YT Tung, WC Chang, PS Chen, TC Chang and ST Chang. Ultrasound‐assisted extraction of phenolic antioxidants from Acacia confusa flowers and buds. J. Sep. Sci. 2011; 34, 844-51.
C Corbin, T Fidel, EA Leclerc, E Barakzoy, N Sagot, A Falguiéres, S Renouard, JP Blondeau, C Ferroud, J Doussot, E Lainé and C Hano. Development and validation of an efficient ultrasound assisted extraction of phenolic compounds from flax (Linum usitatissimum L.) seeds. Ultrason. Sonochem. 2015; 26, 176-85.
SH Sung, KH Kim, BT Jeon, SH Cheong, J Park and DH Kim. Antibacterial and antioxidant activities of tannins extracted from agricultural by-products. J. Med. Plant Res. 2012; 6, 3072-9.
A Alupului, I Calinescu and V Lavric. Ultrasonic vs. microwave extraction intensification of active principles from medicinal plants. AIDIC Conf. Ser. 2009; 9, 8.
EA Ainsworth and KM Gillespie. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protoc. 2007; 2, 875-7.
TJ Herald, P Gadgil and M Tilley. High‐throughput micro plate assays for screening flavonoid content and DPPH‐scavenging activity in sorghum bran and flour. J. Sci. Food Agr. 2012; 92, 2326-31.
A Nowak, A Klimowicz, W Duchnik, Ł Kucharski, K Florkowska, A Muzykiewicz, D Wira, J Zielonkabrzezicka, A Siedłowska and K Nadarzewska. Application of green-extraction technique to evaluate of antioxidative capacity of wild population of fireweed (Epilobium angustifolium). Herba Pol. 2019; 65, 18-30.
K Thaipong, U Boonprakob, K Crosby, L Cisneros-Zevallos and DH Byrne. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 2006; 19, 669-75.
BD Craft, AL Kerrihard, R Amarowicz and RB Pegg. Phenol-based antioxidants and the in vitro methods used for their assessment. Compr. Rev. Food Sci. Food Saf. 2012; 11, 148-73.
J Gao, Z Yang, Zhao C, X Tang, Q Jiang and Y Yin. A comprehensive review on natural phenolic compounds as alternatives to in-feed antibiotics. Sci. China Life Sci. 2023; 66, 1518-34.
FMF Roleira, EJ Tavares-da-Silva, CL Varela, SC Costa, T Silva, J Garrido and F Borges. Plant derived and dietary phenolic antioxidants: Anticancer properties. Food Chem. 2015; 183, 235-58.
MA Asgar. Anti-diabetic potential of phenolic compounds: A review. Int. J. Food Prop. 2013; 16, 91-103.
S Wonghirundecha, S Benjakul, and P Sumpavapol. Total phenolic content, antioxidant and antimicrobial activities of stink bean (Parkia speciosa Hassk.) pod extracts. Songklanakarin J. Sci. Technol. 2014; 36, 301-8.
Y Gao, S Ma, M Wang and XY Feng. Characterization of free, conjugated, and bound phenolic acids in seven commonly consumed vegetables. Molecules 2017; 22, 1878.
L Štěrbová, PH Čepková, I Viehmannová and DC Huansi. Effect of thermal processing on phenolic content, tocopherols and antioxidant activity of sacha inchi kernels. J. Food Process. Preserv. 2017; 41, e12848.
D Jacobo‐Velázquez and L Cisneros‐Zevallos. Correlations of antioxidant activity against phenolic content revisited: A new approach in data analysis for food and medicinal plants. J. Food Sci. 2009; 74, R107-R113.
P Terpinc, B Čeh, NP Ulrih and H Abramovič. Studies of the correlation between antioxidant properties and the total phenolic content of different oil cake extracts. Ind. Crop. Prod. 2012; 39, 210-7.
G Piluzza and S Bullitta. Correlations between phenolic content and antioxidant properties in twenty-four plant species of traditional ethnoveterinary use in the Mediterranean area. Pharmaceut. Biol. 2011; 49, 240-7.
E Roselló-Soto, FJ Martí-Quijal, A Cilla, PES Munekata, JM Lorenzo, F Remize and FJ Barba. Influence of temperature, solvent and pH on the selective extraction of phenolic compounds from tiger nuts by-products: Triple-TOF-LC-MS-MS characterization. Molecules 2019; 24, 797.
A Ghasemzadeh, HZE Jaafar, MFM Bukhori, MH Rahmat and A Rahmat. Assessment and comparison of phytochemical constituents and biological activities of bitter bean (Parkia speciosa Hassk.) collected from different locations in Malaysia. Chem. Cent. J. 2018; 12, 12.
N Kakatum and Y Sudjaroen. Screening of anti-tyrosinase and anti-bacterial activities of ethanolic extract from stink bean (Parkia speciosa Hassk) seed. Int. J. Pharmaceut. Res. 2020; 12, 3583.
F Cadoná, R Dantas, GHD Mello and F Silva-Jr. Natural products targeting into cancer hallmarks: An update on caffeine, theobromine, and (+)-catechin. Crit. Rev. Food Sci. Nutr. 2022; 62, 7222-41.
S Liu, AR Khan, X Yang, B Dong, J Ji and G Zhai. The reversal of chemotherapy-induced multidrug resistance by nanomedicine for cancer therapy. J. Contr. Release 2021; 335, 1-20.
J Wang and YF Jiang. Natural compounds as anticancer agents: Experimental evidence. World J. Exp. Med. 2012; 2, 45-57.
A Naeem, P Hu, M Yang, J Zhang, Y Liu, W Zhu and Q Zheng. Natural products as anticancer agents: Current status and future perspectives. Molecules 2022; 27, 8367.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






