Recombinant Bacteriocin Rhamnosin Suppresses Malignant Phenotypes of Colorectal Cancer Cells
DOI:
https://doi.org/10.48048/tis.2025.10057Keywords:
Bacteriocins, Lactic acid bacteria, Lactobacillus rhamnosus, Recombinant rhamnosin, Colorectal cancer, anticancer, Cell proliferation, Cell motility, ApoptosisAbstract
Colorectal cancer (CRC), the third most diagnosed cancer worldwide, is believed to worsened by microbiome dysbiosis, which promotes carcinogenesis through inflammation and genetic instability. Lactic acid bacteria (LAB), known for their beneficial metabolites, produce bacteriocins with potential anticancer properties. Here we hypothesized that recombinant rhamnosin (rRN), derived from the prebacteriocin gene of Lactobacillus rhamnosus, may inhibit CRC progression by targeting oncogenic pathways associated to proliferation, metastasis, and chemoresistance. We evaluated the anticancer activity of rRN in HT-29 and Caco-2 CRC cells, along with normal oral keratinocyte (NOK-SI) cells, using cell viability (MTT assay), apoptosis (Hoechst/propidium iodide dual nuclear staining), migration and invasion (Boyden chamber assay), and RT-qPCR to measure the expression of genes regulating growth, motility, and drug resistance. The results revealed that rRN dose-dependently inhibited CRC cell viability (IC50: 25.3 and 14.5 µg/mL for HT-29 and Caco-2, respectively) with minimal toxicity to NOK-SI cells (IC50: >32 µg/mL). When combined with 5-fluorouracil (5-FU), it enhanced the growth inhibitory effect. rRN induced apoptosis and suppressed migration and invasion by 70 - 80 %. Transcriptional profiling revealed the downregulation of genes related to cell growth (CCNB1, CCND1 and EGFR), motility (FN1, PXN and SNAI1), and drug resistance (ABCG2, MRP1 and MRP2), while CCNE1 and VIM remained unaffected, underscoring rRN’s selective targeting of CRC progression pathways. These findings establish rRN as a multitarget therapeutic candidate for CRC, addressing proliferation, metastasis, and chemoresistance - key challenges in CRC relapse. Future studies should validate protein-level mechanisms, such as caspase activation and BCL-2/Bax dynamics, and evaluate rRN’s efficacy in preclinical models. This work underscores the potential of rRN, a LAB-derived bacteriocin, as a novel adjuvant for CRC treatment.
HIGHLIGHTS
- Gut microbiome imbalance promotes colorectal cancer (CRC) development, while lactic acid bacteria (LAB) produce beneficial bacteriocins with potential anticancer properties.
- This study produced recombinant rhamnosin (rRN), derived from the prebacteriocin gene of Lactobacillus rhamnosus, a type of LAB.
- rRN exhibits anticancer effects by inhibiting CRC cell proliferation, enhancing chemosensitivity, inducing apoptosis, and suppressing migration and invasion.
- rRN modulates oncogenic pathways by downregulating genes associated with cell growth (CCNB1, CCND1 and EGFR), motility (FN1, PXN and SNAI1), and drug resistance (ABCG2, MRP1 and MRP2).
GRAPHICAL ABSTRACT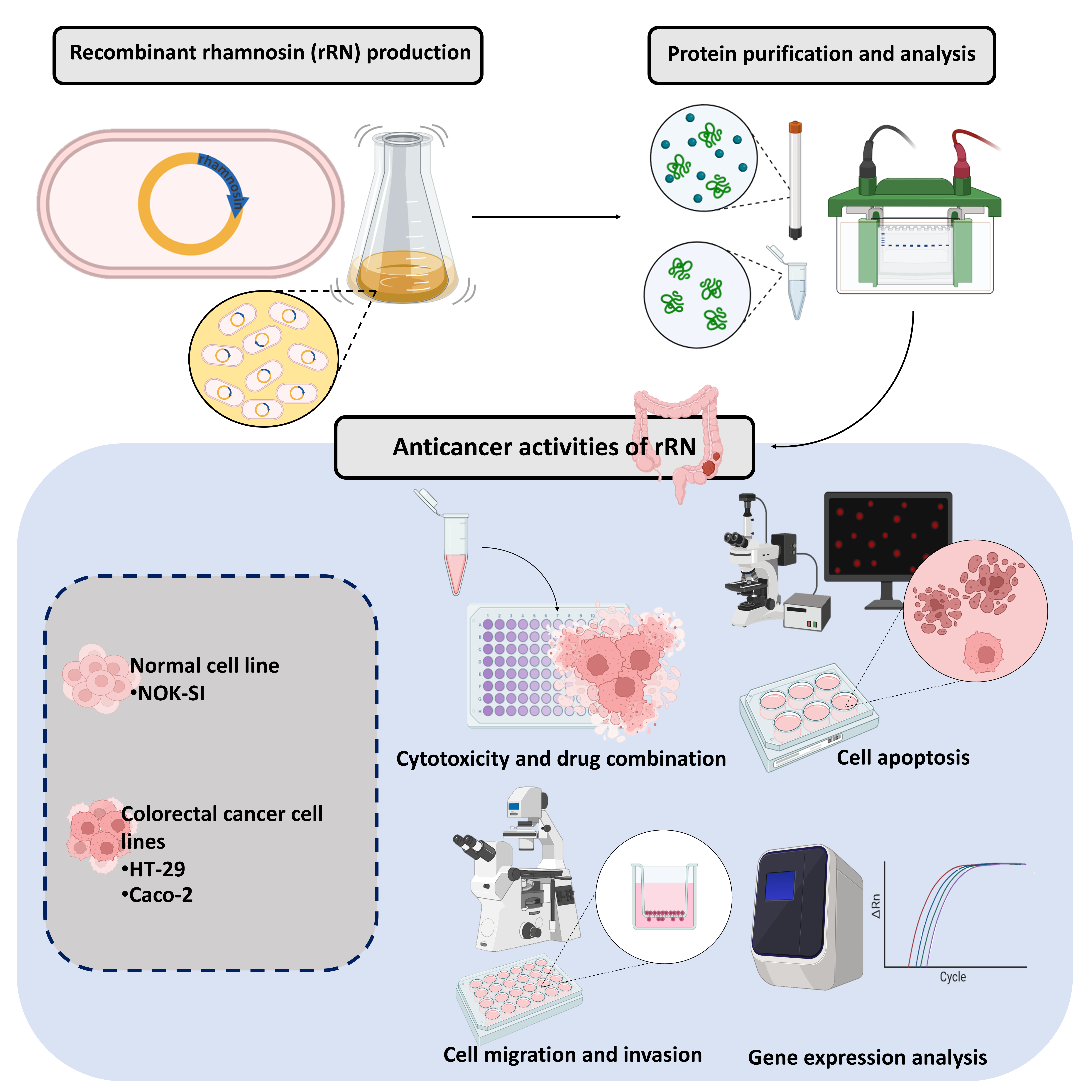
Downloads
References
F Bray, J Ferlay, I Soerjomataram, RL Siegel, LA Torre and A Jemal. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. A Cancer Journal for Clinicians 2018; 68(6), 394-424.
J Douaiher, A Ravipati, B Grams, S Chowdhury, O Alatise and C Are. Colorectal cancer-global burden, trends, and geographical variations. Journal of Surgical Oncology 2017; 115(5), 619-630.
T Sawicki, M Ruszkowska, A Danielewicz, E Niedzwiedzka, T Arlukowicz and KE Przybylowicz. A review of colorectal cancer in terms of epidemiology, risk factors, development, symptoms and diagnosis. Cancers 2021; 13(9), 2025.
H Fadlallah, J El Masri, H Fakhereddine, J Youssef, C Chemaly, S Doughan and W Abou-Kheir. Colorectal cancer: Recent advances in management and treatment. World Journal of Clinical Oncology 2024; 15(9), 1136-1156.
WA Hammond, A Swaika and K Mody. Pharmacologic resistance in colorectal cancer: A review. Therapeutic Advances in Medical Oncology 2016; 8(1), 57-84.
J Kaur, J Elms, AL Munn, D Good and MQ Wei. Immunotherapy for non-small cell lung cancer (NSCLC), as a stand-alone and in combination therapy. Critical Reviews in Oncology /Hematology 2021; 164, 103417.
TR Klaenhammer. Bacteriocins of lactic acid bacteria. Biochimie 1988; 70(3), 337-349.
F De Filippis, E Pasolli and D Ercolini. The food-gut axis: Lactic acid bacteria and their link to food, the gut microbiome and human health. FEMS Microbiology Reviews 2020; 44(4), 454-489.
E Pessione. Lactic acid bacteria contribution to gut microbiota complexity: Lights and shadows. Frontiers in Cellular and Infection Microbiology 2012; 2, 86.
A Darbandi, A Asadi, M Mahdizade Ari, E Ohadi, M Talebi, M Halaj Zadeh, A Darb Emamie, R Ghanavati and M Kakanj. Bacteriocins: Properties and potential use as antimicrobials. Journal of Clinical Laboratory Analysis 2022; 36(1), e24093.
SC Yang, CH Lin, CT Sung and JY Fang. Antibacterial activities of bacteriocins: Application in foods and pharmaceuticals. Frontiers in Microbiology 2014; 5, 241.
M Zimina, O Babich, A Prosekov, S Sukhikh, S Ivanova, M Shevchenko and S Noskova. Overview of global trends in classification, methods of preparation and application of bacteriocins. Antibiotics 2020; 9(9), 553.
S Kaur and S Kaur. Bacteriocins as potential anticancer agents. Frontiers in Pharmacology 2015; 6, 272.
Y Wang, Y Wang, T Sun and J Xu. Bacteriocins in cancer treatment: Mechanisms and clinical potentials. Biomolecules 2024; 14(7), 831.
AM Molujin, S Abbasiliasi, A Nurdin, PC Lee, JA Gansau and R Jawan. Bacteriocins as potential therapeutic approaches in the treatment of various cancers: A review of in vitro studies. Cancers 2022; 14(19), 4758.
SD Todorov, CM Dioso, MT Liong, LA Nero, K Khosravi-Darani and IV Ivanova. Beneficial features of pediococcus: From starter cultures and inhibitory activities to probiotic benefits. World Journal of Microbiology and Biotechnology 2022; 39(1), 4.
H Zhao, R Sood, A Jutila, S Bose, G Fimland, J Nissen-Meyer and PK Kinnunen. Interaction of the antimicrobial peptide pheromone Plantaricin A with model membranes: Implications for a novel mechanism of action. Biochimica et Biophysica Acta 2006; 1758(9), 1461-1474.
K Kerdkumthong, W Chanket, P Runsaeng, S Nanarong, K Songsurin, P Tantimetta, C Angsuthanasombat, A Aroonkesorn and S Obchoei. Two recombinant bacteriocins, rhamnosin and lysostaphin, show synergistic anticancer activity against gemcitabine-resistant cholangiocarcinoma cell lines. Probiotics and Antimicrobial Proteins 2024; 16(3), 713-725.
YP Shih, WM Kung, JC Chen, CH Yeh, AH Wang and TF Wang. High-throughput screening of soluble recombinant proteins. Protein Science 2002; 11(7), 1714-1719.
M Khaledi, HEG Ghaleh, MA Abyazi, HM Hosseini and R Golmohammadi. The anticancer properties of probiotic species. Journal of Applied Biotechnology Reports 2023; 10(4), 1123-1139.
T Utsugi, AJ Schroit, J Connor, CD Bucana and IJ Fidler. Elevated expression of phosphatidylserine in the outer membrane leaflet of human tumor cells and recognition by activated human blood monocytes. Cancer Research 1991; 51(11), 3062-3066.
F Schweizer. Cationic amphiphilic peptides with cancer-selective toxicity. European Journal of Pharmacology 2009; 625(1-3), 190-194.
S Riedl, B Rinner, M Asslaber, H Schaider, S Walzer, A Novak, K Lohner and D Zweytick. In search of a novel target - phosphatidylserine exposed by non-apoptotic tumor cells and metastases of malignancies with poor treatment efficacy. Biochimica et Biophysica Acta 2011; 1808(11), 2638-2645.
DW Hoskin and A Ramamoorthy. Studies on anticancer activities of antimicrobial peptides. Biochimica et Biophysica Acta 2008; 1778(2), 357-375.
AL Hilchie, CD Doucette, DM Pinto, A Patrzykat, S Douglas and DW Hoskin. Pleurocidin-family cationic antimicrobial peptides are cytolytic for breast carcinoma cells and prevent growth of tumor xenografts. Breast Cancer Research 2011; 13(5), R102.
C Cesa-Luna, JM Alatorre-Cruz, R Carreno-Lopez, V Quintero-Hernandez and A Baez. Emerging applications of bacteriocins as antimicrobials, anticancer drugs, and modulators of the gastrointestinal microbiota. Polish Journal of Microbiology 2021; 70(2), 143-159.
O Moroni, E Kheadr, Y Boutin, C Lacroix and I Fliss. Inactivation of adhesion and invasion of food-borne Listeria monocytogenes by bacteriocin-producing Bifidobacterium strains of human origin. Applied and Environmental Microbiology 2006; 72(11), 6894-6901.
YC Yue, BY Yang, J Lu, SW Zhang, L Liu, K Nassar, XX Xu, XY Pang and JP Lv. Metabolite secretions of Lactobacillus plantarum YYC-3 may inhibit colon cancer cell metastasis by suppressing the VEGF-MMP2/9 signaling pathway. Microbial Cell Factories 2020; 19(1), 213.
S Hume, GL Dianov and K Ramadan. A unified model for the G1/S cell cycle transition. Nucleic Acids Research 2020; 48(22), 12483-12501.
P Nisticò, MJ Bissell and DC Radisky. Epithelial-mesenchymal transition: General principles and pathological relevance with special emphasis on the role of matrix metalloproteinases. Cold Spring Harbor Perspectives in Biology 2012; 4(2), a011908.
EM Leslie, RG Deeley and SP Cole. Multidrug resistance proteins: Role of P-glycoprotein, MRP1, MRP2, and BCRP (ABCG2) in tissue defense. Toxicology and Applied Pharmacology 2005; 204(3), 216-237.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






