The Role of Monosodium Glutamate in Neural Crest Cell Migration Impacts Congenital Heart Defects and the Protective Effect of Folic Acid in Chick Embryo Models
DOI:
https://doi.org/10.48048/tis.2025.9890Keywords:
Congenital heart defects, Monosodium glutamate, Molecular docking, Folic acid, Homocysteine, Cardiac neural crest cell, Chick embryoAbstract
Introduction: Congenital heart defects are among the most common birth anomalies, characterized by malformations of cardiac structures such as the pharyngeal arches, aorticopulmonary septum, and outflow tract. Disruptions in neural crest cell function during embryogenesis have been also associated with congenital heart defects. This study investigates the teratogenic effects of monosodium glutamate on the critical signaling pathways of cardiac neural crest cells in cardiac morphogenesis, and folic acid mitigates this adverse effect in chick embryo models. Materials and methods: Using a combined network pharmacology and molecular biology approach, we identified molecular targets potentially associated with monosodium glutamate-induced congenital heart defects. Fertilized chick eggs were divided into control, monosodium glutamate exposure and folic acid treatment groups, and the effects on cardiac tissue structures were evaluated in 3 and 6-day-old embryos. Wingless-related integration site and bone morphogenetic protein signaling proteins were analyzed via immunostaining and immunoblotting techniques, while homocysteine levels were quantified using an ELISA assay. Results and discussion: These findings revealed that the molecular targets shared through network pharmacology and molecular docking analyses were closely linked to neural crest cell function. Monosodium glutamate exposure resulted in a significant reduction in neural crest cell populations and downregulation of Wingless-related integration site and bone morphogenetic protein signaling protein expression (p < 0.05). Additionally, homocysteine levels were controlled by monosodium glutamate exposure, suggesting a metabolic influence on cardiac development. Treatment with folic acid mitigates this adverse effect by lowering homocysteine levels. Conclusion: In conclusion, this study provides novel evidence that monosodium glutamate-induced teratogenesis disrupts the protein signaling molecules and metabolic pathways critical to embryonic heart development. This leads to congenital heart defects analogous to those observed in humans. These insights offer a foundation for understanding the impact of environmental teratogens on congenital heart anomalies and the role of neural crest cell-associated signaling pathways in cardiac morphogenesis. Moreover, the treatment with folic acid mitigates an adverse effect on cardiac morphogenesis by lowering homocysteine levels.
HIGHLIGHTS
- MSG exposure disrupts cardiac neural crest cell function by reducing populations and downregulating Wnt and BMP signaling pathways, contributing to congenital heart defects.
- Structural abnormalities, including thinner ventricular walls, impaired septation, and blood congestion, were observed in MSG exposure embryos, highlighting its teratogenic impact on heart development.
- Elevated homocysteine levels in MSG exposure embryos suggest a metabolic mechanism linking MSG exposure to the increased risk of CHDs, and treatment with folic acid mitigates this adverse effect by lowering homocysteine levels.
GRAPHICAL ABSTRACT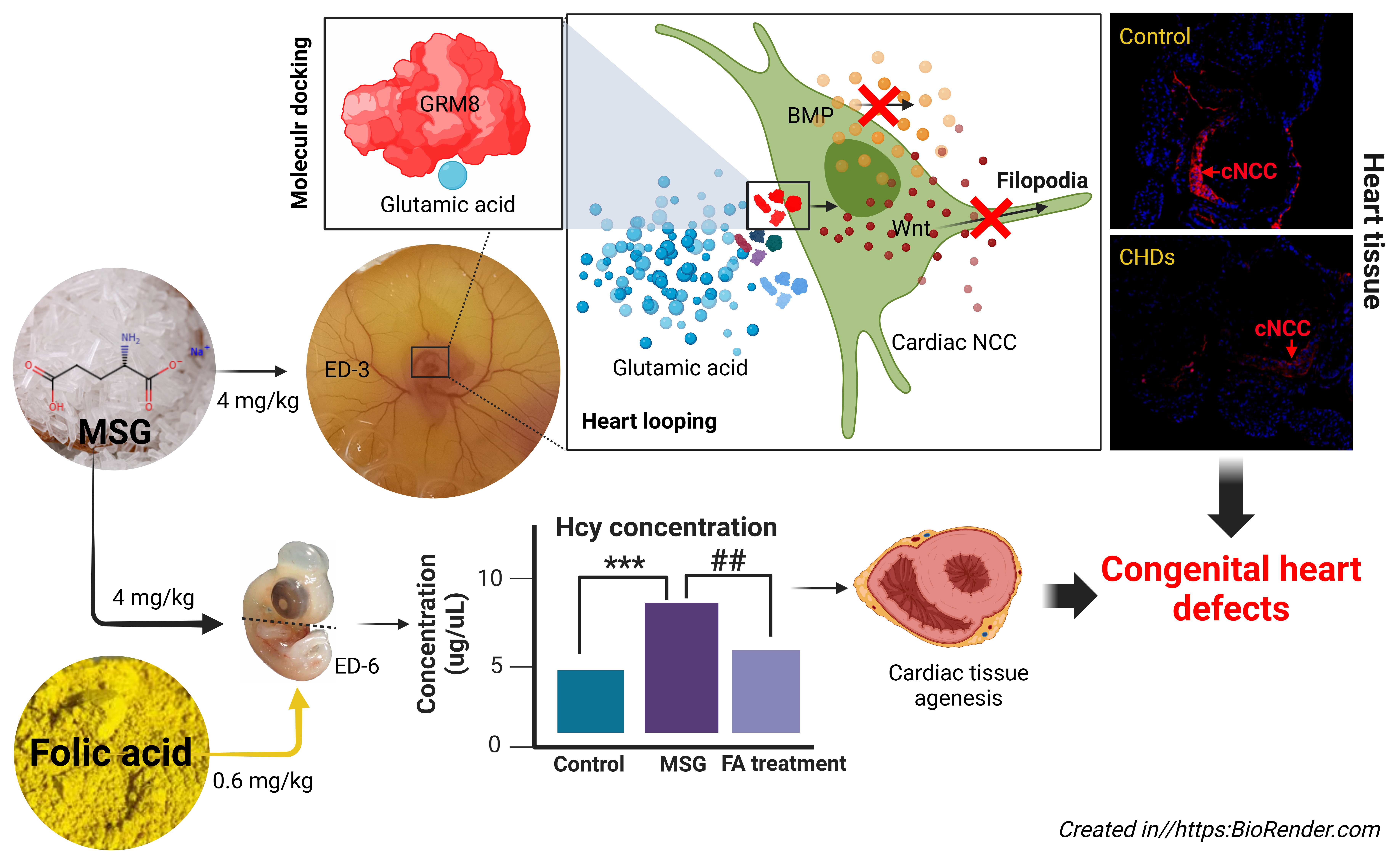
Downloads
References
Y Liu, S Chen, L Zühlke, GC Black, MK Choy, N Li and BD Keavney. Global birth prevalence of congenital heart defects 1970 - 2017: Updated systematic review and meta-analysis of 260 studies. International Journal of Epidemiology 2019; 48(2), 455-463.
R Kučienė and V Dulskienė. Selected environmental risk factors and congenital heart defects. Medicina 2008; 44(11), 827.
S Baldacci, F Gorini, M Santoro, A Pierini, F Minichilli and F Bianchi. Environmental and individual exposure and the risk of congenital anomalies: A review of recent epidemiological evidence. Epidemiologia e Prevenzione 2018; 42(3-4), 1-34.
LX Liu and Z Arany. Maternal cardiac metabolism in pregnancy. Cardiovascular Research 2014; 101(4), 545-553.
F Lamberto, I Peral-Sanchez, S Muenthaisong, M Zana, S Willaime-Morawek and A Dinnyés. Environmental alterations during embryonic development: Studying the impact of stressors on pluripotent stem cell-derived cardiomyocytes. Genes 2021; 12(10), 1564.
TP Fleming, C Sun, O Denisenko, L Caetano, A Aljahdali, JM Gould and P Khurana. Environmental exposures around conception: Developmental pathways leading to lifetime disease risk. International Journal of Environmental Research and Public Health 2021; 18(17), 9380.
SP Chakraborty. Patho-physiological and toxicological aspects of monosodium glutamate. Toxicology Mechanisms and Methods 2019; 29(6), 389-396.
S Erhardt, M Zheng, X Zhao, TP Le, TO Findley and J Wang. The cardiac neural crest cells in heart development and congenital heart defects. Journal of Cardiovascular Development and Disease 2021; 8(8), 89.
HSW Haroun. Teratogenicity and teratogenic factors. MOJ Anatomy and Physiology 2017; 3(1), 1-5.
AS Mahaliyana, MFA Fasmina, AMTB Alahakoon and GMGMM Wickrama. Toxicity effects of monosodium glutamate (MSG) on embryonic development of zebrafish (Danio rerio); A promising model to study excitotoxins. International Journal of Scientific and Research Publications 2016; 6(3), 229-234.
NAA Elnaga, M Sarhan and H Mansour. Teratogenicity of monosodium glutamate on pregnant rats and their fetuses. The Egyptian Journal of Hospital Medicine 2019; 74(8), 1737-1747.
S Cerrizuela, GA Vega-Lopez and MJ Aybar. The role of teratogens in neural crest development. Birth Defects Research 2020; 112(8), 584-632.
E Takimoto and DA Kass. Role of oxidative stress in cardiac hypertrophy and remodeling. Hypertension 2007; 49(2), 241-248.
S Pintarasri, M Santiparadon, A Kamnate, C Naktubtim, T Koomsang and W Payuhakrit. A novel’s evidence of Msg-induced craniofacial defects in chick embryo models. Trends in Science 2024; 21(12), 8718.
TL Creazzo, RE Godt, L Leatherbury, SJ Conway and ML Kirby. Role of cardiac neural crest cells in cardiovascular development. Annual Review of Physiology1998; 60, 267-286.
AL Hopkins. The role of network pharmacology in understanding multifactorial diseases. Nature Reviews Drug Discovery 2022; 21(1), 39-56.
X Chen, CC Yan, X Zhang, X Zhang, F Dai, J Yin and Y Zhang. Drug-target interaction prediction: Databases, web servers and computational models. Briefings in Bioinformatics 2016; 17(4), 696-712.
J Lin and H Nagase. The road not taken with pyrrole-imidazole polyamides: Off-target effects and genomic binding. Biomolecules 2020; 10(4), 544.
A Negri, V Naponelli, F Rizzi and S Bettuzzi. Molecular targets of epigallocatechin - Gallate (EGCG): A special focus on signal transduction and cancer. Nutrients 2018; 10(12), 1936.
W Tang, ML Martik, Y Li and ME Bronner. Cardiac neural crest contributes to cardiomyocytes in amniotes and heart regeneration in zebrafish. Elife 2019; 8, e47929.
O Schussler, L Gharibeh, P Mootoosamy, N Murith, V Tien, AL Rougemont, T Sologashvili, E Suuronen, Y Lecarpentier and M Ruel. Cardiac neural crest cells: Their rhombomeric specification, migration, and association with heart and great vessel anomalies. Cellular and Molecular Neurobiology 2021; 41(2), 403-429.
ME Bronner and NM LeDouarin. Development and evolution of the neural crest: An overview. Developmental Biology 2012; 366(1), 2-9.
R Pal and A Khanna. Role of smad-and wnt-dependent pathways in embryonic cardiac development. Stem cells and Development 2006; 15(1), 29-39.
A Mohan, R Kumar, V Kumar and M Yadav. Homocysteine, vitamin B12 and folate level: Possible risk factors in the progression of chronic heart and kidney disorders. Current Cardiology Reviews 2023; 19(4), 66-83.
S Pintarasri, V Plakornkul, Y Viravud, W Payuhakrit and T Rungruang. Folic acid attenuates MSG-induced teratogenicity during a 2-month pregnancy by preventing neural crest cell destruction and malformation in chick embryo models. Trends in Sciences 2023; 20(7), 6656.
SM Hazzaa, ES El-Roghy, MA Abd Eldaim and GE Elgarawany. Monosodium glutamate induces cardiac toxicity via oxidative stress, fibrosis, and P53 proapoptotic protein expression in rats. Environmental Science and Pollution Research 2020; 27(17), 20014-20024.
RN Wang, J Green, Z Wang, Y Deng, M Qiao, M Peabody, Q Zhang, J Ye, Z Yan, Z Yan, S Denduluri, O Idowu, M Li, C Shen, A Hu, RC Haydon, R Kang, J Mok, MJ Lee, HL Luu and LL Shi. Bone morphogenetic protein (BMP) signaling in development and human diseases. Genes & Diseases 2014; 1(1), 87-105.
L Hebert, P Hillman, C Baker, M Brown, A Ashley-Koch, JE Hixson, AC Morrison, H Northrup and KS Au. Burden of rare deleterious variants in WNT signaling genes among 511 myelomeningocele patients. PLoS One 2020; 15(9), e0239083.
XJ Zhu, YD Liu, ZM Dai, X Zhang, XQ Yang, Y Li, M Qiu, J Fu, W Hsu, YP Chen and Z Zhang. BMP-FGF signaling axis mediates Wnt-induced epidermal stratification in developing mammalian skin. PLoS Genetics 2014; 10(10), e1004687.
N Itasaki and S Hoppler. Crosstalk between Wnt and bone morphogenic protein signaling: A turbulent relationship. Developmental Dynamics: An Official Publication of the American Association of Anatomists 2010; 239(1), 16-33.
G Chen, C Deng and YP Li. TGF-β and BMP signaling in osteoblast differentiation and bone formation. International Journal of Biological Sciences 2012; 8(2), 272-288.
IA Mohamed, N El-Badri and A Zaher. Wnt signaling: The double-edged sword diminishing the potential of stem cell therapy in congenital heart disease. Life Sciences 2019; 239, 116937.
Y Li, J Du, S Deng, B Liu, X Jing, Y Yan, Y Liu, J Wang, X Zhou and Q She. The molecular mechanisms of cardiac development and related diseases. Signal Transduction and Targeted Therapy 2024; 9(1), 368.
S Silvestro, V Calcaterra, G Pelizzo, P Bramanti and E Mazzon. Prenatal hypoxia and placental oxidative stress: insights from animal models to clinical evidences. Antioxidants 2020; 9(5), 414.
N Mollo, R Scognamiglio, A Conti, S Paladino, L Nitsch and A Izzo. Genetics and molecular basis of congenital heart defects in Down syndrome: Role of extracellular matrix regulation. International Journal of Molecular Sciences 2023; 24(3), 2918.
S Sambu, U Hemaram, R Murugan and AA Alsofi. Toxicological and teratogenic effect of various food additives: An updated review. BioMed Research International 2022; 2022, 6829409.
JI Hoffman and S Kaplan. The incidence of congenital heart disease. Journal of the American College of Cardiology 2002; 39(12), 1890-1900.
A Zanfirescu, A Ungurianu, AM Tsatsakis, GM Nițulescu, D Kouretas, A Veskoukis, D Tsoukalas, AB Engin, M Aschner and D Margină. A review of the alleged health hazards of monosodium glutamate. Comprehensive Reviews in Food Science and Food Safety 2019; 18(4), 1111-1134.
GJ Udom, BR Abdulyekeen, MO Osakwe, AN Ezejiofor, CN Orish, FC Orish, C Frazzoli and OE Orisakwe. Reconsideration of the health effects of monosodium glutamate: From bench to bedside evidence. Journal of Environmental Science and Health, Part C 2024; 43, 1-31.
A Mahajan, D Sapehia, S Thakur, PS Mohanraj, R Bagga and J Kaur. Effect of imbalance in folate and vitamin B12 in maternal/parental diet on global methylation and regulatory miRNAs. Scientific Reports 2019; 9(1), 17602.
B Li, Y Xu, D Pang, Q Zhao, L Zhang, M Li, W Li, G Duan and C Zhu. Interrelation between homocysteine metabolism and the development of autism spectrum disorder in children. Frontiers in Molecular Neuroscience 2022; 15, 947513.
PR Brauer and BJ Tierney. Consequences of elevated homocysteine during embryonic development and possible modes of action. Current Pharmaceutical Design 2004; 10(22), 2719-2732.
V Iacobazzi, V Infantino, A Castegna and G Andria. Hyperhomocysteinemia: Related genetic diseases and congenital defects, abnormal DNA methylation and newborn screening issues. Molecular Genetics and Metabolism 2014; 113(1-2), 27-33.
RM Shawky, AR Ramy, SMN El-Din, SM Abd Elmonem and MA Abd Elmonem. Abnormal maternal biomarkers of homocysteine and methionine metabolism and the risk of congenital heart defects. Egyptian Journal of Medical Human Genetics 2018; 19(1), 7-12.
L Koklesova, A Mazurakova, M Samec, K Biringer, SM Samuel, D Büsselberg, P Kubatka and O Golubnitschaja. Homocysteine metabolism as the target for predictive medical approach, disease prevention, prognosis, and treatments tailored to the person. EPMA Journal 2021; 12, 477-505.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






