Modulation of Gut Microbiota in Rats by Hydrolyzed-Glucomannan from Amorphophallus oncophyllus
DOI:
https://doi.org/10.48048/tis.2025.9551Keywords:
Porang glucomannan hydrolysate, Ribosomal Intergenic Spacer Analysis (RISA), 16S rRNA sequencing, Gut microbiota modulation, Prebiotic, Short-chain fatty acidAbstract
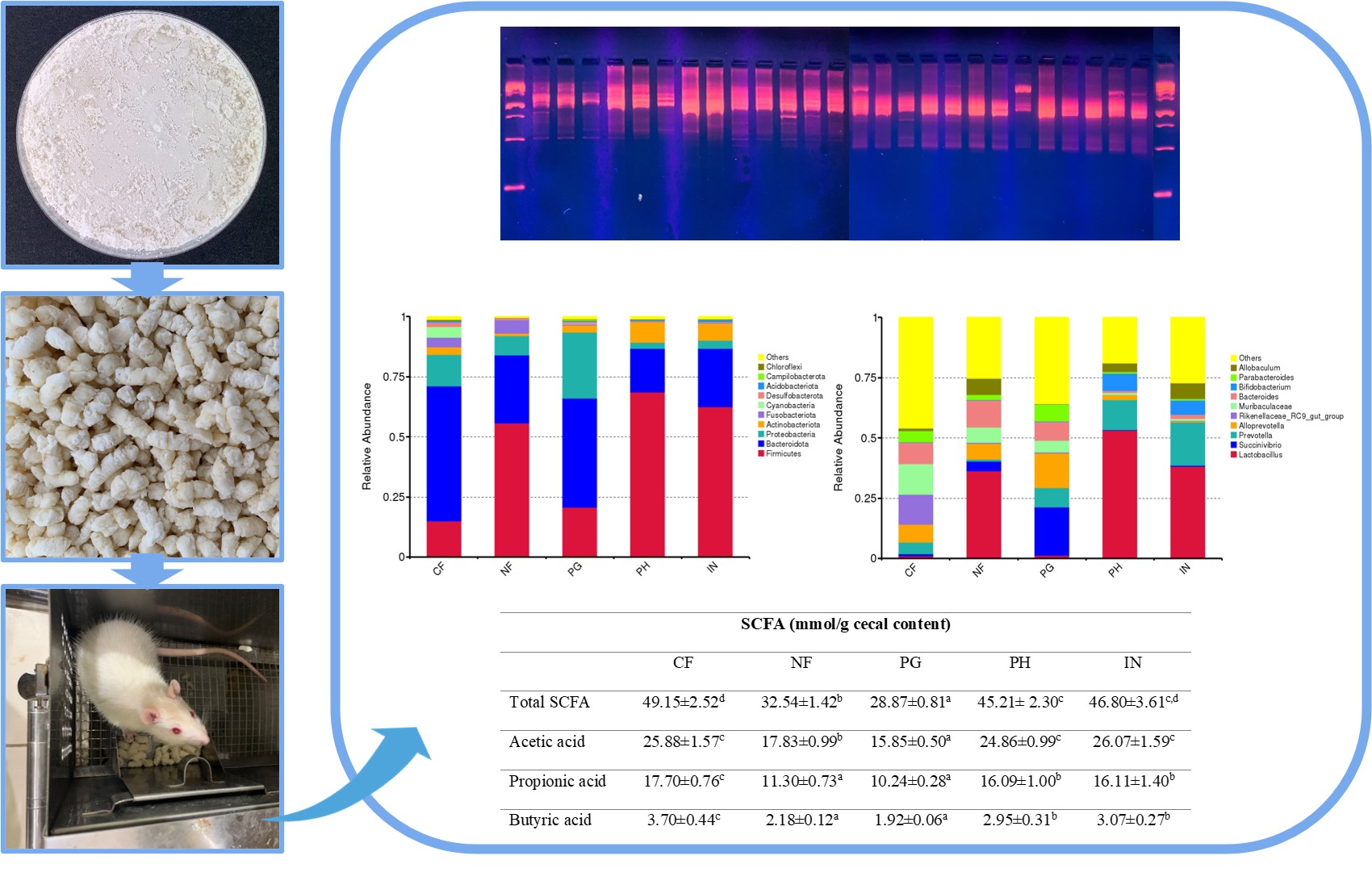
Gut microbiota plays a crucial role in maintaining host health, with dietary fibers being key modulators. This study evaluates the effects of porang glucomannan hydrolysate (PH), a novel prebiotic, on gut microbiota and short-chain fatty acid (SCFA) production in rats. For 21 days, rats were fed diets containing cellulose (CF), a fiber-free diet (NF), porang glucomannan (PG), PH, or inulin (IN). Gut microbiota composition was assessed using Ribosomal Intergenic Spacer Analysis (RISA) and 16S rRNA sequencing, while SCFA levels were measured via gas chromatography. The findings revealed differences in gut microbiota composition at the phylum level: the CF, NF, and PG groups were predominantly composed of Bacteroidota, Firmicutes, and Proteobacteria, whereas the PH and IN groups were mainly dominated by Firmicutes, Bacteroidota, and Actinobacteriota. Notably, at the genus level, SCFA-producing bacterial groups, such as Lactobacillus, Allobaculum, Bifidobacterium, and Blautia, were identified only in the PH and IN groups. This was further corroborated by the higher SCFA concentrations found in the PH and IN groups compared to the other treatment groups. These findings suggest that the inclusion of PH in rats’ diets positively affects the modulation of gut microbiota and increases SCFA concentrations in the cecum, showing effects similar to those of inulin, a commercial prebiotic. Consequently, PH holds the potential as a functional food that supports gastrointestinal health.
HIGHLIGHTS
- The hydrolysis process of porang glucomannan (PG) significantly altered the composition of gut microbiota in rats. Ribosomal Intergenic Spacer Analysis (RISA) and beta diversity measurements indicated that the group receiving PG formed a distinct cluster compared to the group given porang glucomannan hydrolysate (PH).
- The gut microbiota in the PH group closely resembled that of rats fed with inulin (IN).
- The PH group had a greater abundance of short-chain fatty acid (SCFA)-producing bacteria, including Lactobacillus, Allobaculum, Bifidobacterium, and Blautia, which were absent in the PG group. Consequently, SCFA levels were significantly higher in the PH group than in the PG group.
GRAPHICAL ABSTRACT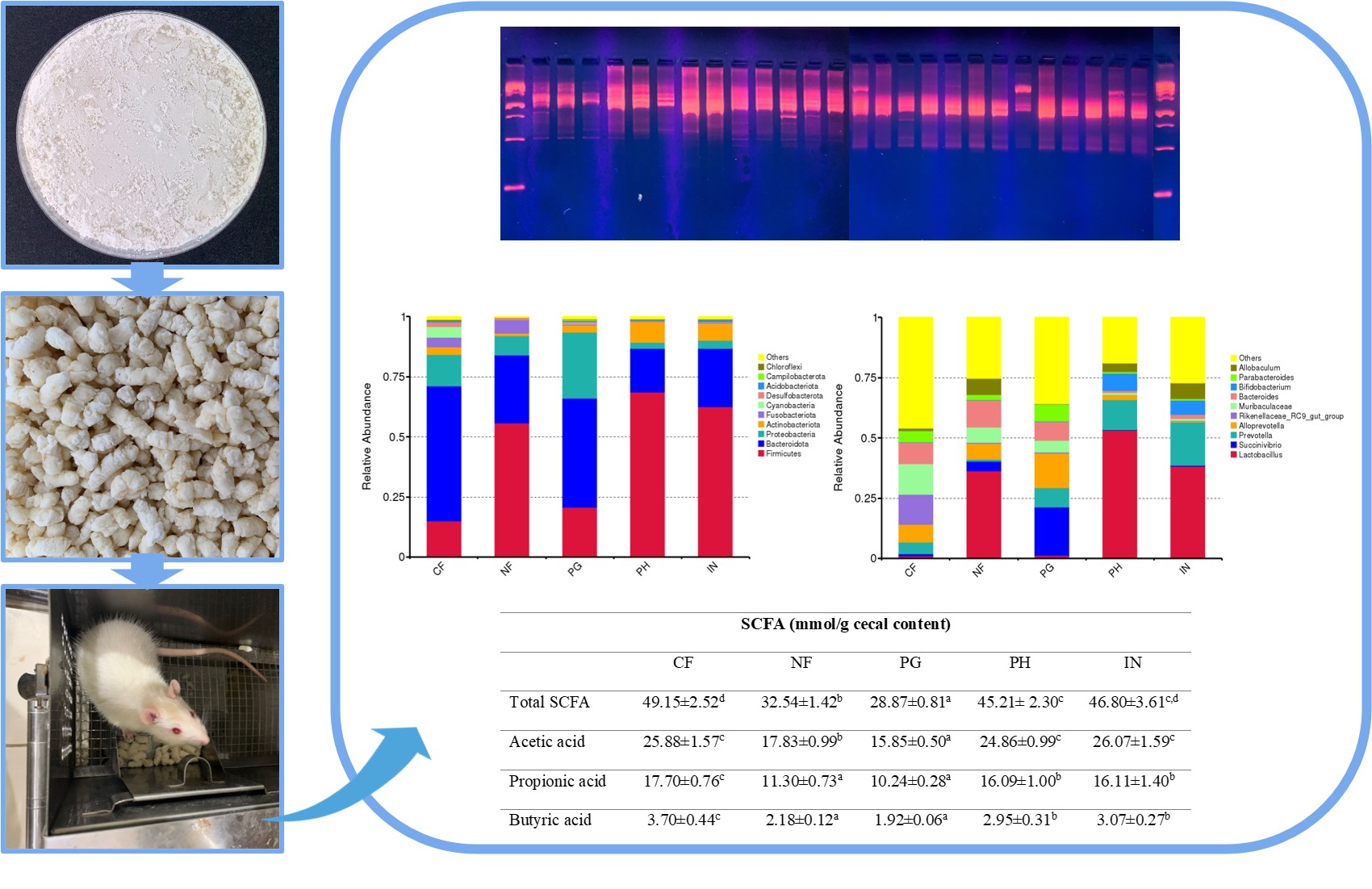
Downloads
References
K Maheshwari, P Musyuni, A Moulick, H Mishra, A Ekielski, PK Mishra and G Aggarwal. Unveiling the microbial symphony: Next-Gen sequencing and bioinformatics insights into the human gut microbiome. Health Sciences Review 2024; 11(S1), 100173.
C Zhang, X Pi, X Li, J Huo and W Wang. Edible herbal source-derived polysaccharides as potential prebiotics: Composition, structure, gut microbiota regulation, and its related health effects. Food Chemistry 2024; 458, 140267.
M Čoklo, DR Maslov and SK Pavelić. Modulation of gut microbiota in healthy rats after exposure to nutritional supplements. Gut Microbes 2020; 12(1), 1779002.
P Cronin, SA Joyce, PW O’Toole and EM O’Connor. Dietary fibre modulates the gut microbiota. Nutrients 2021; 13(5), 1655.
NB Danneskiold-Samsøea, HDFQ Barros, R Santos, JL Bicas, CBB Cazarin, L Madsen, K Kristiansen, GM Pastore, S Brix and MRM Júnior. Interplay between food and gut microbiota in health and disease. Food Research International 2019; 115, 23-31.
H Li, G Wang, X Yan, X Hu and J Li. Effects of acetyl groups on the prebiotic properties of glucomannan extracted from Artemisia sphaerocephala Krasch seeds. Carbohydrate Polymers 2024; 330, 121805.
D Ouyang, J Deng, K Zhou, Y Liang, Y Chen, D Wang, J Zhong, Y Sun and M Li. The effect of deacetylation degree of konjac glucomannan on microbial metabolites and gut microbiota in vitro fermentation. Journal of Functional Foods 2020; 66, 103796.
P Stribling and F Ibrahim. Dietary fibre definition revisited - The case of low molecular weight carbohydrates. Clinical Nutrition ESPEN 2023; 55, 340-356.
JY Yin, LY Ma, MY Xie, SP Nie and JY Wu. Molecular properties and gut health benefits of enzyme-hydrolyzed konjac glucomannans. Carbohydrate Polymers 2020; 237, 116117.
R Satiti, T Utami, J Widada and E Harmayani. Characterization and prebiotic activity in vitro of hydrolyzed glucomannan extracted from fresh porang tuber (Amorphophallus oncophyllus). Trends in Sciences 2025; 22(1), 8617.
A Anggela, E Harmayani, W Setyaningsih and S Wichienchot. Prebiotic effect of porang oligo-glucomannan using fecal batch culture fermentation. Food Science and Technology 2021; 42(1), 1-7.
B Gómez, B Míguez, R Yáñez and JL Alonso. Manufacture and properties of glucomannans and glucomannooligosaccharides derived from konjac and other sources. Journal of Agricultural and Food Chemistry 2017; 65(10), 2019-2031.
G Paturi, S Mishra, DI Hedderley and JA Monro. Gut microbiota responses to dietary fibre sources in rats fed starch-based or quasi-human background diets. Journal of Functional Foods 2021; 83, 104565.
PG Reeves, FH Nielsen and GC Fahey. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition and Ad Hoc Writing Committee on the reformulation of the AIN-76A diet. Journal of Nutrition 1993; 123(11), 1939-1951.
ES Rahayu, IH Rusdan, A Athennia, RZ Kamil, PC Pramesi, Y Marsono, T Utami and J Widada. Safety assessment of indigenous probiotic strain Lactobacillus plantarum Dad-13 isolated from Dadih using Sprague Dawley rats as a model. American Journal of Pharmacology and Toxicology 2019; 14(1), 38-47.
Z Yu and WW Mohn. Bacterial diversity and community structure in an aerated lagoon revealed by ribosomal intergenic spacer analyses and 16S ribosomal DNA sequencing. Applied and Environmental Microbiology 2001; 67(4), 1565-1574.
G Baskara, N Gofar, U Harun, J Widada, PA Hardini, R Wandri and D Asmono. Profiling of rhizobacteria to alleviate drought stress in oil palm using ribosomal intergenic spacer analysis. IOP Conference Series: Earth and Environmental Science 2024; 1308(1), 012035.
T Magoč and LS Steven. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011; 27(21), 2957-2963.
NA Bokulich, S Subramanian, JJ Faith, D Gevers, JI Gordon, R Knight, DA Mills and JG Caporaso. Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nature Methods 2013; 10(1), 57-59.
RC Edgar, BJ Haas, JC Clemente, C Quince and R Knight. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011; 27(16), 2194-220.
BJ Haas, D Gevers, AM Earl, M Feldgarden, DV Ward, G Giannoukos, D Ciulla, D Tabbaa, SK Highlander, E Sodergren, B Methé, TZ DeSantis, Human Microbiome Consortium, JF Petrosino, R Knight and BW Birren. Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Research 2011; 21(3), 494-504.
RC Edgar. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nature methods 2013; 10(10), 996-998.
SF Altschul, W Gish, W Miller, EW Myers and DJ Lipman. Basic local alignment search tool. Journal of Molecular Biology 1990; 215(3), 403-410.
Q Wang, GM Garrity, JM Tiedje and JR Cole. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied and environmental microbiology 2007; 73(16), 5261-5267.
C Quast, E Pruesse, P Yilmaz, J Gerken, T Schweer, P Yarza, J Peplies and FO Glöckner. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Research 2013; 41(D1), D590-D596.
RC Edgar. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research 2004; 32(5), 1792-1797.
E Harmayani, V Aprilia and Y Marsono. Characterization of glucomannan from Amorphophallus oncophyllus and its prebiotic activity in vivo. Carbohydrate Polymers 2014; 112(3), 475-479.
D Desnilasari, E Harmayani, J Widada and Nurliyani. Using kefir glucomannan to modify the diversity and composition of cecum bacterial in Sprague Dawley rats with metabolic syndrome. IOP Conference Series: Materials Science and Engineering, 2021; 1011(1), 012009.
S Yeh, M Lin and H Chen. Partial hydrolysis enhances the inhibitory effects of konjac glucomannan from Amorphophallus konjac C. Koch on DNA damage induced by fecal water in Caco-2 cells. Food Chemistry 2010; 119(2), 614-618.
M Wang, Y Chen, Y Wang, Y Li, H Zheng, F Ma, CW Ma, X Zhang, B Lu, Z Xie and Q Liao. The effect of probiotics and polysaccharides on the gut microbiota composition and function of weaned rats. Food and Function 2018; 9(3), 1864-1877.
Y Zhang, Y Zhao, W Yang, G Song, P Zhong, Y Ren and G Zhong. Structural complexity of konjac glucomannan and its derivatives governs the diversity and outputs of gut microbiota. Carbohydrate Polymers 2022; 292, 119639.
Y Li, T Gong, H Lu, S Ma and X Liu. In vitro fermentation characteristics of oxidized konjac glucomannan and its modulation effects on gut microbiota. Food Hydrocolloids 2023; 141(2), 108693.
TM Cantu-Jungles and BR Hamakera. New view on dietary fiber selection for predictable shifts in gut microbiota. MBio 2020; 11(1), e02179-02119.
Y Li, Y Kang, Y Du, M Chen, L Guo, X Huang, T Li, S Chen, F Yang, F Yu, J Hong and X Kong. Effects of konjaku flour on the gut microbiota of obese patients. Frontiers in Cellular and Infection Microbiology 2022; 12, 771748.
H Wang, X Zhang, S Wang, H Li, Z Lu, J Shi and Z Xu. Mannan-oligosaccharide modulates the obesity and gut microbiota in high-fat diet-fed mice. Food and Function 2018; 9(7), 3916-3929.
RZ Kamil, A Murdiati, M Juffrie, J Nakayama and ES Rahayu. Gut microbiota and short-chain fatty acid profile between normal and moderate malnutrition children in Yogyakarta, Indonesia. Microorganisms 2021; 9(1), 1-15.
JY Ou, JQ Huang, Y Song, SW Yao, XC Peng, MF Wang and SY Ou. Feruloylated oligosaccharides from maize bran modulated the gut microbiota in rats. Plant Foods for Human Nutrition 2016; 71(2), 123-128.
YF Li, CR Zhu, XL Gong, HL Li, LK Xiong, KJ Wang and GS Liu. Beneficial effects of probiotic treatment on gut microbiota in very low birth weight infants. Gastroenterology Research and Practice 2019; 2019, 3682836.
X Zhang, JJ Aweya, ZX Huang, ZY Kang, ZH Bai, KH Li, XT He, Y Liu, XQ Chen and KL Cheong. In vitro fermentation of Gracilaria lemaneiformis sulfated polysaccharides and its agaro-oligosaccharides by human fecal inocula and its impact on microbiota. Carbohydrate Polymers 2020; 234, 115894.
Y Wang, W Zhang, J Wang, C He and X Zhu. Effect of selenium-rich konjac glucomannan on intestinal microbial diversity in mice. Journal of Biological Regulators and Homeostatic Agents 2023; 37(6), 2877-2887.
Y Li, H Lu, C Liao and X Liu. Oxidized konjac glucomannan: A safe dietary fiber influencing mouse gut microbiota. Food Chemistry: X 2024; 21, 101089.
W Fusco, MB Lorenzo, M Cintoni, S Porcari, E Rinninella, F Kaitsas, E Lener, MC Mele, A Gasbarrini, MC Collado, G Cammarota and G Ianiro. Short-chain fatty-acid-producing bacteria: key components of the human gut microbiota. Nutrients 2023; 15(9), 2211.
T Chen, W Long, C Zhang, S Liu, L Zhao and BR Hamaker. Fiber-utilizing capacity varies in Prevotella- versus Bacteroides-dominated gut microbiota. Scientific Reports 2017; 7(1), 2594.
Z Zheng, W Lyu, Y Ren, X Li, S Zhao, H Yang and Y Xiao. Allobaculum involves in the modulation of intestinal ANGPTLT4 expression in mice treated by high-fat diet. Frontiers in Nutrition 2021; 8, 690138.
RZ Kamil, A Murdiati, M Juffrie and ES Rahayu. Gut microbiota modulation of moderate undernutrition in infants through gummy Lactobacillus plantarum Dad-13 consumption: A randomized double-blind controlled trial. Nutrients 2022; 14(5), 1049.
YN Larasati, E Harmayani, J Widada, N Nurliyani and A Perdinan. Caecal bacterial composition of broiler chickens affected by porang glucomannan. Journal of the Indonesian Tropical Animal Agriculture 2021; 46(3), 187-198.
J Deng, X Zou, Y Liang, J Zhong, K Zhou, J Zhang, M Zhang, Z Wang, Y Sun and M Li. Hypoglycemic effects of different molecular weight konjac glucomannans via intestinal microbiota and SCFAs mediated mechanism. International Journal of Biological Macromolecules 2023; 234, 122941.
P Portincasa, L Bonfrate, M Vacca, MD Angelis, I Farella, E Lanza, M Khalil, DQH Wang, M Sperandio and AD Ciaula. Gut microbiota and short chain fatty acids: Implications in glucose homeostasis. International Journal of Molecular Sciences 2022; 23(3), 1105.
Y Li, HS Faden and L Zhu. The response of the gut microbiota to dietary changes in the first two years of life. Frontiers in Pharmacology 2020; 11, 334.
O Hernandez, ML Sanz, S Kolida, RA Rastall and FJ Moreno. In vitro fermentation by human gut bacteria of proteolytically digested caseinomacropeptide nonenzymatically glycosylated with prebiotic carbohydrates. Journal of Agricultural and Food Chemistry 2011; 59(22), 11949-11955.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






