Characterization and Prebiotic Activity in Vitro of Hydrolyzed Glucomannan Extracted from Fresh Porang Tuber (Amorphophallus Oncophyllus)
DOI:
https://doi.org/10.48048/tis.2024.8617Keywords:
Porang glucomannan (PG), Porang glucomannan hydrolysate (PGH), Enzymatic hydrolysis, Characteristics, Prebiotic activity scoreAbstract
The technology for direct extraction of porang glucomannan (PG) from fresh tubers offers a faster and simpler process, yielding high-purity glucomannan. However, PG’s high viscosity and low solubility limit its use in the food, pharmaceutical, and health industries. Enzymatic hydrolysis of PG has the potential to improve these characteristics and enhance its prebiotic activity. This study aimed to examine the characteristics and prebiotic activity of porang glucomannan hydrolysate (PGH) derived from glucomannan extracted directly from the fresh tubers. PG was hydrolyzed under optimal conditions at 37.6 °C for 3 h, pH of 6.8, and an E/S of 0.8 % (w/w). Analysis of PGH included the degree of polymerization (DP), molecular weight (MW), viscosity, and solubility. Changes in the morphology, molecular structure, and composition of PGH were assessed through scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), and high-performance liquid chromatography (HPLC), respectively. Prebiotic activity analysis was conducted by determining the prebiotic activity score in vitro. The results revealed that PGH was composed of 58 % mannohexaose, 40 % mannotriose, and 2 % mannobiose. Notably, PGH exhibited reduced granule size and a porous surface. FTIR analysis indicated a structural change in the PGH molecule after hydrolysis. The DP of PG decreased by 1000 times, and the MW decreased by 60 times, leading to a 700-fold reduction in the viscosity of PGH. In contrast, the solubility of PGH increased threefold, attributed to the disruption of glycosidic and hydrogen bonds between and within glucomannan molecules during hydrolysis. These modifications in PGH characteristics make it more readily fermentable by several Lactobacilli and Bifidobacteria without stimulating the growth of E. coli, resulting in a positive prebiotic activity score. As a result, PGH shows promise as a source of prebiotics and can be utilized in beverage products to enrich their nutritional value, consequently producing functional food.
HIGHLIGHTS
- The hydrolysis of porang glucomannan (PG) altered its molecular structure and resulted in the formation of porang glucomannan hydrolysate (PGH) containing 58 % mannohexaose, 40 % mannotriose, and 2 % mannobiose.
- The degree of polymerization and molecular weight of PG decreased significantly by 1000 times and 60 times, respectively, leading to reduced viscosity and improved solubility of PGH.
- The changes in PGH characteristics due to hydrolysis made it more readily fermentable by various strains of Lactobacilli and Bifidobacteria, resulting in a positive prebiotic activity score.
GRAPHICAL ABSTRACT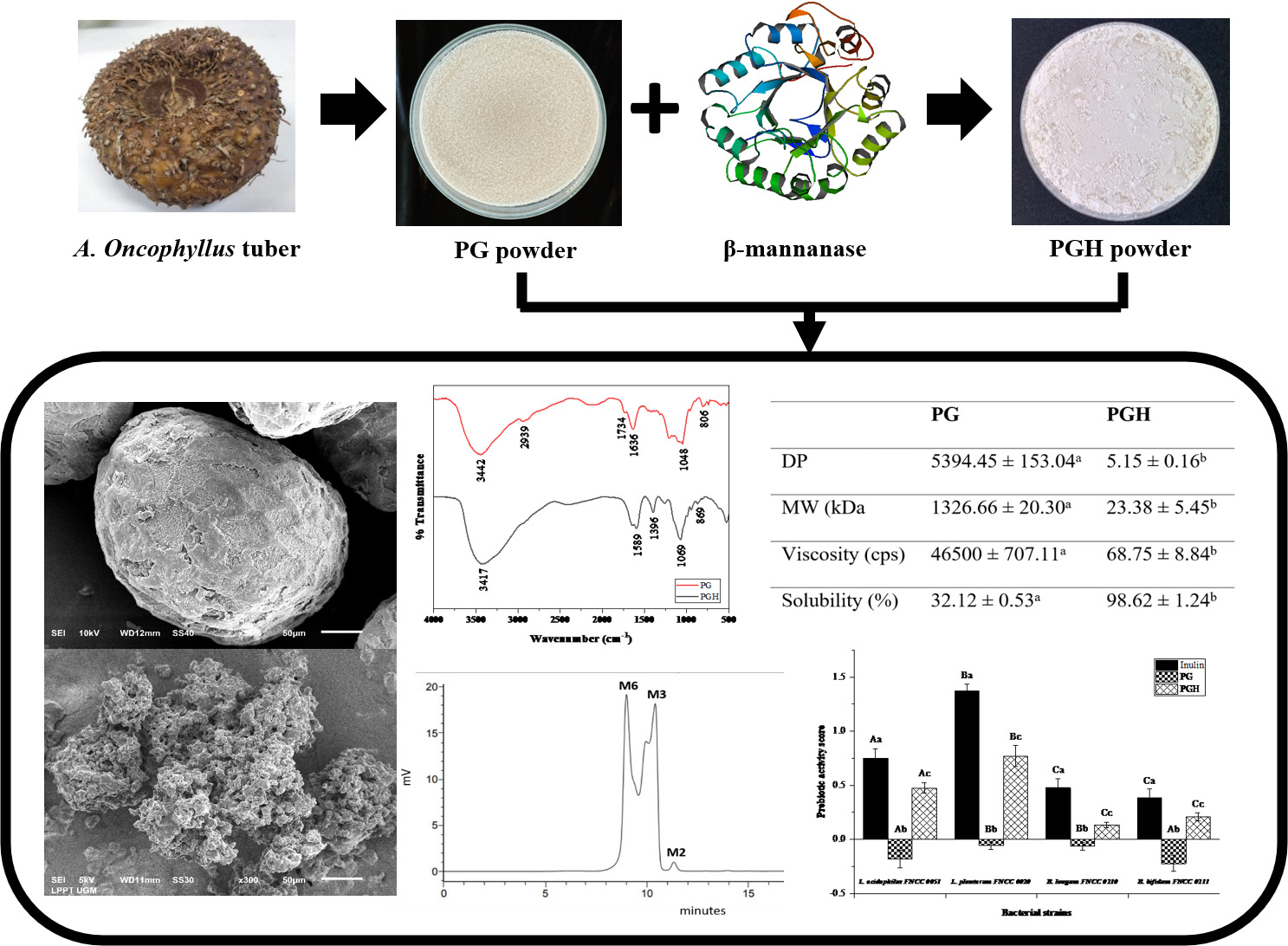
Downloads
References
RF Tester and FH Al-Ghazzewi. Glucomannan and nutrition. Mannans and health, with a special focus on glucomannans. Food Research International 2013; 50(1), 394-391.
R Tester and F Al-Ghazzewi. Glucomannan and nutrition. Food Hydrocolloids 2017; 68, 246-254.
E Harmayani, V Aprilia and Y Marsono. Characterization of glucomannan from Amorphophallus oncophyllus and its prebiotic activity in vivo. Carbohydrate Polymers 2014; 112, 475-479.
A Lopez-Rubio, P Tarancon, LG Gomez-Mascaraque, M Martinez-Sanz, MJ Fabra, JC Martinez and S Fiszman. Development of glucomannan-chitosan interpenetrating hydrocolloid networks (IHNs) as a potential tool for creating satiating ingredients. Food Hydrocolloids 2016; 60, 533-542.
Y Zhang, C Tong, Y Chen, X Xia, S Jiang, C Qiu and J Pang. Advances in the construction and application of konjac glucomannan-based delivery systems. International Journal of Biological Macromolecules 2024; 262(1), 129940.
AA Anggraeni, P Triwitono, LA Lestari and E Harmayani. Evaluation of glucomannan as a fat replacer in the dough and cookies made from fermented cassava flour and soy protein concentrate. Food Chemistry 2024; 434, 137452.
IWR Widarta, A Rukmini, U Santoso, Supriyadi and S Raharjo. Optimization of oil-in-water emulsion capacity and stability of octenyl succinic anhydride-modified porang glucomannan (Amorphophallus muelleri Blume). Heliyon 2022; 8(5), e09523.
B Azhar, S Gunawan, ERF Setyadi, L Majidah, F Taufany, L Atmaja and HW Aparamarta. Purification and separation of glucomannan from porang tuber flour (Amorphophallus muelleri) using microwave assisted extraction as an innovative gelatine substituent. Heliyon 2022; 9(11), e21972.
L Guo, HD Goff, M Chen and F Zhong. The hydration rate of konjac glucomannan after consumption affects its in vivo glycemic response and appetite sensation and in vitro digestion characteristics. Food Hydrocolloids 2022; 122, 107102.
S Gurusmatika, K Nishi, E Harmayani, Y Pranoto and T Sugahara. Immunomodulatory activity of octenyl succinic anhydride modified porang (Amorphophallus oncophyllus) glucomannan on mouse macrophage-like J774.1 cells and mouse primary peritoneal macrophages. Molecules 2017; 22(7), 1187.
J Liu, Q Xu, J Zhang, X Zhou, F Lyu, P Zhao and Y Ding. Preparation, composition analysis and antioxidant activities of konjac oligo-glucomannan. Carbohydrate Polymers 2015; 130, 398-404.
M Chua, K Chan, TJ Hocking, PA Williams, CJ Perry and TC Baldwin. Methodologies for the extraction and analysis of konjac glucomannan from corms of Amorphophallus konjac K. Koch. Carbohydrate Polymers 2012; 87(3), 2202-2210.
AC Kumoro, THA Yuganta, R Ratnawati and DS Retnowati. Effect of catalyst concentration and reaction time on the extraction of glucomannan from porang (Amorphophallus oncophyllus) flour via acid hydrolysis. IOP Conference Series: Materials Science and Engineering 2016; 162, 012020.
A Yanuriati, DW Marseno, Rochmadi and E Harmayani. Characteristics of glucomannan isolated from fresh tuber of porang (Amorphophallus muelleri Blume). Carbohydrate Polymers 2017; 156, 56-63.
DH Wardhani, JA Vazquez, DA Ramdani, A Lutfiati, N Aryanti and H Cahyono. Enzymatic purification of glucomannan from Amorphophallus oncophyllus using α-amylase. Bioscience Journal 2019; 35(1), 277-288.
C Wang, P Lai, M Chen and H Chen. Antioxidative capacity produced by Bifidobacterium- and Lactobacillus acidophilus-mediated fermentations of konjac glucomannan and glucomannan oligosaccharides. Journal of the Science of Food and Agriculture 2008; 88(7), 1294-1300.
T Pan, S Peng, Z Xu, B Xiong, C Wen, M Yao and J Pang. Synergetic degradation of konjac glucomannan by γ-ray irradiation and hydrogen peroxide. Carbohydrate Polymers 2013; 93(2), 761-767.
Y Zhao, J Wang, Y Zhang, R He, Y Du and G Zhong. Hydration properties and mesoscopic structures of different depolymerized konjac glucomannan: Experiments and molecular dynamics simulations. Food Hydrocolloids 2024; 151, 109853.
CA Ariestanti, V Seechamnanturakit, E Harmayani and S Wichienchot. Optimization on production of konjac oligo-glucomannan and their effect on the gut microbiota. Food Science & Nutrition 2019; 7(2), 788-796.
Anggela, E Harmayani, W Setyaningsih and S Wichienchot. Prebiotic effect of porang oligo-glucomannan using fecal batch culture fermentation. Food Science and Technology 2021; 42, e06321.
A Dinoto, CC Watumlawar and Yopi. In vitro modulation of human intestinal microbiota by mannoligosaccharides synthesized from Amorphophallus muelleri glucomannan. Microbiology Indonesia 2016; 7(4), 144-151.
AH Safitri, Yopi and A Meryandini. Enzymatic hydrolysis of porang by Streptomyces violascens BF 3.10 mannanase for the production of mannooligosaccharides. Media Peternakan 2014; 37(3), 143-214.
A Anggela, W Setyaningsih, S Wichienchot and E Harmayani. Oligo-glucomannan production from porang (Amorphophallus oncophyllus) by enzymatic hydrolysis using β-mannanase. Indonesian Food and Nutrition Progress 2020; 17(1), 23-27.
X Shi, J Yin, L Zhang, X Huang and S Ni. Studies on O-acetyl-glucomannans from Amorphophallus species: Comparison of physicochemical properties and primary structures. Food Hydrocolloids 2019; 89, 503-511.
J Chen, D Liu, B Shi, H Wang, Y Cheng and W Zhang. Optimization of hydrolysis conditions for the production of glucomanno-oligosaccharides from konjac using β-mannanase by response surface methodology. Carbohydrate Polymers 2013; 93(1), 81-88.
I Ratcliffe, PA Williams, C Viebke and J Meadows. Physicochemical characterization of konjac glucomannan. Biomacromolecules 2005; 6(4), 1977-1986.
X Du, J Li, J Chen and B Li. Effect of degree of deacetylation on physicochemical and gelation properties of konjac glucomannan. Food Research International 2012; 46(1), 270-278.
J Rungruangsaphakun and S Keawsompong. Optimization of hydrolysis conditions for the mannooligosaccharides copra meal hydrolysate production. 3 Biotech 2018; 8(3), 169.
J Huebner, RL Wehling and RW Hutkins. Functional activity of commercial prebiotics. International Dairy Journal 2007; 17(7), 770-775.
ES Rahayu, IH Rusdan, A Athennia, RZ Kamil, PC Pramesi, Y Marsono, T Utami and J Widada. Safety assessment of indigenous probiotic strain Lactobacillus plantarum Dad-13 isolated from dadih using Sprague Dawley rats as a model. American Journal of Pharmacology and Toxicology 2019; 14(1), 38-47.
S Zhang, H Hu, L Wang, F Liu and S Pan. Preparation and prebiotic potential of pectin oligosaccharides obtained from citrus peel pectin. Food Chemistry 2018; 244, 232-237.
X Luo, X Yao, C Zhang, X Lin and B Han. Preparation of mid-to-high molecular weight konjac glucomannan (MHKGM) using controllable enzyme-catalyzed degradation and investigation of MHKGM properties. Journal of Polymer Research 2012; 19, 989.
YR Herrero, KL Camas and A Ullah. Characterization of biobased materials. In: S Ahmed and Annu (Eds.). Advanced applications of biobased materials: Food, biomedical, and environmental applications. Elsevier Scince, Amsterdam, Netherland, 2023.
ABD Nandiyanto, R Ragadhita and M Fiandini. Interpretation of fourier transform infrared spectra (FTIR): A practical approach in the polymer/plastic thermal decomposition. Indonesian Journal of Science and Technology 2023; 8(1), 113-126.
S Mahanta, J Shree, SC Santra, D Moulick and Akbar Hossain. Deciphering of mycogenic nanoparticles by spectroscopic methods. In: M Kuddus, IZ Ahmad and CM Hussain (Eds.). Myconanotechnology and application of nanoparticles in biology: Fundamental concepts, mechanism and industrial applications. Academic Press, Massachusetts, 2023.
RK Suryawanshi and N Kango. Production of mannooligosaccharides from various mannans and evaluation of their prebiotic potential. Food Chemistry 2021; 334, 127428.
P Tripetch, S Lekhavat, S Devahastin and C Borompichaichartkul. Antioxidant activities of konjac glucomannan hydrolysates of different molecular weights at different values of pH. Foods 2023; 12(18), 3406.
Y Li, H Liu, Y Xie, KI Shabani and X Liu. Preparation, characterization and physicochemical properties of konjac glucomannan depolymerized by ozone assisted with microwave treatment. Food Hydrocolloids 2021; 119, 106878.
Y Zhang, Y Zhao, W Yang, G Song, P Zhong, Y Ren and G Zhong. Structural complexity of Konjac glucomannan and its derivatives governs the diversity and outputs of gut microbiota. Carbohydrate Polymers 2022; 292, 119639.
T Hongbo, W Lan, L Yanping and D Siqing. Effect of acidolysis and oxidation on structure and properties of konjac glucomannan. International Journal of Biological Macromolecules 2019; 130, 378-387.
Z Chen, S Wang, L Shang, P Zhou, J Li and B Li. An efficient and simple approach for the controlled preparation of partially degraded konjac glucomannan. Food Hydrocolloids 2020; 108, 106017.
B Cai, X Yi, Q Han, J Pan, H Chen, H Sun and P Wan. Structural characterization of oligosaccharide from Spirulina platensis and its effect on the faecal microbiota in vitro. Food Science and Human Wellness 2022; 11(1), 109-118.
J Yin, L Ma, M Xie, S Nie and J Wu. Molecular properties and gut health benefits of enzyme-hydrolyzed konjac glucomannans. Carbohydrate Polymers 2020; 237, 116117.
L Wang, G Huang, T Xu and J Xiao. Characterization of carboxymethylated konjac glucomannan for potential application in colon-targeted delivery. Food Hydrocolloids 2019; 94, 354-362.
WHV Zyl, SH Rose, K Trollope and JF Gorgens. Fungal β-mannanases: Mannan hydrolysis, heterologous production and biotechnological applications. Process Biochemistry 2010; 45(8), 1203-1213.
M Zhang, X Chen, Z Zhang, C Sun, L Chen, H He, B Zhou and Y Zhang. Purification and functional characterization of endo-β-mannanase MAN5 and its application in oligosaccharide production from konjac flour. Applied Microbiology and Biotechnology 2009; 83(5), 865-873.
J Liu, A Basit, T Miao, F Zheng, H Yu, Y Wang, W Jiang and Y Cao. Secretory expression of β-mannanase in Saccharomyces cerevisiae and its high efficiency for hydrolysis of mannans to mannooligosaccharides. Applied Microbiology and Biotechnology 2018; 102, 10027-10041.
W Xu, M Han, W Zhang, F Zhang, F Lei, K Wang and J Jiang. Production of manno-oligosaccharide from Gleditsia microphylla galactomannan using acetic acid and ferrous chloride. Food Chemistry 2021; 346, 128844.
GR Gibson, R Hutkins, ME Sanders, SL Prescott, RA Reimer, SJ Salminen, K Scott, C Stanton, KS Swanson, PD Cani, K Verbeke and G Reid. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nature Reviews Gastroenterology & Hepatology 2017; 14(8), 491-502.
J Yang, N Vittori, W Wang, Y Shi, JL Hoeflinger, MJ Miller and Y Pan. Molecular weight distribution and fermentation of mechanically pre-treated konjac enzymatic hydrolysates. Carbohydrate Polymers 2017; 159, 58-65.
F Li, X Sun, W Yu, C Shi, X Zhang, H Yu and F Ma. Enhanced konjac glucomannan hydrolysis by lytic polysaccharide monooxygenases and generating prebiotic oligosaccharides. Carbohydrate Polymers 2021; 253, 117241.
ME Kustyawati, ME Nurlita, EG Fadhallah and S Rizal. Prebiotic activity of Lactobacillus casei grown on medium containing Hylocereus undatus extract and its use in the fermentation of goat’s milk kefir. Biodiversitas Journal of Biological Diversity 2022; 23(12), 6513-6519.
A Song, Y Mao, K Siu and J Wu. Bifidogenic effects of Cordyceps sinensis fungal exopolysaccharide and konjac glucomannan after ultrasound and acid degradation. International Journal of Biological Macromolecules 2018; 111, 587-594.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






