Anti-Bacterial Potential of Multi-strain Probiotics Lactiplantibacillus plantarum (Strain Dad-13 and FNCC-0250) and Lacticaseibacillus paracasei GMRMP-001 against Escherichia coli FNCC-0091 In Vitro and In Vivo
DOI:
https://doi.org/10.48048/tis.2024.8280Keywords:
Escherichia coli, Gut histology analysis, Multi-strains, Probiotic, Short chain fatty acidAbstract
Lactiplantibacillus plantarum Dad-13, Lactiplantibacillus plantarum FNCC-0250 and Lacticaseibacillus paracasei GMRMP-001 are probiotics strain isolated from Indonesian local and traditional fermented foods. This study aims to evaluate the administration of multi-strain probiotics at dose of 109 CFU/mL/day for 14 days against the diarrhea-causing bacteria Escherichia coli FNCC-0091 at a dose of 1011 CFU/mL/day. Thirty rats were randomized and the divided into 5 groups, including control group, skim milk group, single-strain group, multi-strain group and E. coli group. Antibacterial activity was analyzed in vitro, feed intake, body weight were analyzed, microbial analysis was carried out on feces and cecum, short chain fatty acids were analyzed and intestinal morphology was measured. The result showed that the single-strain and multi-strain inhibition zone in in vitro test produces a very strong inhibitory potential (> 9 mm). The number of L. plantarum and L. paracasei increased and the number of E. coli decreased significantly in the feces and cecum of Rat indicating the survival rate of the probiotic bacteria in the digestive tract and their ability to fight pathogenic bacteria. The concentration of acetic acid, propionic acid and butyric acid compounds also increased, indicating that the digestive tract is at a low pH so that it can inhibit the growth of pathogenic bacteria in rats. In addition, consumption of multi-strain probiotics does not have a negative effect on intestinal morphology. The conclusion is that multi-strain probiotics are able to inhibit pathogenic bacteria in vivo and in vitro.
HIGHLIGHTS
- Multi-strain probiotics isolated from traditional Indonesian fermented foods can reduce the potential for diarrhea.
- Single-strain and multi-strain have the same effectiveness in inhibiting pathogenic bacteria as seen from the growth of the bacteria.
- Digestive conditions were damaged in rat that were not given probiotic intervention, but when they received probiotic intervention, digestive conditions improved.
GRAPHICAL ABSTRACT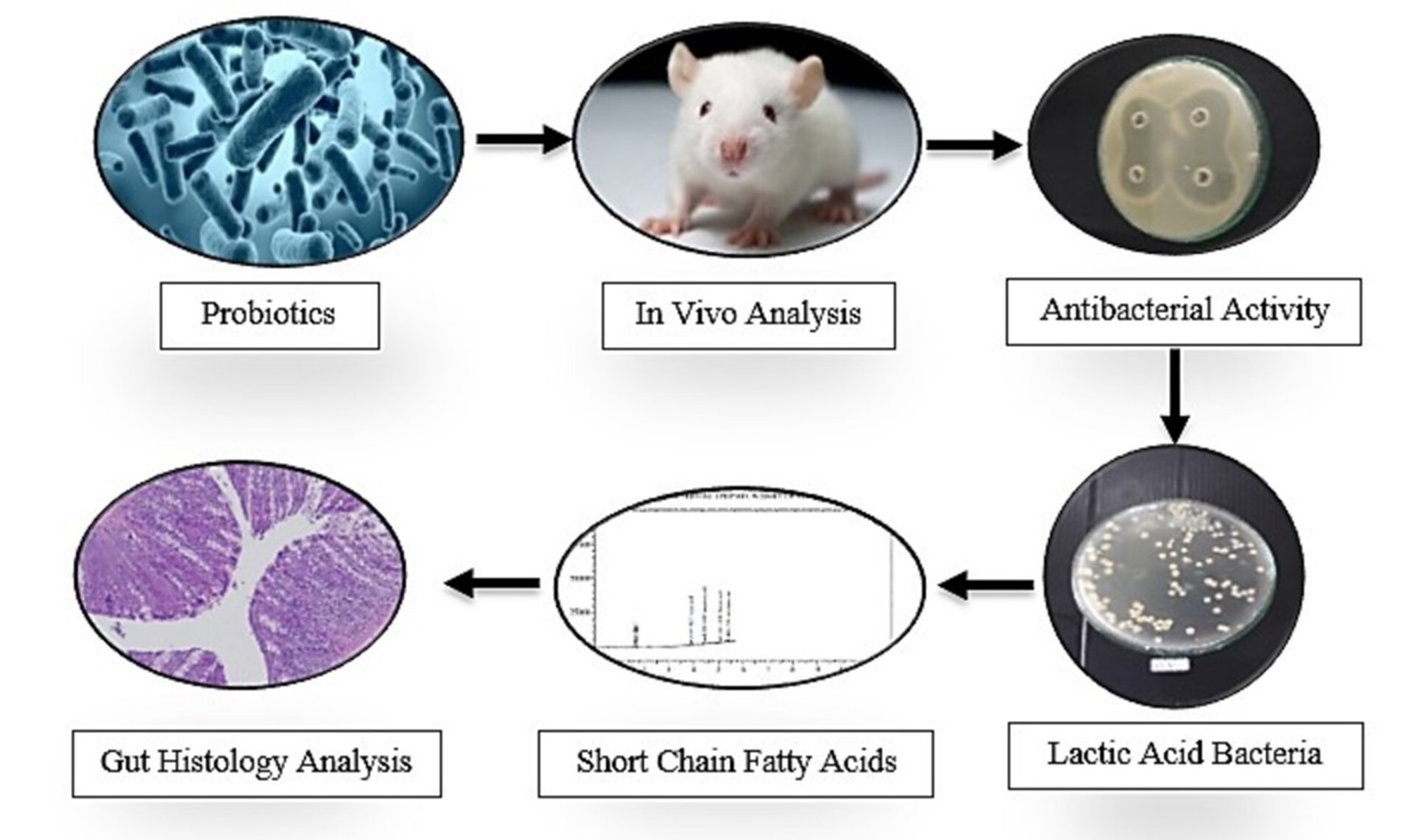
Downloads
References
Y Hu, Z Mengna, L Zhaoxin, LV Fengxia, Z Haizhen and B Xiaomei. L. johnsonii, L. plantarum, and L. rhamnosus alleviated enterohaemorrhagic Escherichia coli-induced diarrhoea in mice by regulating gut microbiota. Microb. Pathog. 2021; 154, 104856.
World Health Organization, Available at: www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease, accessed November 2022.
Food and Agricultural Organization and World Health Organization. Guidelines for the evolution of probiotics in food. Food and Agricultural Organization and World Health Organization, Ontario, Canada, 2002.
S Rodriguez-Sanchez, P Fernandez-Pacheco, S Sesena, C Pintado and ML Palop. Selection of probiotic Lactobacillus strains with antimicrobial activity to be used as biocontrol agents in food industry. LWT 2021; 143, 111142.
CM Galdeano, SI Cazorla, JML Dumit, E Vélez and G Perdigón. Beneficial effects of probiotic consumption on the immune system. Ann. Nutr. Metab. 2019; 74, 115-24.
NP Shah, FA Jolene and RR Ramakanth. Populations of Lactobacillus acidophilus, Bifidobacterium spp. and Lactobacillus casei in commercial fermented milk products. Biosci. Microflora 2000; 19, 35-9.
ES Rahayu, Y Agung, Mariyatun, W Linda, U Tyas and W Koichi. Molecular characteristic of indigenous probiotics strains from Indonesia. Int. J. Probiotics Prebiotics 2015; 10, 109-16.
BT Sumaryati, U Tyas and Suparmo. The effect of Escherichia coli infection and administration of Lactobacillus plantarum Dad-13 on the fecal microbiota wistar rats. agriTECH 2009; 29, 165-70.
AO Rahmi. 2023, Selection of lactic acid bacteria from Indonesian Dadih originated from Alahan Panjang, West Sumatera, as a probiotic candidate. Master Thesis. Gadjah Mada University, Yogyakarta, Indonesia.
Z Noor, NC Muhammad, I Retno and Sardjono. Screening of Lactobacillus plantarum producing lactic acid for mocaf fermentation. agriTECH 2017; 37, 437-42.
W Mastuti and FY Era. Effectiveness of single and multi-strain probiotics against Escherichia coli in vitro. J. Sci. Tech. 2018; 7, 178-87.
DK Ratna, M Evita, ES Rahayu, MN Cahyanto, R Wikandari and T Utami. Indigenous lactic acid bacteria from halloumi cheese as a probiotic candidate of Indonesian origin. Int. J. Probiotics Prebiotics 2021; 16, 39-44.
PG Reeves, FH Nielsen and GC Fahey. AIN-93 purified diets for laboratory rodents: Final report of the American institute of nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993; 123, 1939-51.
JS Zhou, Q Shu and KJ Rutherfurd. Safety assessment of potential probiotic lactic acid bacterial strains Lactobacillus rhamnosus HN001, Lactobacillus acidophilus HN017, and Bifidobacterium lactis HN019 in BALB/c Rat. Int. J. Food Microbiol. 2000; 56, 87-96.
C Bujalance, M Jiménez-Valera and E Moreno. A selective differential medium for Lactobacillus plantarum. J. Microbiol. Methods 2006; 66, 572-5.
Q Zhang, Y Pan, M Wang, L Sun, Y Xi, M Li and Q Zeng. In vitro evaluation of probiotic properties of lactic acid bacteria isolated from the vagina of yak (Bos grunniens). PeerJ 2022; 10, 15-35.
J Ferdouse, S Paul, T Chowdhury, F Ali, S Islam and TJ Hossain. Probiotic characteristics of Pediococcus pentosaceus and Apilactobacillus kunkeei strains: The lactic acid bacteria isolated from Bangladeshi natural honey. Appl. Food Biotechnol. 2023; 10, 33-45.
ES Rahayu, HR Ilzamha, A Armita, ZK Rafli, CP Putrika, M Yustinus, U Tyas and W Jaka. Safety assessment of indigenous probiotic strain Lactobacillus plantarum Dad-13 isolated from dadih using sprague dawley rats as a model. Am. J. Pharmacol. Toxicol. 2019; 14, 38-47.
AY Ikhsani. 2016, Safety evaluation of the indigenous probiotic strain Lactobacillus plantarum Mut 7 using the sprague dawley rat model. Master Thesis. Gadjah Mada University, Yogyakarta, Indonesia.
M Kechagia, D Basoulis, S Konstantopoulou, D Dimitriadi, K Gyftopoulou, N Skarmoutsou and EM Fakiri. Health benefits of probiotics: A review. ISRN Nutr. 2013; 2013, 481651.
XF Chen, X Chen and X Tang. Short-chain fatty acid, acylation and cardiovascular diseases. Clin. Sci. 2020; 134, 657-76.
A Takeuchi and H Sprinz. Electron-microscope studies of experimental Salmonella infection in the preconditioned guinea pig: II. Response of the intestinal mucosa to the invasion by Salmonella typhimurium. Am. J. Pathol. 1967; 51, 137-61.
VO Oyetayo, FC Adetuyi and FA Akinyosoye. Safety and protective effect of Lactobacillus acidophilus and Lactobacillus casei used as probiotic agent in vivo. Afr. J. Biotechnol. 2003; 2, 448-52.
Y Mayangsari, S Natsumi, X Wenxi, M Chinatsu, T Yuki, U Osamu, S Tomohiro, S Yoshiharu, Y Yoshinari and S Takuya. 3,5,7,3’,4’-Pentamethoxyflavone enhances the barrier function through transcriptional regulation of the tight junction in human intestinal Caco-2 cells. J. Agric. Food Chem. 2021; 69, 10174-83.
Y Mayangsari, O Mayu, M Chinatsu, T Yuki, U Osamu, S Tomohiro, S Yoshiharu, Y Yoshinari and S Takuya. 5,7-Dimethoxyflavone enhances barrier function by increasing occludin and reducing claudin-2 in human intestinal Caco-2 cells. J. Funct. Foods 2021; 85, 104641.
S Koch and A Nusrat. The life and death of epithelia during inflammation: Lessons learned from the gut. Ann. Rev. Pathol. Mech. Dis. 2012; 7, 35-60.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






