Exploration of Antituberculosis Potential of Ni(II) and Zn(II) Serine-Tyrosine Dithiocarbamate Complexes: Synthesis, Characterization, In Silico Profile, and In Vitro Evaluation
DOI:
https://doi.org/10.48048/tis.2025.10800Keywords:
Metal-complexes, Peptide-dithiocarbamate, Antituberculosis, M. tuberculosis, Molecular dockingAbstract
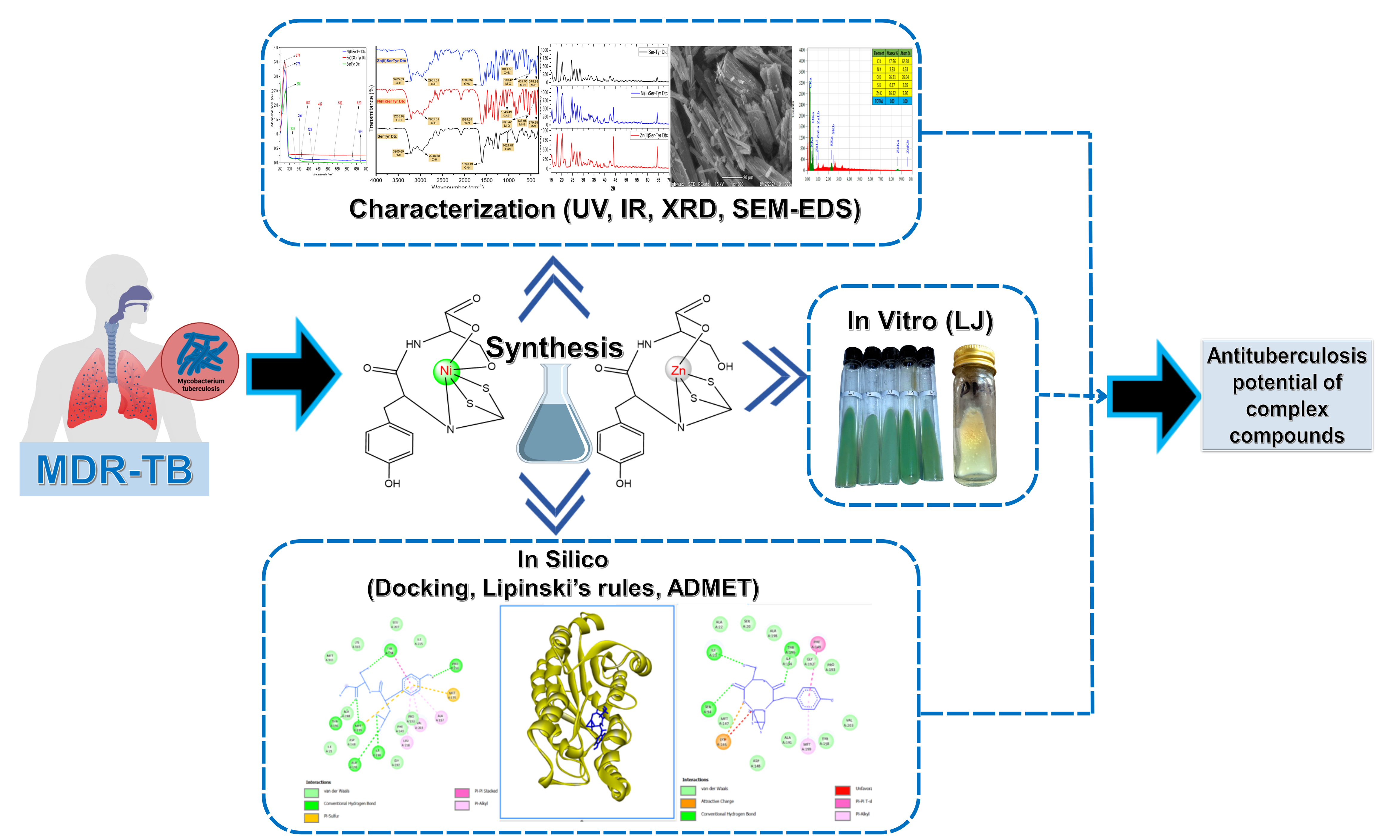
Tuberculosis (TB) remains a global health challenge, with millions of new cases and deaths reported each year. The emergence of drug resistance and treatment side effects has driven the development of new therapeutic agents, including the use of dithiocarbamate metal complex compounds. This study aims to synthesize and characterize Ni(II) and Zn(II) complexes with serine-tyrosine dithiocarbamate ligands and evaluate their antituberculosis bioactivity. The method in this study involves the synthesis and characterization of Ni(II) and Zn(II) complexes with serine-tyrosine dithiocarbamate, including melting point determination, conductivity measurement, and spectroscopic analyses (UV-Vis, FT-IR, XRD, and SEM-EDS). Furthermore, the bioactivity of the synthesized metal complexes was evaluated using both in silico approaches (Lipinski's rule of 5, ADMET profiling, and molecular docking) and in vitro testing using the Lowenstein-Jensen method. The synthesis and characterization results show that the successfully synthesized metal complexes are solid, non-electrolyte, stable, and have typical characteristics of dithiocarbamate complexes. In silico analysis shows compliance with Lipinski’s rule and most ADMET parameters, indicating potential as oral drug candidates. Molecular docking revealed strong interactions, particularly for the Ni(II)Ser-Tyr Dtc complex with the highest binding score ( 95.0923 kJ/mol), exceeding INH ( 65.8232 kJ/mol). In vitro results confirmed antibacterial activity by the absence of M. tuberculosis H37Rv colony growth. These findings suggest the metal complexes have potential as antituberculosis agents.
HIGHLIGHTS
- Ni(II) and Zn(II) metal complexes with serine-tyrosine dithiocarbamate ligands were successfully synthesized and characterized (UV-Vis, FT-IR, XRD, SEM-EDS).
- The metal complexes met Lipinski’s rule and most ADMET criteria, with Ni(II)Ser-Tyr Dtc showing the strongest binding to Enoyl-ACP reductase, outperforming isoniazid.
- In vitro assays confirmed antituberculosis activity, indicated by the absence of tuberculosis H37Rv colony growth.
- The results highlight the potential of these metal complexes as promising antituberculosis drug candidates.
GRAPHICAL ABSTRACT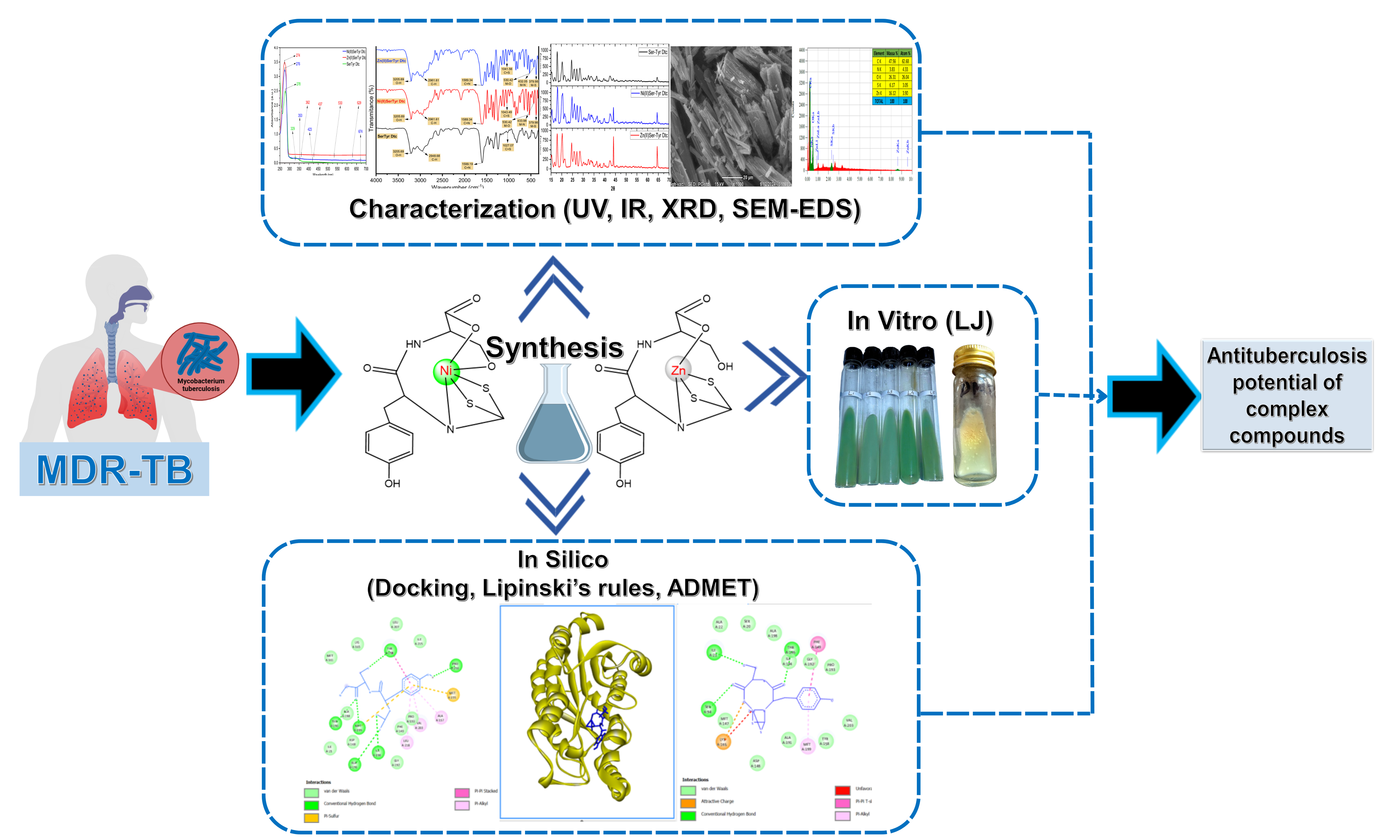
Downloads
References
World Health Organization. Global Tuberculosis Report 2023. Geneva, Switzerland, 2024.
AJ Reis, JLD Pizzol, R Gattelli, AV Groll, DF Ramos, IB Ramis, A Kritski, JRLE Silva and PEAD Silva. Thesis and dissertations examining tuberculosis in brazil between 2013 and 2019: An overview. Journal of the Brazilian Society of Tropical Medicine 2022; 55, e0198-e2022.
I Gajic, N Tomic, B Lukovic, M Jovicevic, D Kekic, M Petrovic, M Jankovic, A Trudic, DM Culafic, M Milenkovic and N Lukovic. A comprehensive overview of antibacterial agents for combating multidrug-resistant bacteria: the current landscape, development, future opportunities, and challenges. Antibiotics 2025; 14(3), 221.
MN Patel, AJ Patel, MN Nandpal, MA Raval, RJ Patel, AA Patel, KR Paudel, PM Hansbro, SK Singh, G Gupta, K Dua and SG Patel. Advancing against drug-resistant tuberculosis: an extensive review, novel strategies and patent landscape. Naunyn-Schmiedeberg's Archives of Pharmacology 2025; 398, 2127-2150.
K Srinivasulu, KH Reddy, K Anuja, D Dhanalakshmi and YB Nagamani. Design, synthesis, spectral characterization, dna binding and antibacterial studies of ternary metal complexes with 1,10 phenanthroline and 2-acetylthiophene-4-phenyl-3-thiosemicarbazone. Trends in Sciences 2022; 19(16), 5653.
N Kumar, R Kaushal and P Awasthi. Transition metal complexes as antibacterial agents: An overview. Springer Nature, New York, 2025.
CD Natale, ID Benedictis, AD Benedictis and D Marasco. Metal-peptide complexes as promising antibiotics to fight emerging drug resistance: New perspectives in tuberculosis. Antibiotics 2020; 9(6), 337.
SK Nandanwar and HJ Kim. Anticancer and antibacterial activity of transition metal complexes. ChemistrySelect 2019; 4(5), 1706-1721.
NO Boadi, M Degbevi, SA Saah, M Badu, LS Borquaye and NK Kortei. Antimicrobial properties of metal piperidine dithiocarbamate complexes against Staphylococcus aureus and Candida albicans. Scientific African 2021; 12, e00846.
AS Alfahdawi, MS Sawsan SE Saleh and MM Saleh. Synthesis and study of N, N' -(ethane-1,2-diyl)bis(1-phenyl methanimine) and their complex derivative with in-vivo and in-vitro Bacterial biological study. Egyptian Journal of Chemistry 2021; 64(6), 2879-2888.
TA Saiyed, JO Adeyemi and DC Onwudiwe. The structural chemistry of Zinc(II) and Nickel(II) dithiocarbamate complexes. Open Chemistry 2021; 19(1), 974-986.
S Balakrishnan, S Duraisamy, M Kasi, S Kandasamy, R Sarkar and A Kumarasamy. Syntheses, physicochemical characterization, antibacterial studies on potassium morpholine dithiocarbamate Nickel (II), Copper (II) metal complexes and their ligands. Heliyon 2019; 5(5), e01687.
N Baartzes, T Stringer, R Seldon, DF Warner, CD Kock, PJ Smith and GS Smith. Synthesis, characterization and antimicrobial evaluation of mono- and polynuclear ferrocenyl derived amino and imino complexes. Journal of Organometallic Chemistry 2016; 809, 79-85.
SA Khan, W Ahmad, KS Munawar and S Kanwal. Synthesis, spectroscopic characterization and biological evaluation of Ni(II), Cu(II) and Zn(II) complexes of diphenyldithiocarbamate. Indian Journal of Pharmaceutical Sciences 2018; 80(3), 480-488.
NJ Porter, NH Christianson, C Decroos and DW Christianson. Structural and functional influence of the glycine-rich loop g302gggy on the catalytic tyrosine of histone deacetylase 8. Biochemistry 2016; 55(48), 6718-6729.
S Medici, M Peana, VM Nurchi and MA Zoroddu. The involvement of amino acid side chains in shielding the nickel coordination site: An NMR study. Molecules 2013; 18(10), 12396-12414.
E Kasotakis and A Mitraki. Silica biotemplating by self-assembling peptides via serine residues activated by the peptide amino terminal group. Peptide Science 2012; 98(6), 501-509.
A Chollet, L Maveyraud, C Lherbet and V Bernardes-Genisson. An overview on crystal structures of inha protein: Apo-form, in complex with its natural ligands and inhibitors. European Journal of Medicinal Chemistry 2018; 146, 318-343.
DEV Pires, TL Blundell and DB Ascher. pkCSM: Predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. Journal of Medicinal Chemistry 2015; 58(9), 4066-4072.
MBD Avila, G Bitencourt-Ferreira and WFD Azevedo. structural basis for inhibition of enoyl-[acyl carrier protein] reductase (InhA) from Mycobacterium tuberculosis. Current medicinal chemistry 2020; 27(5), 745-759.
EP Istyastono. Docking studies of curcumin as a potential lead compound to develop novel dipeptydyl peptidase-4 inhibitors. Indonesian Journal of Chemistry 2009; 9(1), 132-136.
AN Usoltsev, IA Shentseva, VR Shayapov, PE Plyusnin, IV korolkov, PA Abramov, MN Sokolov and SA Adonin. Iodide bismuth(iii) complexes with 1-ethyl-3-methylpyridinium: Structure, thermal stability, and optical properties. Russian Journal of Inorganic Chemistry 2022; 67, 1979-1986.
RA Arfah, E Pratiwi, I Raya, H Natsir, P Taba, H Rasyid, M Alfliadhi, AB Khaerunnisa, R Irfandi and S Jarre. Design, synthesis and characterization of Mn(II) cysteine-tyrosine dithiocarbamate complex for against the cancer on MCF-7 breast cancer cell line. Asian Pacific Journal of Cancer Prevention 2024; 25(9), 3251-3261.
AK Mishra, N Manav and NK Kaushik. Organotin(IV) complexes of thiohydrazones: Synthesis, characterization and antifungal study. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2005; 61(13-14), 3097-3101.
S Santi, AW Wahab, I Raya and A Ahmad. Synthesis and interaction of adenosine-5’-triphosphate with rare earth metal Europium (Eu3+). AIP Conference Proceedings 2020; 2296, 020074.
B Annuar, JO Hill and RJ Magee. Nickel(II) and Copper(II) complexes of mono-ethanol and di-ethanol-dithiocarbamic acid. Journal of Inorganic and Nuclear Chemistry 1974; 36(6), 1253-1257.
I Sakiyan, R Ozdemir and H Ogutcu. Synthesis, characterization, and antimicrobial activities of new N-(2-hydroxy-1-naphthalidene)-amino acid (L-Tyrosine, L-Arginine, and L-Lysine) Schiff bases and their Manganese (III) complexes. Synthesis and Reactivity in Inorganic, Metal-Organic, and Nano-Metal Chemistry 2014; 44(3), 417-423.
M Ghorbanloo, M Jaworska, P Paluch, GD Li and L Zhou. Synthesis, characterization, and catalytic activity for thioanisole oxidation of homogeneous and heterogeneous binuclear Manganese(II) complexes with amino acid-based ligands. Transition Metal Chemistry 2013; 38, 511-521.
J Dharmaraja, J Balamurugan and S Shobana. Synthesis, structural elucidation, microbial, antioxidant and nuclease activities of some novel divalent M(II) complexes derived from 5-fluorouracil and l-tyrosine. Journal of Saudi Chemical Society 2017; 21(1), S67-S76.
S Santi, A Wahab, I Raya, A Ahmad and M Maming. Synthesis, spectroscopic (FT-IR, UV-Visible) study, and homo-lumo analysis of adenosine triphosphate (ATP) doped trivalent terbium. Journal of Molecular Structure 2021; 1237, 130398.
E Pratiwi, I Raya, H Natsir, R Irfandi, P Taba, R Arfah, H Rasyid, Y Hala, S Kasim, AB Khaerunnisa, B Ilham, M Mazaya, Y Tanzil and D Luthfiana. Investigations of Ni(II)cysteine-tyrosine dithiocarbamate complex: Synthesis, characterization, molecular docking, molecular dynamic, and anticancer activity on MCF-7 breast cancer cell line. Asian Pacific Journal of Cancer Prevention 2024; 25(4), 1301-1313.
LZ Benet, CM Hosey, O Ursu and TI Oprea. BDDCS, the Rule of 5 and drugability. Advanced Drug Delivery Reviews 2016; 101, 89-98.
CA Lipinski, F Lombardo, BW Dominy and PJ Feeney. experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews 2012; 46(1-3), 3-26.
J Mosquera, I Garcia and LM Liz-Marzan. Cellular uptake of nanoparticles versus small molecules: A matter of size. Accounts of Chemical Research 2018; 51(9), 2305-2313.
K Adachi, T Sasaki, A Arai, M Shimizu and H Yamazaki. Impact of variability of in silico and in vitro octanol/water partition coefficients of compounds on the input parameters and results of simplified human physiologically based pharmacokinetic models after virtual oral administrations. The Journal of Toxicological Sciences 2024; 49(10), 459-466.
R Raj. Analysis of non-structural proteins, NSPs of SARS-CoV-2 as targets for computational drug designing. Biochemistry and Biophysics Reports 2021; 25, 100847.
I Nyamba, CB Sombie, M Yabre, H Zime-Diawara, J Yameogo, S Ouedraogo, A Lechanteur, R Semde and B Evrard. Pharmaceutical approaches for enhancing solubility and oral bioavailability of poorly soluble drugs. European Journal of Pharmaceutics and Biopharmaceutics 2024; 204, 114513.
EV Radchenko, AS Dyabina, VA Palyulin and NS Zefirov. Prediction of human intestinal absorption of drug compounds. Russian Chemical Bulletin 2016; 65, 576-580.
KMA Azzam. SwissADME and pkCSM webservers predictors: An integrated online platform for accurate and comprehensive predictions for in silico ADME/T properties of artemisinin and its derivatives. Engineering and Technology 2022; 325(2), 14-21.
LM Lagares, Y Perez-Castillo, N Minovski and M Novic. Structure-function relationships in the human p-glycoprotein (abcb1): Insights from molecular dynamics simulations. International journal of molecular sciences 2021; 23(1), 362.
N Holford and DS Yim. Volume of distribution. Translational and Clinical Pharmacology 2016; 24(2), 74-77.
JH Lee, S Basith, M Cui, B Kim and S Choi. In silico prediction of multiple-category classification model for cytochrome P450 inhibitors and non-inhibitors using machine-learning method. SAR and QSAR in Environmental Research 2017; 28(10), 863-874.
A Spicakova, P Kraus, T Gucky, V Krystof, M Strnad, V Bazgier, M Otyepka, V Kubickova, M Poruba, Z Racova, I Zapletalova and P Anzenbacher. In vitro and in silico studies of interaction of synthetic 2,6,9-trisubstituted purine kinase inhibitors BPA-302, BP-21 and BP-117 with liver drug-metabolizing cytochromes P450. Physiological Research 2020; 69(4), S627-S636.
R Watanabe, R Ohashi, T Esaki, H Kawashima, Y Natsume-Kitatani, C Nagao and K Mizuguchi. Development of an in silico prediction system of human renal excretion and clearance from chemical structure information incorporating fraction unbound in plasma as a descriptor. Scientific Reports 2019; 9, 18782.
JC Hancox and AF James. Refining insights into high-affinity drug binding to the human ether-a-go-go-related gene potassium channel. Molecular pharmacology 2008; 73(6), 1592-1595.
X Duan, X Xiang and J Xie. Crucial components of Mycobacterium type II fatty acid biosynthesis (Fas-II) and their inhibitors. FEMS Microbiology Letters 2014; 360(2), 87-99.
SR Khan, Y Manialawy and AG Siraki. Isoniazid and host immune system interactions: A proposal for a novel comprehensive mode of action. British Journal of Pharmacology 2019; 176(24), 4599-4608.
MAFA Manan and DB Cordes. Cadmium(II) schiff base complex containing 5-fluoroisatin moiety: Synthesis, characterization, antibacterial activity and structural studies. Trends in Sciences 2022, 19(18), 5796.
CI Yeo, ERT Tiekink and J Chew. Insights into the antimicrobial potential of dithiocarbamate anions and metal-based species. Inorganics 2021; 9(6), 48.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






