Anticancer Activity of Pyrazoline A - D on MCF7 Breast Cancer Cell Line and Molecular Docking on Proliferation Proteins
DOI:
https://doi.org/10.48048/tis.2025.10503Keywords:
Anticancer, Breast cancer, N-phenyl-Pyrazoline, Molecular dockingAbstract
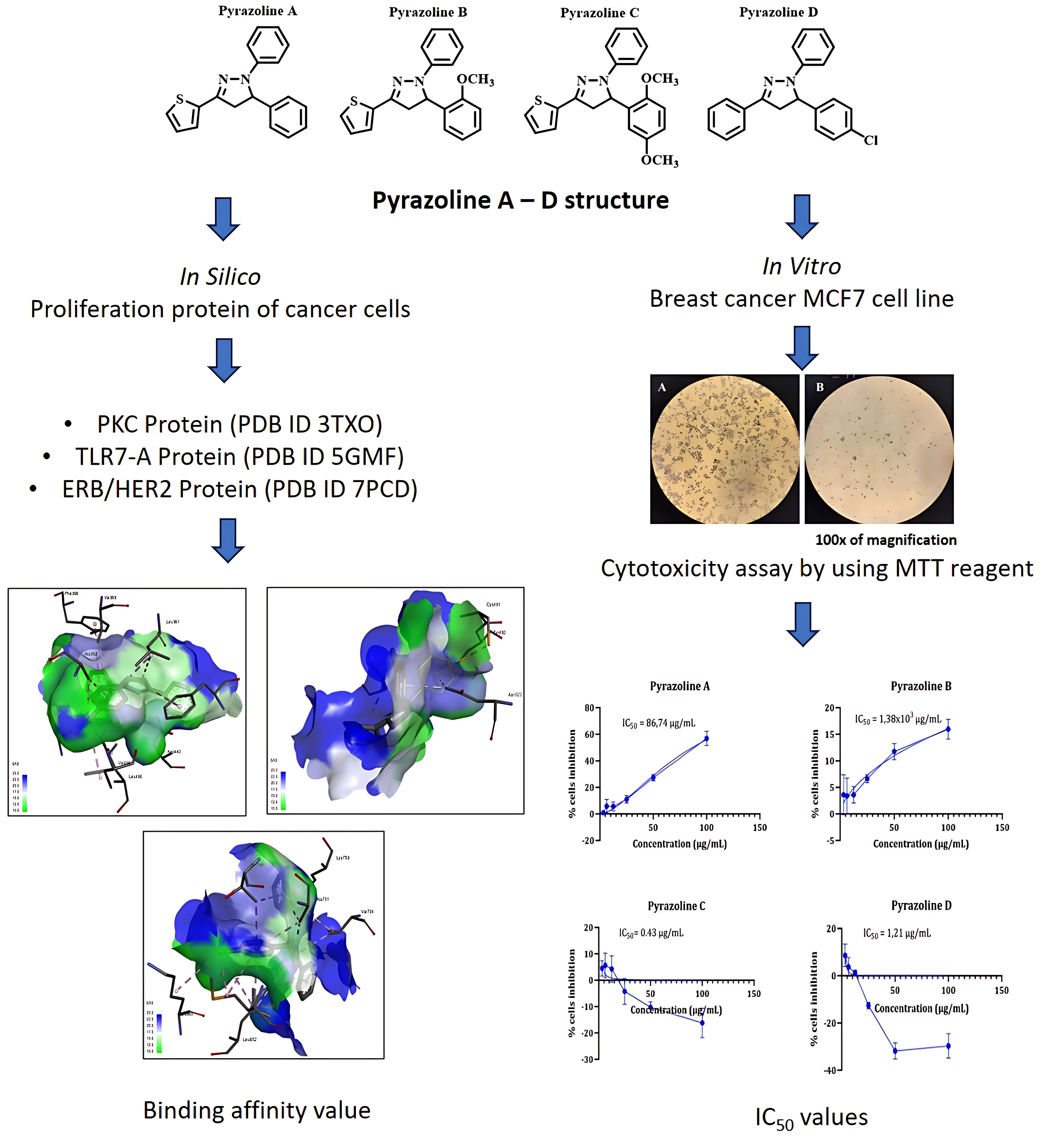
Pyrazoline is a versatile heterocyclic compound with excellent biological activity, especially as a new therapeutic agent for anticancer. This study examined the binding of N-pyrazolines A - D in MCF7 cells to PKC, TRL7, and ERB/HER-2 proteins. N-phenyl-pyrazoline derivatives A, B, C, and D were evaluated for their anticancer activity against the MCF7 breast cancer cell line and their binding affinity to proliferation-related proteins PKC, TLR7, and ERB/HER-2, using in vitro and in silico methods. Pyrazoline C and D exhibited high anticancer activity with IC50 values of 0.43 and 1.21 µg/mL, respectively, while pyrazoline A showed moderate activity (IC50 = 86.74 µg/mL) and pyrazoline B was inactive (IC50 > 1,000 µg/mL). Molecular docking studies revealed that pyrazoline D had the best binding affinity to PKC (–9.532 kcal/mol), TLR7 (–5.964 kcal/mol), and ERB/HER-2 (–8.416 kcal/mol) compared to the other pyrazolines and native ligands. Pyrazoline C also showed better binding affinity than the native ligand for TLR7-A and similar binding affinity to PKC and ERB/HER-2. The binding interactions of pyrazolines C and D involved pi-sigma, pi-pi stacked, alkyl, pi-alkyl, and specific hydrogen-bonding interactions with key amino acid residues in the target proteins. These findings suggest that pyrazolines C and D have potential as anticancer agents with anti-proliferative activities.
HIGHLIGHTS
- Pyrazoline C and D exhibited high anticancer activity, Pyrazoline A showed moderate activity, while pyrazoline B was inactive against MCF7 cells.
- Pyrazoline D had the best binding affinity to PKC, TLR7, and ERB/HER-2 proteins.
- Binding interactions involved pi-sigma, pi-pi stacked, alkyl, pi-alkyl, and hydrogen bonds.
- Pyrazolines C and D are potential anticancer agents with anti-proliferative activity.
GRAPHICAL ABSTRACT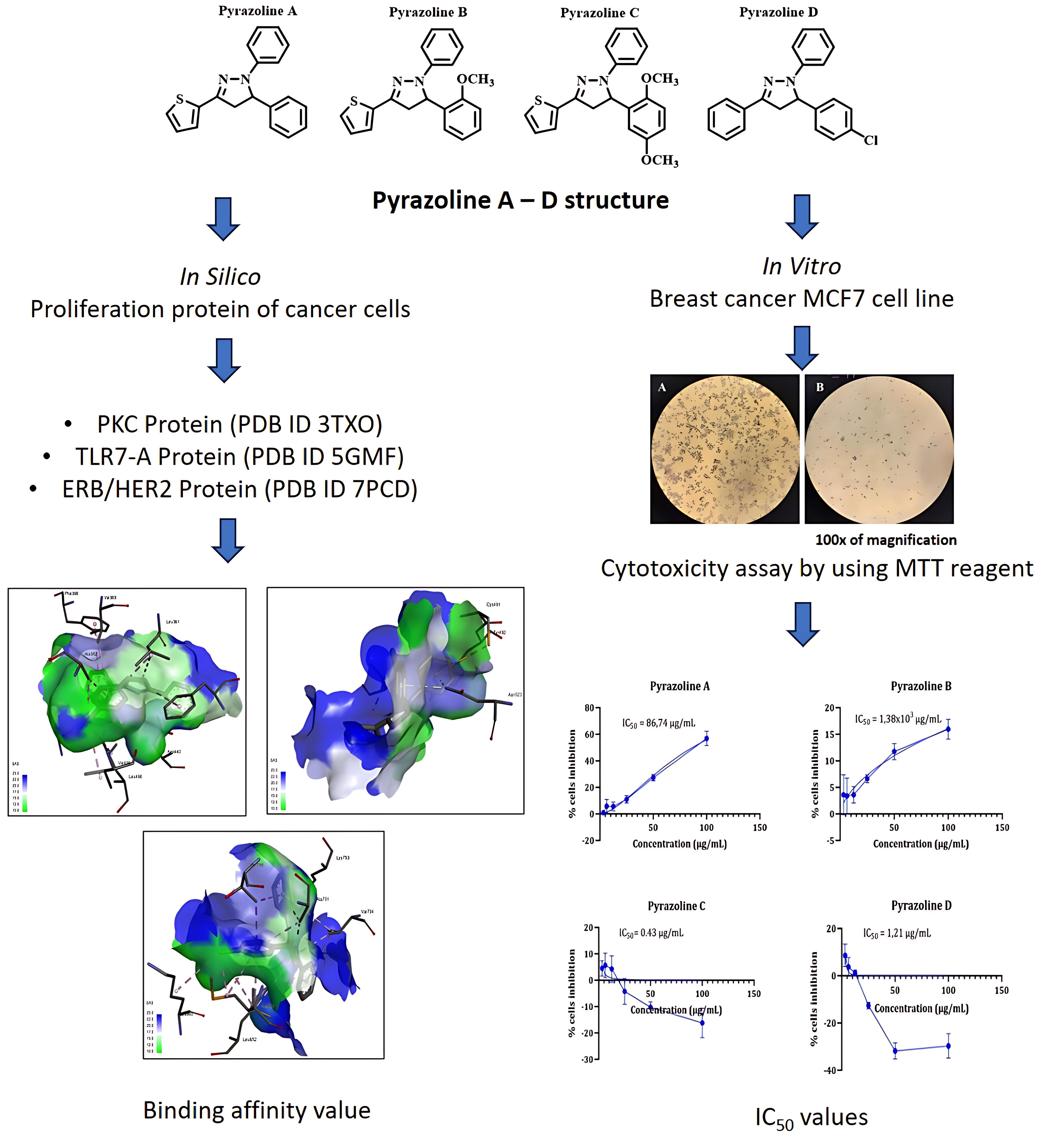
Downloads
References
D Satria, SB Waruwu, EN Sholikhah, M Mustofa, PB Satriyo, TD Wahyuningsih, HL Wiraswati and E. Damayanti. In-silico analysis of N-phenyl-pyrazoline as potential of human epiderma growth receptor-2 (HER-2) inhibitor using molecular docking and MD simulation. Rasayan Journal of Chemistry 2024; 17(2), 356-362.
C Admoun and HN Mayrovitz. The etiology of breast cancer. In: HN Mayrovitz (Ed.). Breast cancer. Exon Publications, Brisbane, Australia, 2022, p. 21-30.
AJ Alejandro, J Urtreger, MG Kazanietz and EDBDK Joffé. Contribution of individual PKC isoforms to breast cancer progression. IUBMB Life 2012; 64(1), 18-26.
V Albert, G Piendl, D Yousseff, H Lammert, M Hummel, O Ortmann, W Jagla, A Gaumann, AK Wege and G Brockhoff. Protein kinase C targeting of luminal (T‑47D), luminal / HER2 ‑ positive (BT474), and triple negative (HCC1806) breast cancer cells in ‑ vitro with AEB071 (Sotrastaurin) is efficient but mediated by subtype specific molecular effects. Archives of Gynecology and Obstetrics 2022; 306(4), 11974-1210.
W Lin, J Huang, Z Yuan, S Feng, Y Xie and W Ma. Protein kinase C inhibitor chelerythrine selectively inhibits proliferation of triple-negative breast cancer cells. Scientific Reports 2015; 7(1), 2022.
J Zhou, L Zhang, S Liu, D Derubeis and D Zhang. Toll-like receptors in breast cancer immunity and immunotherapy. Frontiers in Immunology 2024; 15, 1418025.
M Le, J Lipniarska, P Majka, W Kopyt, M Lejman and J. Zawitkowska. The role of TRL7 / 8 Agonists in cancer therapy, with special emphasis on hematologic malignancies. Vaccines 2023; 1(2), 277
SA Eccles. The epidermal growth factor receptor/Erb-B/HER family in normal and malignant breast biology. The International Journal of Developmental Biology 2011; 55(7-9), 685-696.
AW Hamburger. The role of ErbB3 and its binding partners in breast cancer progression and resistance to hormone and tyrosine kinase directed therapies. Journal of Mammary Gland Biology and Neoplasia 2008; 13(2), 225-233.
TD Wahyuningsih, AAT Suma and E Astuti. Synthesis, anticancer activity, and docking study of N-acetyl pyrazolines from veratraldehyde. Journal of Applied Pharmaceutical Science 2019; 9(3), 14-20.
I Chunaifah, RE Venilita, PJP Tjitda, E Astuti and TD Wahyuningsih. Thiophene-based N-phenyl pyrazolines: synthesis, anticancer activity, molecular docking and ADME study. Journal of Applied Pharmaceutical Science 2024; 14(4), 63-71
M Mustofa, PB Satriyo, AAT Suma, SS Waskitha, TD Wahyuningsih and EN Sholikhah. A potent EGFR inhibitor, N-phenyl-pyrazoline derivative suppresses aggressiveness and cancer stem cell-like phenotype of cervical cancer cells. Drug Design, Development and Therapy 2022; 16, 2325-2339.
TA Ghifari, FF Wulan, E Astuti, V Suryanti, DEK Putri, H Nur and TD Wahyuningsih. Synthesis of furan-based pyrazoline as an anticancer agent: An in vitro and in silico approach toward COX-2 inhibition. Journal of Molecular Structure 2025; 1337, 142125.
D Satria, SB Waruwu, EN Sholikhah, Mustofa, PB Satriyo, TD Wahyuningsih, HL Wiraswati and E Damayanti. In silico study of anticancer activity of pyrazoline C and M as potential selective of cyclooxygenase-2 (Cox-2) inhibitor using molecular docking and MD simulations. International Journal of Applied Pharmaceutics 2024; 16(4), 1-5.
AAT Suma, TD Wahyuningsih and M Mustofa. Synthesis, cytotoxicity evaluation and molecular docking study of n-phenylpyrazoline derivatives. Indonesian Journal of Chemistry 2019; 19(4), 1081-1090.
PB Satriyo, M Mustofa, TD Wahyuningsih, E Damayanti, HL Wiraswati, D Satria, MH Bashari and EN Sholikhah. N-phenyl pyrazoline derivative inhibits cell aggressiveness and enhances paclitaxel sensitivity of triple negative breast cancer cells. Scientific Reports 2024; 14(1), 13200.
HL Wiraswati, MH Bashari, NM Alfarafisa, IF Ma’ruf, EN Sholikhah, TD Wahyuningsih, PB Satriyo, M Mustofa, D Satria and E Damayanti. Pyrazoline B-paclitaxel or doxorubicin combination drugs show synergistic activity against cancer cells: In silico study. Advances and Applications in Bioinformatics and Chemistry 2024; 17, 33-46.
HL Wiraswati, MH Bashari, NM Alfarafisa, IF Ma’ruf, EN Sholikhah, TD Wahyuningsih, PB Satriyo, M Mustofa, D Satria and E Damayanti. Virtual screening of anticancer activity of chalcones and pyrazolines as potential EGFR, VEGFR, and cytochrome P450 inhibitors. Journal of Pharmacy & Pharmacognosy Research 2023; 11(4), 699-713.
FF Wulan, TD Wahyuningsih, E Astuti and N Prasetyo. Towards targeting EGFR and COX-2 inhibitors: Comprehensive computational studies on the role of chlorine group in novel thienyl-pyrazoline derivative. Journal of Biomolecular Structure and Dynamics 2024; 42(19), 9857-9872.
O Trott and AJ Olson. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry 2020; 31(2), 455-461.
D Studio. Dassault Systemes BIOVIA, Discovery studio modelling environment. Dassault Systèmes, France, 2025.
K Haider, M Shafeeque, S Yahya and MS Yar. A comprehensive review on pyrazoline based heterocyclic hybrids as potent anticancer agents. European Journal of Medicinal Chemistry Reports 2021; 5(8), 100042.
BS Gabr, AR Shalabi, MF Said and RF George. 3,5-Disubstituted pyrazoline as a promising core for anticancer agents: Mechanisms of action and therapeutic potentials. Future Medicinal Chemistry 2025; 17(6), 725-745.
B Kurban, BN Sağlık, D Osmaniye, S Levent, Y Özkay and ZA Kaplancıklı. Synthesis and anticancer activities of pyrazole-thiadiazole-based EGFR inhibitors. ACS Omega 2023; 8(34), 31500-31509.
M Rana, R Arif, FI Khan, V Maurya, R Singh, MI Faizan, S Yasmeen, SH Dar, R Alam, A Sahu, T Ahmad and Rahisuddin. Pyrazoline analogs as potential anticancer agents and their apoptosis, molecular docking, MD simulation, DNA binding and antioxidant studies. Bioorganic Chemistry 2021; 108(28), 104665.
G Roman. Anticancer activity of coumarin-pyrazoline hybrids: A mini-review. Chemistry Africa 2024; 7(5), 2307-2319.
D Matiadis and M Sagnou. Pyrazoline hybrids as promising anticancer agents: An up-to-date overview. International Journal of Molecular Sciences 2020; 21(15), 5507.
Y Song, S Feng, J Feng, J Dong, K Yang, Z Liu and X Qiao. Synthesis and biological evaluation of novel pyrazoline derivatives containing indole skeleton as anti-cancer agents targeting topoisomerase II. European Journal of Medicinal Chemistry 2020; 200, 112459.
HM Hafez, BAM Said, AM Sayed, E Alatwi, BGM Youssif, S Bräse and HAM El-Sherief. Design, synthesis, antiproliferative activity, and molecular dynamics simulation of pyrazoline-based derivatives as dual EGFR and HER-2 inhibitors. RSC Advances 2025; 15(12), 9265-9279.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






