Potential of Neolignan from Piper Crocatum as Antimicrobial Against Pathogenic Oral Microbes and Its Prospects as an Oral Medication
DOI:
https://doi.org/10.48048/tis.2026.11608Keywords:
Red betel (Piper crocatum), Neolignan, Antimicrobial, Oral Microbes, ADMET, Drug likeness analysis, In vitro, In silicoAbstract
Red Betel (Piper crocatum) is one of the potential medicinal plants in several traditional medicines, one of which is in the treatment of microbial dental and oral infections. This activity comes from the presence of active secondary metabolite compounds such as neolignan groups which have unique and diverse structures and activities. The aim of this study was to isolate antimicrobial active compounds from red betel leaves and predict their inhibitory mechanism against DNA ligase and lanosterol 14α demethylase enzymes. This study was conducted by isolating red betel leaf extract using column chromatography method in a bioassay guided. The compounds obtained were analysed using UV-Vis, IR, NMR, MS and their antimicrobial activity was tested in vitro and in silico against S. mutans, S. sanguinis, E. faecalis bacteria, and C. albicans fungi. Two compounds were successfully isolated from P. crocatum leaves, namely crocatin B (1) and crocatin A (2). crocatin B (1) was found to be capable of inhibiting the growth of S. mutans ATCC 25175, S. sanguinis ATCC 10556 and E. faecalis ATCC 29212. crocatin A demonstrated inhibitory activity against S. sanguinis. The binding affinity of crocatin B (1) and crocatin A (2) to the DNA ligase enzyme was –6.14 and –7.27 kcal/mol, respectively, while the binding affinity of crocatin B (1) and crocatin A (2) to lanosterol 14α demethylase was –7.91 and –9.04 kcal/mol, respectively. It is evident from the analysis of pharmacokinetic (ADMET) parameters that both crocatin B (1) and crocatin A (2) demonstrate a satisfactory alignment with the predicted pharmacokinetic parameters. In the context of drug likeness analysis, crocatin B and crocatin A do not demonstrate any violation of the established parameters and are thus classified within the toxicity class 4 category. The data demonstrate the potential of crocatin B (1) and crocatin A (2) as antimicrobials against oral pathogenic microbes, as evidenced by in vitro and in silico studies.
HIGHLIGHTS
- crocatin A and crocatin B were successfully isolated from red betel leaves.
- This first report on the antimicrobial activity of crocatin A and B against oral pathogenic microbes (in vitro)
- crocatin A and B have the potential to inhibit the enzymes DNA ligase and lanosterol 14α-demethylase (in silico).
- ADMET studies and drug-likeness analysis indicate that crocatin A and B can be used as candidate drug ingredients.
GRAPHICAL ABSTRACT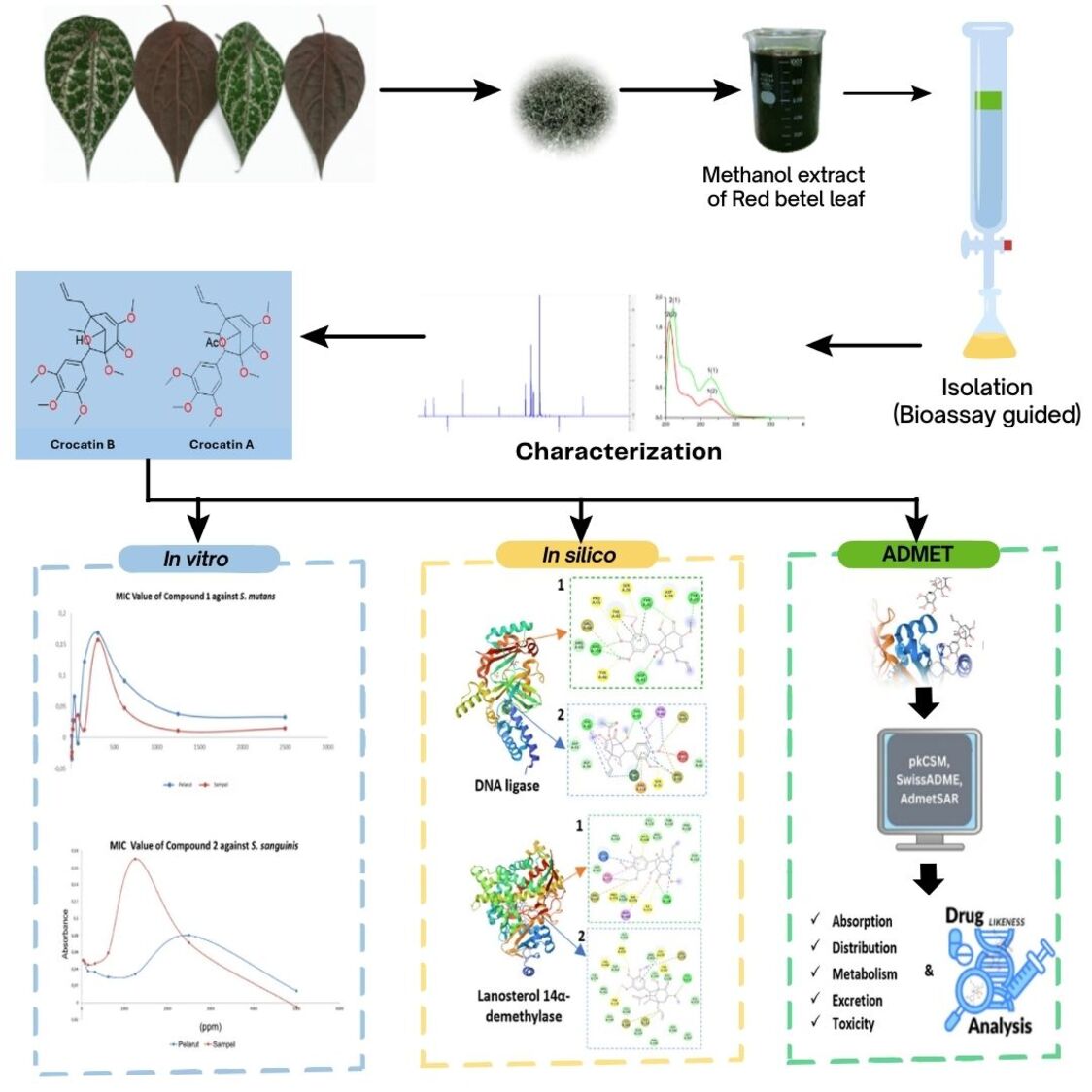
Downloads
References
N Parfati and T Windono. Red betel (Piper crocatum Ruiz & Pav) literature review. Media Pharmaceutica Indonesiana 2016; 1(2), 106-115.
D Arbain, Nofrizal, N Syafni, F Ismed, S Yousuf and MI Choudhary. Bicyclo[3.2.1]octanoid neolignans from Indonesian red betle leaves (Piper crocatum Ruiz & Pav.). Phytochemistry Letters 2017; 24, 163-166.
YJ Chai YJ Chai, Y Go, HQ Zhou, HX Li, SJ Lee, YJ Park, W Widowatib, R Rizal, YH Kim, SY Yang and W Li. Unusual bicyclo[3.2.1]octanoid neolignans from leaves of Piper crocatum and their effect on pyruvate dehydrogenase activity. Plants 2021; 10(9), 1855.
FO Enwa. Mechanisms of antimicrobial actions of phytochemicals against enteric pathogens - A Review. Journal of Pharmaceutical Biological and Chemical Sciences 2014; 2(2), 77-85.
Emrizal, A Fernando, R Yuliandari, K Rullah, NR Indrayani, A Susanty, R Yerti, F Ahmad, HM Sirat and D Arbain. Cytotoxic activities of fractions and two isolated compounds from Sirih Merah (Indonesian red betel), Piper crocatum Ruiz & Pav. Procedia Chemistry 2014; 13, 79-84.
D Kurnia, S Lestari, T Mayanti, M Gartika and D Nurdin. Anti-infection of oral microorganisms from herbal medicine of Piper crocatum Ruiz & Pav. Drug Design, Development and Therapy 2024; 18, 2531-2553.
S Lestari, D Kurnia, T Mayanti and L Heliawati. Antimicrobial activities of stigmasterol from Piper crocatum in vitro and in Silico. Journal of Chemistry 2024; 2024(1), 2935516.
N Singburaudom. Hydroxychavicol from Piper betel leave is an antifungal activity against plant pathogenic fungi. Journal of Biopesticides 2015; 8(2), 82-92.
T Siswina, MM Rustama, D Sumiarsa, E Apriyanti, H Dohi and D Kurnia. Antifungal constituents of Piper crocatum and their activities study using ADMET and drug-likeness Analysis. Molecules 2023; 28(23), 7705.
E Eijk, B Wittekoek, EJ Kuijper and WK Smits. DNA replication proteins as potential targets for antimicrobials in drug-resistant bacterial pathogens. The Journal of Antimicrobial Chemotherapy 2017; 72(5), 1275-1284.
K Bilotti, V Potapov, JM Pryor, AT Duckworth, JL Keck and GJS Lohman. Mismatch discrimination and sequence bias during end-joining by DNA ligases. Nucleic Acids Research 2022; 50(8), 4647-4658.
KP Ha and AM Edwards. DNA Repair in Staphylococcus aureus. Microbiology and Molecular Biology Reviews: MMBR 2021; 85(4), e0009121.
WD Nes, M Chaudhuri and DJ Leaver. Druggable sterol metabolizing enzymes in infectious diseases: Cell targets to therapeutic leads. Biomolecules 2024; 14(3), 249.
T Siswina, MM Rustama, D Sumiarsa and D Kurnia. Phytochemical profiling of Piper crocatum and its antifungal activity as Lanosterol 14 alpha demethylase CYP51 inhibitor: A review. F1000Research 2023; 11, 1115.
KS Gajiwala and C Pinko. Structural rearrangement accompanying NAD+synthesis within a bacterial DNA ligase crystal. Structure 2004; 12(8), 1449-1459.
MV Keniya, M Sabherwal, RK Wilson, MA Woods, AA Sagatova, JDA Tyndall and BC Monk. Crystal structures of full-length lanosterol 14α-Demethylases of prominent fungal pathogens candida albicans and candida glabrata provide tools for antifungal discovery. Antimicrobial Agents and Chemotherapy 2018; 62(11), e01134-18.
D Kurnia, SA Putri, SG Tumilaar, A Zainuddin, HDA Dharsono and MF Amin. In silico study of antiviral activity of polyphenol compounds from Ocimum basilicum by molecular docking, ADMET and drug-likeness analysis. Advances and Applications in Bioinformatics and Chemistry : AABC 2023; 16, 37-47.
NV Wendersteyt, DS Wewengkang and SS Abdullah. Uji aktivitas antimikroba dari ekstrak dan fraksi ascidian herdmania momus dari perairan pulau bangka likupang terhadap pertumbuhan mikroba Staphylococcus aureus, Salmonella typhimurium dan Candida albicans. Pharmacon 2021; 10(1), 706-712.
MF Yahaya, JM Yelwa, S Abdullahi, JB Umar, AM Abubakar and M Babakura. Chemical compositions, FTIR and larvicidal activity of essential oils extracted from aromatic plants. European Scientific Journal 2019; 15(24), 110.
KC Lin, B Muthiah, HP Chang, T Kasai and YP Chang. Halogen-related photodissociation in atmosphere: Characterisation of atomic halogen, molecular halogen and hydrogen halide. International Reviews in Physical Chemistry 2020; 40(1), 1-50.
TI Purwantiningsih, W Haumein and J Presson. Air rebusan daun sirih sebagai antibakteri alami untuk mencegah mastitis. Jurnal Ilmu dan Teknologi Peternakan Tropis 2020; 7(3), 252.
AS Ravipati, N Reddy and SR Koyyalamudi. Biologically active compounds from the genus Uncaria (Rubiaceae). Studies in Natural Products Chemistry 2014; 43, 381-408.
CM Mouzié, MGF Guefack, BY Kianfé, HU Serondo, BK Ponou, X Siwe-Noundou, RB Teponno, RWM Krause, V Kuete and LA Tapondjou. A new chalcone and antimicrobial chemical constituents of dracaena stedneuri. Pharmaceuticals 2022; 15(6), 725.
J Leszczynski, A Kaczmarek-Kedziera, T Puzyn, MG Papadopoulos, H Reis and MK Shukla. Handbook of computational chemistry. Springer, Cham, Switzerland.
Y Guttman and Z Kerem. Computer-aided (In Silico) modeling of cytochrome P450-Mediated food-drug interactions (FDI). International Journal of Molecular Sciences 2022; 23(15), 8498.
F İslamoğlu and E Hacıfazlıoğlu. Investigation of the usability of some triazole derivative compounds as drug active ingredients by ADME and molecular docking properties. Moroccan Journal of Chemistry 2022; 10(4), 861-880.
M Jeleń, B Morak-Młodawska, M Dołowy and A Konefał. Study of the Lipophilicity of Tetracyclic Anticancer Azaphenothiazines. Biomolecules 2025; 15(8), 1194.
N Pavlović, NM Sopta, D Mitrović, D Zaklan, AT Petrović, N Stilinović and S Vukmirović. Principal component analysis (PCA) of molecular descriptors for improving permeation through the Blood–Brain barrier of quercetin analogues. International Journal of Molecular Sciences 2024; 25(1), 192.
D Mitrović and N Pavlović. In silico approach in the development of structural analogues of resveratrol with improved distribution in the central nervous system. Kragujevac Journal of Science 2024; 46(1), 59-71.
H Sardar. Drug like potential of Daidzein using SwissADME prediction: In silico approaches. PHYTONutrients 2023; 2(1), 02-08.
CA Lipinski. Lead- and drug-like compounds: The rule-of-five revolution. Drug Discovery Today: Technologies 2004; 1(4), 337-341.
F Cotinguiba, HM Debonsi, RV Silva, RM Pioli, RA Pinto, LG Felippe, SN López, MJ Kato and M Furlan. Amino acids L-phenylalanine and L-lysine involvement in trans and cis piperamides biosynthesis in two Piper species. Brazilian Journal of Biology 2023; 82, e268505.
JE Poulton and VS Butt. Purification and properties of S-adenosyl-l-methionine: Caffeic acid o-methyltransferase from leaves of spinach Beet (Beta vulgaris L.). Biochimica et Biophysica Acta (BBA) – Enzymology 1975; 403(2), 301-314.
JM Humphreys, MR Hemm and C Chapple. New routes for lignin biosynthesis defined by biochemical characterization of recombinant ferulate 5-hydroxylase, a multifunctional cytochrome P450-dependent monooxygenase. Proceedings of the National Academy of Sciences of the United States of America 1999; 96(18), 10045-10050.
C Paniagua, A Bilkova, P Jackson, S Dabravolski, W Riber, V Didi, J Houser, N Gigli-Bisceglia, M Wimmerova, E Budínská, T Hamann and J Hejatko. Dirigent proteins in plants: Modulating cell wall metabolism during abiotic and biotic stress exposure. Journal of Experimental Botany 2017; 68(13), 3287-3301.
HMA El-Lateef, MM Khalaf, M Kandeel, AA Amer, AA Abdelhamid and A Abdou. Designing, characterization, biological, DFT and molecular docking analysis for new FeAZD, NiAZD and CuAZD complexes incorporating 1-(2-hydroxyphenylazo)−2-naphthol (H2AZD). Computational Biology and Chemistry 2023; 105, 107908.
S Sato, M Dacher and H Kurumizaka. Nucleosome structures built from highly divergent histones: Parasites and giant DNA viruses. Epigenomes 2022; 6(3), 22.
P Augustijns, B Wuyts, B Hens, P Annaert, J Butler and J Brouwers. A review of drug solubility in human intestinal fluids: Implications for the prediction of oral absorption. European Journal of Pharmaceutical Sciences 2014; 57, 322-332.
G Falcón-Cano, C Molina and MÁ Cabrera-Pérez. ADME prediction with KNIME: In silico aqueous solubility consensus model based on supervised recursive random forest approaches. ADMET & DMPK 2020; 8(3), 251-273.
MF Khan, N Nahar, RB Rashid, A Chowdhury and MA Rashid. Computational investigations of physicochemical, pharmacokinetic, toxicological properties and molecular docking of betulinic acid, a constituent of Corypha taliera (Roxb.) with Phospholipase A2 (PLA2). BMC Complementary and Alternative Medicine 2018; 18(1), 48.
XL Ma, C Chen and J Yang. Predictive model of blood-brain barrier penetration of organic compounds. Acta Pharmacologica Sinica 2005; 26(4), 500-512.
VM Alves, EN Muratov, A Zakharov, NN Muratov, CH Andrade and A Tropsha. Chemical toxicity prediction for major classes of industrial chemicals: Is it possible to develop universal models covering cosmetics, drugs and pesticides. Food and Chemical Toxicology 2018; 112, 526-534.
AA Toropov, AP Toropova, I Raska, D Leszczynska and J Leszczynski. Comprehension of drug toxicity: Software and databases. Computers in Biology and Medicine 2014; 45(1), 20-25.
MN Drwal, P Banerjee, M Dunkel, MR Wettig and R Preissner. ProTox: A web server for the in silico prediction of rodent oral toxicity. Nucleic Acids Research 2014; 42(W1), W53-W58.
LZ Benet, CM Hosey, O Ursu and TI Oprea. BDDCS, the Rule of 5 and drugability. Advanced Drug Delivery Reviews 2016; 101, 89-98.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






