Morphological and Molecular Characterization of Trichotylenchus dispersus (Nematoda: Dolichodoridae), a Newly Recorded Plant-Parasitic Nematode in the Rhizosphere Soil of Tomato Plants in Thailand
DOI:
https://doi.org/10.48048/tis.2024.8796Keywords:
Morphometrics, Molecular characterization, Phylogenetic trees, Plant-parasitic nematodesAbstract
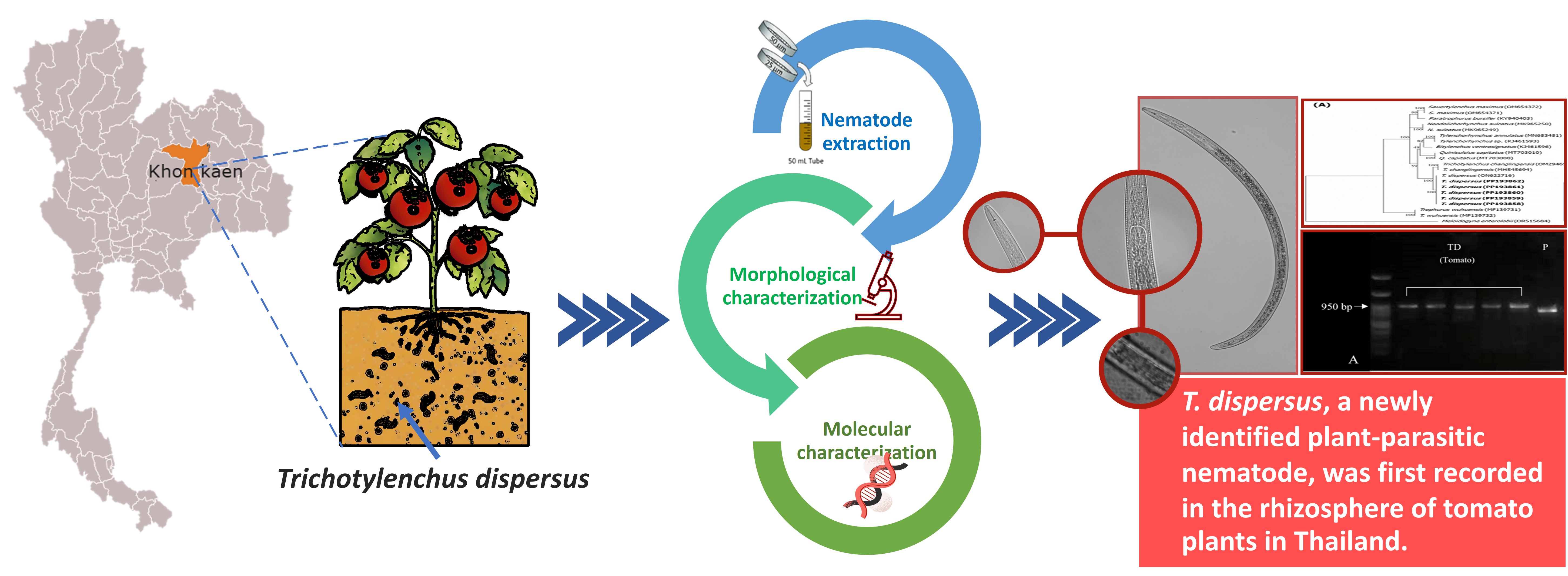
Trichotylenchus sp. is a migratory ectoparasitic nematode (PPN) that causes hypoplasia disease symptoms in economic plants, such as maize and banana. In this study, soil samples were collected from 6 locations in a tomato field in Khon Kaen Province, Thailand, for the purpose of nematode extraction using the Cobb’s sieving and flotation-centrifugation techniques. Morphological and molecular characterization of the collected PPN identified it as Trichotylenchus sp., a PPN not previously found in Thai tomato fields. Morphologically, the nematode has a C-shaped body, a rounded lip region, robust stylet and dorsal gland orifice located 2.0 ± 0.5 µm from the base of basal knobs. The tail is conically shaped, and the cuticle exhibits annulations with 3 incisures in the lateral field. The morphological characteristics observed closely matched Trichotylenchus sp. To confirm the identity, diagnosis was done using Polymerase Chain Reaction (PCR) molecular technique. Nematode DNA amplification was conducted with different target genes using primer sets AB28/TW81 for ITS1-5.8S-ITS2 and D2A/D3B for D2-D3 region of the 28S rRNA. The PCR amplification showed DNA fragments of approximately 950 and 800 bp, respectively. The sequences obtained were compared with those in the NCBI GenBank, which showed a 98.0 - 99.4 % identity match with Trichotylenchus dispersus specimens previously documented in China. In addition, the phylogenetic trees reiterated a nematode grouping with T. dispersus, 100 % bootstrap value. To the best of our knowledge, this is the first report of the presence of T. dispersus in the soils of tomato field in Thailand. This nematode is likely to become a major factor limiting tomato production yields if not properly managed.
HIGHLIGHTS
- Six plant-parasitic nematodes (PPNs) were found to be associated with the tomato gown in field.
- A newly recorded PPN in the genus, Trichotylenchus, found in Thailand, has been documented in this study.
- Morphological and molecular characterization identified PPN as Trichotylenchus disperses.
- This is the first report on incidence of dispersus in a tomato field in Thailand.
GRAPHICAL ABSTRACT
Downloads
References
C Akotowanou, E Adjou, A Olubi, DS Kougblenou, E Ahoussi and D Sohounhloué. The tomato (Solanum lycopersicum L.) in community development: An overview focused on nutritional properties, agronomic constraints, recent achievements and future prospective. Int. J. Front. Biol. Pharm. Res. 2022; 3, 8-16.
Office of Agricultural Economics, Available at: https://mis-app.oae.go.th/service/api, accessed June 2024.
T Arie, H Takahashi, M Kodama and T Teraoka. Tomato as a model plant for plant-pathogen interactions. Plant Biotechnol. 2007; 24, 135-47.
A Seid, C Fininsa, T Mengistu, W Decraemer and W Wesemael. Tomato (Solanum lycopersicum) and root-knot nematodes (Meloidogyne spp.) - a century-old battle. Nematology 2015; 17, 995-1009.
S Sumokaryo. The role of plant parasitic nematodes on productivity reduction of banana and tomato in East Kalimantan, Indonesia. Asian J. Agric. 2017; 1, 40-5.
JW Noling, Available at: https://edis.ifas.ufl.edu/publication/NG032, accessed June 2024.
A Sachan, RK Pandey, S Kumar, N Kashyap and MS Sengar. Survey and distribution of plant parasitic nematodes on tomato at Baruasagar in Jhansi (India). Am. J. Agric. Biol. Sci. 2023; 18, 48-60.
European and Mediterranean Plant Protection Organization, Available at: https://www.eppo.int/MEETINGS/2010_meetings/p_nematodes_ch, accessed June 2024.
N Beesa, A Sasnarukkit, K Jindapunnapat, B Chinnasri and T Chairin. Incidence and characterization of rice root nematodes, Hirschmanniella mucronata, from rice fields in Pathum Thani province, Thailand. Trends Sci. 2021; 18, 486.
P Nimnoi, P Pirankham, K Srimuang and P Ruanpanun. Insights into soil nematode diversity and bacterial community of Thai jasmine rice rhizosphere from different paddy fields in Thailand. PeerJ 2024; 12, e17289.
S Keereewan. Distribution of plant parasitic nematodes on soybean in Thailand. Thai Agric. Res. J. 1988; 6, 40-6.
P Phadungkit, N Beesa, K Jindapunnapat, S Chowpongpang, B Chinnasri and A Sasnarukkit. Morphological and molecular characterization of predominant plant-parasitic nematodes associated with pattawia pineapple-cultivated areas in Prachuap Khiri Khan, Thailand. Trends Sci. 2024; 21, 8017.
N Beesa, A Suwanngam, K Puttawong, P Phanbut, K Jindapunnapat, A Sasnarukkit and B Chinnasri. First report of the root-knot nematode Meloidogyne graminicola on shallot (Allium cepa var. aggregatum) in Thailand. New Dis. Rep. 2023; 47, e12158.
K Jindapunnapat, B Chinnasri, N Beesa and N Chomphuphuang. Molecular phylogeny and morphological studies reveal a 30-year-old rain tree (Samanea saman) maintains populations of Meloidogyne enterolobii, a new host plant in Thailand. J. Phytopathol. 2023; 171, 409-20.
C Boonrin, N Beesa, K Jindapunnapat, A Sasnarukkit, PJ Chen and B Chinnasri. Morphological and molecular identification of Meloidogyne enterolobii populations from different chili-cultivated areas in Ubon Ratchathani province, Thailand. Trends Sci. 2024; 21, 7816.
J Tangkam, N Beesa, A Suwanngam, K Puttawong, P Phanbut, T Korsrisakul, A Sasnarukkit and B Chinnasri. First report of Meloidogyne enterolobii infecting mulberry cv. Chiang Mai 80 (Morus alba) in Thailand. New Dis. Rep. 2024; 50, e12296.
JT Jones, A Haegeman, EG Danchin, HS Gaur, J Helder, MG Jones, T Kikuchi, R Manzanilla-López, JE Palomares-Rius, WM Wesemael and RN Perry. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013; 14, 946-61.
S Bua-art, W Saksirirat, S Kanokmedhakul, A Hiransalee and R Lekphrom. Extraction of bioactive compounds from luminescent mushroom (Neonothopanus nambi) and its effect on root-knot nematode (Meloidogyne incognita). KKU Res. J. 2010; 15, 726-37.
S Saenkhot, A Hiransalee1 and P Thummabenjapone. Species identification of root-knot nematode (Meloidogyne) in cultivated areas of northeast Thailand. Khon Kaen Agric. J. 2012; 40, 250-8.
J Han, AL Colgrove, ND Bowman, NE Schroeder and NM Kleczewski. A survey of plant-parasitic nematodes in Illinois corn fields, 2018 and 2020. Plant Health Prog. 2021; 22, 560-4.
A Chen, Y Hu, Y Wang, Y Mao, D Zhao and F Pan. First report of Trichotylenchus changlingensis infecting maize in Shaanxi, China. Plant Dis. 2023; 107, 587.
Y Chen, Y Han, H Wang, H Xie and C Xu. Redescription and molecular characterization of Trichotylenchus dispersus (Nematoda: Dolichodoridae) in China. Zootaxa 2023; 5231, 552-66.
M Camacho, M Mota, A Lima, L Rusinque and ML Inacio. Extraction of potato cyst nematodes from soil samples: Cobb’s decanting and sieving method vs. Fenwick’s method. Rev. Ciênc. Agrár. 2018; 41, 21-30.
JL Sarah and M Boisseau. Nematode extraction from banana roots by the centrifugal-flotation technique. Fruits 2008; 63, 249-51.
JW Seinhorst. Three new Pratylenchus species with a description of the structure of cephalic framework and of the spermatheca in this genus. Nematologica 1968; 14, 497-510.
A Ryss. A simple express technique to process nematodes for collection slide mounts. J. Nematol. 2017; 49, 27-32.
G Thorne. On the classification of the Tylenchida, new order (Nematode, Phasmidia). Proc. Helminthol. Soc. Wash. 1919; 16, 37-73.
M Holterman, AVD Wurff, HV Megen, T Bongers, O Holovachov, J Bakker and J Helder. Phylum-wide analysis of SSU rDNA reveals deep phyologentic relationships among nematodes and accelerated evolution toward crown clades. Mol. Biol. Evol. 2006; 23, 1792-800.
N Beesa, A Sasnarukkit, K Jindapunnapat, F Tivet, S Bellafiore and B Chinnasri. Species characterization and population dynamics of Hirschmanniella mucronata in lowland rice fields managed under conservation agriculture in Cambodia. J. Saudi Soc. Agric. Sci. 2021; 20, 137-45.
SA Subbotin, D Sturhan, VN Chizhov, N Vovlas and JG Baldwin. Phylogenetic analysis of Tylenchida thorne, 1949 as inferred from D2 and D3 expansion fragments of the 28S rRNA gene sequences. Nematology 2006; 8, 455-74.
GB Nunn. 1992, Nematode molecular evolution. Ph. D. Dissertation. University of Nottingham, Nottingham, United Kingdom.
E Shokoohi and P Masoko. Association of plant-parasitic nematodes and soil physicochemical properties in tomatoes in Turfloop, Limpopo Province, South Africa. Horticulturae 2024; 10, 328.
K Osei, MK Osei, MB Mochiah, JNL Lamptey, G Bolfrey-Arku and JN Berchie. Plant parasitic nematodes associated with tomato in Ghana. Nematol. Mediterr. 2012; 40, 33-7.
A Silva, G Oliveira, R Pastoriza, E Maranhão, E Pedrosa, S Maranhão, L Boiteux, J Pinheiro and J Filho. Search for sources of resistance to Meloidogyne enterolobii in commercial and wild tomatoes. Hortic. Bras. 2019; 37, 188-98.
H Karuri. Root and soil health management approaches for control of plant-parasitic nematodes in sub-Saharan Africa. Crop Protect. 2022; 152, 105841.
Z Haque and M Khan. Belonolaimidae: Belonolaimus species. CABI Digital Library, Oxfordshire, England, 2021.
M Hosseinvand, A Eskandari and R Ghaderi. Morphological and molecular characterisation of Telotylenchus tamariscus n. sp. (Nematoda: Telotylenchinae) recovered from Khuzestan, south-western Iran. Nematology 2020; 22, 1121-9.
LB Chanu, NM Meitei and MM Shah. Two new species of soil nematodes from Manipur, India. J. Parasit. Dis. 2014; 40, 674-8.
U Gozel, BJ Adams, KB Nguyen, RN Inserra, RM Giblin-Davis and LW Duncan. A phylogeny of Belonolaimus populations in Florida inferred from DNA sequences. Nematropica 2006; 36, 155-71.
M Bogale, A Baniya and P DiGennaro. Nematode identification techniques and recent advances. Plants 2020; 9, 1260.
Aİ Keçici, R Bozbuğa, A Öcal, E Yüksel, G Özer, Ş Yildiz, R Lahlali, B Slaats, AA Dababat and M İmren. Diversity and identification of plant-parasitic nematodes in wheat-growing ecosystems. Microorganisms 2022; 10, 1534.
Z Yang, H Zhang, Z Jiang, Y Wu and M Liu. Morphological and molecular characterization of Heterodera ripae, a new record cyst nematode in the rhizosphere soil of Fagopyrum esculentum. Sci. Rep. 2024; 14, 9958.
CM Holguin, DA Rojas, OY Pérez and S Marchant. First detection of the potato cyst nematode, Globodera rostochiensis, infecting potato in the central region of Colombia. Plant Dis. 2023; 107, 2889.
MY Liu, J Liu, W Huang and D Peng. First report of Meloidogyne graminicola on rice in Henan Province, China. Plant Dis. 2021; 105, 3308.
HY Wu, TJ Li, XB Zhou, L Liu and J Gu. First report of root-lesion nematode, Pratylenchus parazeae on maize (Zea mays L.) in China. Plant Dis. 2019; 104, 2889.
J Gu, M Munawar, P Castillo and B Cai. Morphological and molecular characterization of Quinisulcius curvus from China. J. Nematol. 2021; 53, e2021-e2086.
R Ghaderi, A Karegar and E Miraeiz. Morphological characterization of Trichotylenchus gorganiensis n. sp. (Nematoda: Dolichodoridae) and photomicrographs of several other species of the subfamily Telotylenchinae Siddiqi, 1960. Zootaxa 2018; 4526, 447-80.
A Saeedi, M Abdollahi, R Ghaderi and H Charehgani. Plant parasitic nematodes of the families Merliniidae, Telotylenchidae and Pratylenchidae (Nematoda: Tylenchoidea) associated with important field crops of Gorgan region. Sci. J. Agric. 2019; 41, 4.
P Myers, R Espinosa, CS Parr, T Jones, GS Hammond and TA Dewey. Browse animalia, Available at: https://animaldiversity.org, accessed June 2024.
Nemaplex, Available at: http://nemaplex.ucdavis.edu/Taxadata/Hoplidae.htm, accessed June 2024.
AG Whitehead. Tricotylenchus falciformis n.g., n. sp. (Belonolaiminae n. subfam: Tylenchida Thorn, 1949) an associate of grass roots (Hyparrhenia sp.) in southern Tanganyika. Nematologica 1959; 4, 279-85.
E Geraert. The Dolichodoridae of the world-identification of the family Dolichodridae. Academia Press, Gent, Belgium, 2011.
University of Nebraska-Lincoln, Available at: https://nematode.unl.edu/trichosp.htm, accessed June 2024.
K Azizi, A Eskandari, A Karegar, R Ghaderi, SVD Elsen, M Holterman and J Helder. Analyses of morphological and molecular characteristics of Telotylenchinae from Iran point at the validity of the genera Bitylenchus and Sauertylenchus. Zootaxa 2022; 5169, 425-46.
MR Siddiqi. Telotylenchus, a new nematode genus from North India (Tylenchida: Telotylenchinae n.sub-fam.). Nematologica 1960; 5, 73-7.
EM Onkendi, GM Kariuki, M Marais and LN Moleleki. The threat of root-knot nematodes (Meloidogyne spp.) in Africa: A review. Plant Pathol. 2014; 63, 727-37.
DL Coyne, JM Nicol and B Claudius-Cole. Practical plant nematology: A field and laboratory guide. International Institute of Tropical Agriculture (IITA), Cotonou, Benin, 2007.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






