Nematicidal Potential of Green Chiretta Extracts Against Meloidogyne enterolobii and M. incognita: In vitro Assessment for Agricultural Application
DOI:
https://doi.org/10.48048/tis.2024.8151Keywords:
Crude extracts, Green chiretta, Medicinal plants, Nematicidal activity, Root-knot nematodesAbstract
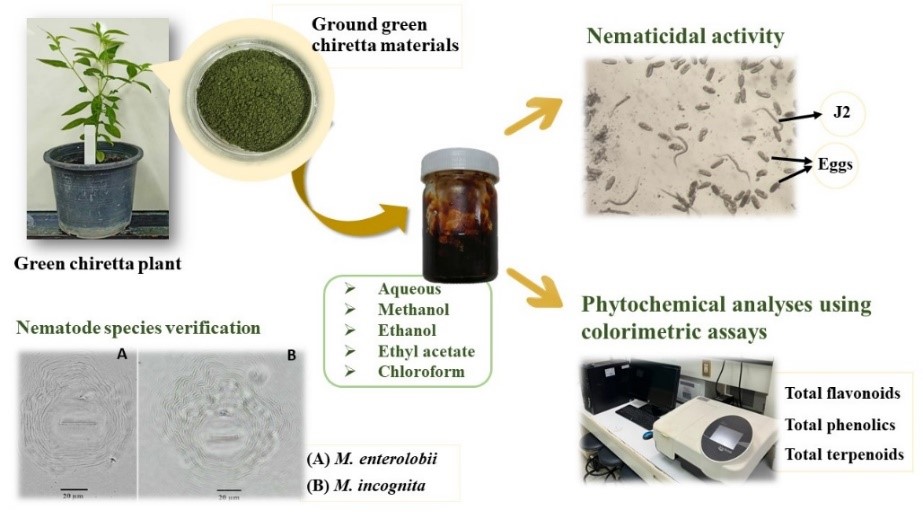
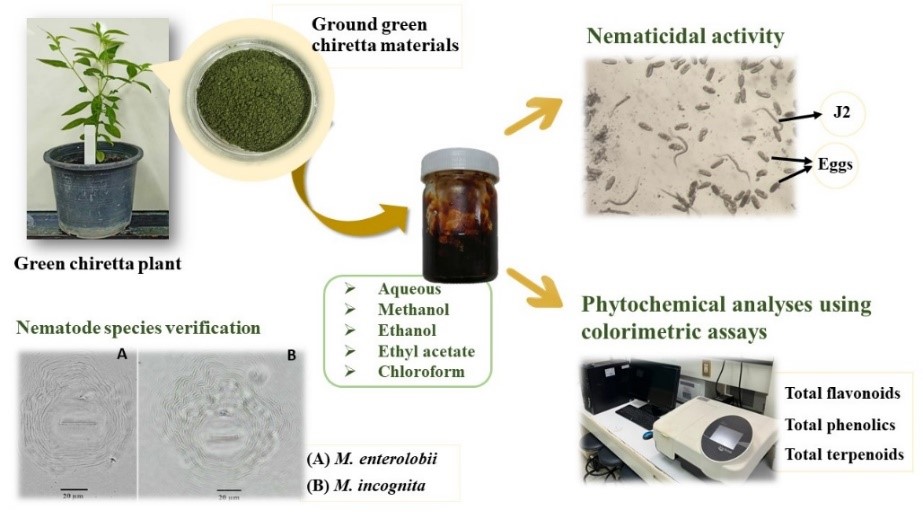
Root-knot nematodes pose a persistent threat to various economically important crops in Thailand, with limited available management strategies for farmers. This study aimed to assess the efficacy of aqueous (AE), methanolic (ME), ethanolic (EE), ethyl acetate (EAE), and chloroform (CE) crude extracts derived from green chiretta in controlling Meloidogyne enterolobii and M. incognita under laboratory conditions. The results demonstrated significant inhibitory effects of all crude extracts, except for CE, against both nematode species. Notably, AE (10 and 20 mg/mL), ME (20 mg/mL), and EE (20 mg/mL) treatments exhibited maximum inhibition of hatching, leading to a considerable reduction in nematode hatch rates by 87.5 - 96.8 % for M. enterolobii and 88.5 - 96.5 % for M. incognita, compared with distilled water (DW). In terms of second-stage juveniles (J2s) mortality, AE (10 and 20 mg/mL), ME (20 mg/mL), and EAE (20 mg/mL) demonstrated the highest nematicidal activity, resulting in mortality rates of 94.2 - 100 % for M. enterolobii and 92.2 - 99.2 % for M. incognita. Chemotaxis assays revealed a repellent effect of all extracts on both nematode species, except for the ethyl acetate extract (EAE), which attracted M. incognita J2s without affecting M. enterolobii J2s. The observed efficacy was attributed to the higher concentrations of bioactive compounds, including flavonoids, phenolics, and terpenoids, in the extracts compared to CE. Based on these in vitro findings, it is suggested that AE, ME, EE, and EAE of green chiretta exhibit significant nematicidal activity against M. enterolobii and M. incognita. Further evaluation under greenhouse and field conditions is warranted to ascertain the practical applicability and effectiveness of these extracts in a real-world agricultural setting.
HIGHLIGHTS
- This study investigates the efficacy of crude extracts from green chiretta against Meloidogyne enterolobii and incognita, common threats to Thai crops. Aqueous, methanolic, ethanolic, ethyl acetate, and chloroform extracts were evaluated for their inhibitory effects on nematode hatching and mortality, with notable efficacy at specific concentrations.
- The mortality rates of nematodes exposed to aqueous, methanolic, and ethyl acetate extracts demonstrated notable nematicidal activity in the range of 92.2 to 100 %.
- The nematode hatch rates exhibited significant nematicidal activity when exposed to aqueous, methanolic, and ethanolic extracts, with a notable range of 87.5 - 96.8 %.
- The observed efficacy is linked to higher concentrations of bioactive compounds, such as flavonoids, phenolics, and terpenoids, present in the extracts compared to chloroform.
- Aqueous, methanolic, ethanolic, and ethyl acetate extracts exhibit significant in vitro nematicidal activity against studied nematodes, showcasing potential agricultural applications.
GRAPHICAL ABSTRACT
Downloads
References
JT Jones, A Haegeman, EGJ Danchin, HS Gaur, J Helder, MGK Jones, T Kikuchi, R Manzanilla-Lopez, JE Palomares-Rius, WML Wesemael and RN Perry. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013; 14, 946-61.
JE Munyaneza and B Bizimungu. Chapter 23 - Management of potato pests and diseases in Africa. Insect Pests of Potato, Global Perspectives on Biology and Management. Copyright, Washington DC, 2022, p. 407-26.
F Gharabadiyan, S Jamali, AA Yazdi, MH Hadizadeh and A Eskandari. Weed hosts of root-knot nematodes in tomato fields. J. Plant Protect. Res. 2012; 52, 230-4.
K Qiao, X Shi, H Wang, X Ji and K Wang. Managing root-knot nematodes and weeds with 1,3-Dichloropropene as an alternative to methyl bromide in cucumber crops in China. J. Agr. Food Chem. 2011; 59, 2362-7.
SK Reddy, S Liu, JC Rudd, Q Xue, P Payton, SA Finlayson, J Mahan, A Akhunova, SV Holalu and N Lu. Physiology and transcriptomics of water-deficit stress responses in wheat cultivars TAM 111 and TAM 112. J. Plant Physiol. 2014; 171, 1289-98.
G Devi and J George. Available at: https://www.biotecharticles.com/Agriculture-Article/Nematodes-and-their-Management-in-Ginger-4085.html, accessed January 2024.
EHC Silva, RS Soares, HO Borges, CA Franco, LT Braz and PLM Soares. Quantification of the damage caused by Meloidogyne enterolobii in okra. Pesquisa Agropecuária Brasileira 2019; 54, e00050.
K Jindapunnapat, B Chinnasri, N Beesa and N Chomphuphuang. Molecular phylogeny and morphological studies reveal a 30-year-old rain tree (Samanea saman) maintains populations of Meloidogyne enterolobii, a new host plant in Thailand. J. Phytopathology 2023; 171, 409-20.
K Jindapunnapat, B Chinnasri and S Kwankuae. Biological control of root-knot nematodes (Meloidogyne enterolobii) in guava by the fungus Trichoderma harzianum. J. Dev. Sustain. Agr. 2013; 8, 110-8.
C Boonrin, N Beesa, K Jindapunnapat, A Sasnarukkit, PJ Chen and B Chinnasri. Morphological and molecular identification of Meloidogyne enterolobii populations from different chili-cultivated areas in Ubon Ratchathani province, Thailand. Trends Sci. 2024; 21, 7816.
N Tangchitsomkid. Management of gall disease in chili. Ministry of Agriculture and Cooperatives, Bangkok, Thailand 2007.
DL Coyne, HH Fourie and M Moens. Current and future management strategies in resource-poor regions. In: RN Perry, M Moens and JL Starr (Eds.). Root-knot nematodes. Wallingford, UK: CABI Publishing, 2009, p. 444-75.
N Viaene, DL Coyne and KG Davies. Biological and cultural management. Plant nematology. CAB International, Wallingford, 2013, p. 383-410.
N Beesa, B Chinnasri, A Sasnarukkit, T Dethoup, K Jindapunnapat, AW Kiriga, K Saikai, S Haukeland and D Coyne. Potential of cavalcade, Centrosema pascuorum, leaves for controlling Meloidogyne javanica and quantification of active phytochemicals. Nematology 2023; 25, 1019-32.
M Ismail, S Fayyaz, A Kowsar, S Javed, I Ali, S Ali, F Hussian and H Ali. Evaluation of nematocidal effects of some medicinal plant extracts against root-knot nematodes (Meloidogyne incognita). Ital. J. Agron. 2020; 15, 1475.
H Kesba, A Abdel-Rahman, S Sayed and A Al-Sayed. Screening the nematicidal potential of indigenous medicinal plant extracts against Meloidogyne incognita under lab and greenhouse conditions. Egypt. J. Biol. Pest Contr. 2021; 31, 81.
A Zareen, MJ Zaki and N Javed. Nematicidal activity of ginger and its effect on the efficacy of Pasteuria penetrans for the control of root knot nematodes on tomato. Asian J. Plant Sci. 2003; 2, 858-60.
SK Mishra, NS Sangwan and RS Sangwan. Plant review Andrographis paniculata (Kalmegh): A review. Phcog. Rev. 2007; 1, 283-98.
MS Hossain, Z Urbi, A Sule and KMH Rahman. Andrographis paniculata (Burm. f.) Wall. ex Nees: A review of ethnobotany, phytochemistry, and pharmacology. Sci. World J. 2014; 2014, 274905.
SR Naik and A Hule. Evaluation of immunomodulatory activity of an extract of andrographolides from Andographis paniculata. Planta Med. 2009; 75, 785-91.
M Nisha, S Anusree and S Sooraj. Efficacy of Andrographis paniculata (Burm.f.) Nees against root-knot nematode in pepper, Piper nigrum L. J. Entomol. Zool Stud. 2019; 7, 539-45.
M Tigano, KD Siqueira, P Castagnone-Sereno, K Mulet, P Queiroz, MD Santos, C Teixeira, M Almeida, J Silva and R Carneiro. Genetic diversity of the root-knot nematode Meloidogyne enterolobii and development of a scar marker for this guava damaging species. Plant Pathol. 2010; 59, 1054-61.
O Randig, M Bongiovanni, RMDG Carneiro and P Castagnone-Sereno. Genetic diversity of root-knot nematodes from Brazil and development of scar markers specific for the coffee-damaging species. Genome 2002; 45, 862-70.
N Beesa, A Sasnarukkit, K Jindapunnapat, B Chinnasri and T Chairin. Incidence and characterization of rice root nematodes, Hirschmanniella mucronata, from rice fields in Pathum Thani province, Thailand. Trends Sci. 2021; 18, 486.
M Talavera-Rubia, MD Vela-Delgado and S Verdejo-Lucas. Nematicidal efficacy of milbemectin against root-knot nematodes. Plants 2020; 9, 839.
M Catalkaya and Z Devran. Integrated management of Mi-1 virulent Meloidogyne incognita (Kofoid & White, 1919) Chitwood, 1949 (Tylenchida: Meloidogynidae) in greenhouse tomatoes. Turk. J. Entomol. 2019; 43, 157-69.
A Banji, B Goodluck, O Oluchi. and F Stephen. Antimicrobial and antioxidant activities of crude methanol extract and fractions of Andrographis paniculata leaf (Family: Acanthaceae) (Burm. f.) Wall. Ex Nees. Jordan J. Biol. Sci. 2018; 11, 23-30.
AC Kumoro, M Hasan and H Singh. Effects of solvent properties on the soxhlet extraction of diterpenoid lactones from Andrographis paniculata leaves. ScienceAsia 2009; 35, 306-9.
LG. Wade. Organic chemistry. Prentice-Hall, New York, 1987.
T Banerjee, A Singh, S Kumar, T Dhanani, NA Gajbhiye, TK Koley, A Maurya and J Filgona. Ovicidal and larvicidal effects of extracts from leaves of Andrographis paniculata (Burm. f.) Wall.ex Nees against field isolates of human hookworm (Ancylostoma duodenale). J. Ethnopharmacol. 2019; 235, 489-500.
W Chao and B Lin. Isolation and identification of bioactive compounds in Andrographis paniculata (Chuanxinlian). Chin. Med. 2010; 5, 17.
J Chen and B Song. Natural nematicidal active compounds: recent research progress and outlook. J. Integr. Agr. 2021; 20, 2015-31.
DJ Chitwood. Nematicidal compounds from plants. Phytochemical Resources for medicine and agriculture. Plenum Press, New York, 1992, 185-204.
N Beesa, K Jindapunnapat, B Chinnasri and T Chairin. Nematode development and changes in enzymatic defensive activity in rice plants upon Meloidogyne graminicola infection for preliminary screening of resistant cultivars. Songklanakarin J. Sci. Tech. 2022; 44, 26-31.
K Jindapunnapat, ND Reetz, MH MacDonald, G Bhagavathy, B Chinnasri, N Soonthornchareonnon, A Sasnarukkit, KR Chauhan, DJ Chitwood and SLF Meyer. Activity of vetiver extracts and essential oil against Meloidogyne incognita. J. Nematol. 2018; 50, 147-62.
R Petrikovszki, F Toth and PI Nagy. Aqueous extracts of organic mulch materials have nematicide and repellent effect on Meloidogyne incognita infective juveniles: a laboratory study. J. Nematol. 2023; 55, 20230037.
C Sukkanon, T Karpkird, M Saeung, T Leepasert, A Panthawong, W Suwonkerd, MJ Bangs and T Chareonviriyaphap. Excito-repellency activity of Andrographis paniculata (Lamiales: Acanthaceae) against colonized mosquitoes. J. Med. Entomol. 2020; 57, 192-203.
M Govindarajan and R Sivakumar. Adulticidal and repellent properties of indigenous plant extracts against Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). Parasitol. Res. 2012; 110, 1607-20.
SP Adiguna, JA Panggabean, RT Swasono, SI Rahmawati, F Izzati, A Bayu, MY Putra, C Formisano and C Giuseppina. Evaluations of andrographolide-rich fractions of Andrographis paniculata with enhanced potential antioxidant, anticancer, antihypertensive, and anti-inflammatory activities. Plants 2023; 12, 1220.
S Nagajothi, P Mekala, A Raja, MJ Raja and P Senthilkumar. Andrographis paniculata: Qualitative and quantitative phytochemical analysis. J. Phcog. Phytochemistry 2018; 7, 1251-3.
S Sinha and R Raghuwanshi. Evaluation of phytochemical, antioxidant and reducing activity in whole plant extract of Andrographis paniculata (Burm.f.) Wall. ex Nees. Biosci. Biotechnol. Res. Comm. 2020, http://dx.doi.org/10.21786/bbrc/13.4/15.
HT Nguyen, VM Do, TT Phan and DTN Huynh. The potential of ameliorating COVID-19 and sequelae from Andrographis paniculata via bioinformatics. Biol. Biol. Insights 2023; 17, 11779322221149622.
LA Pavun, S Uskokovic-markovic, M Jelikic-stankov, D Dikanovic and P Durdevic. Determination of flavonoids and total polyphenol contents in commercial apple juices. Czech J. Food Sci. 2018; 36, 233-8.
A Lukowski, R Jagiello, P Robakowski and D Adamczyk. Adaptation of a simple method to determine the total terpenoid content in needles of coniferous trees. Plant Sci. 2022; 314, 111090.
G Aydinl and S Mennan. Identification of root-knot nematodes (Meloidogyne spp.) from greenhouses in the middle black sea region of Turkey. Turk. J. Zool. 2016; 40, 675-85.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






