Efficacy of Potassium Silicate in Suppressing Meloidogyne enterolobii on Okra (Abelmoschus esculentus L.)
DOI:
https://doi.org/10.48048/tis.2026.11933Keywords:
Biostimulant, Nematode management, Potassium, Root-knot nematodes, Silicon, Soil-borne diseaseAbstract
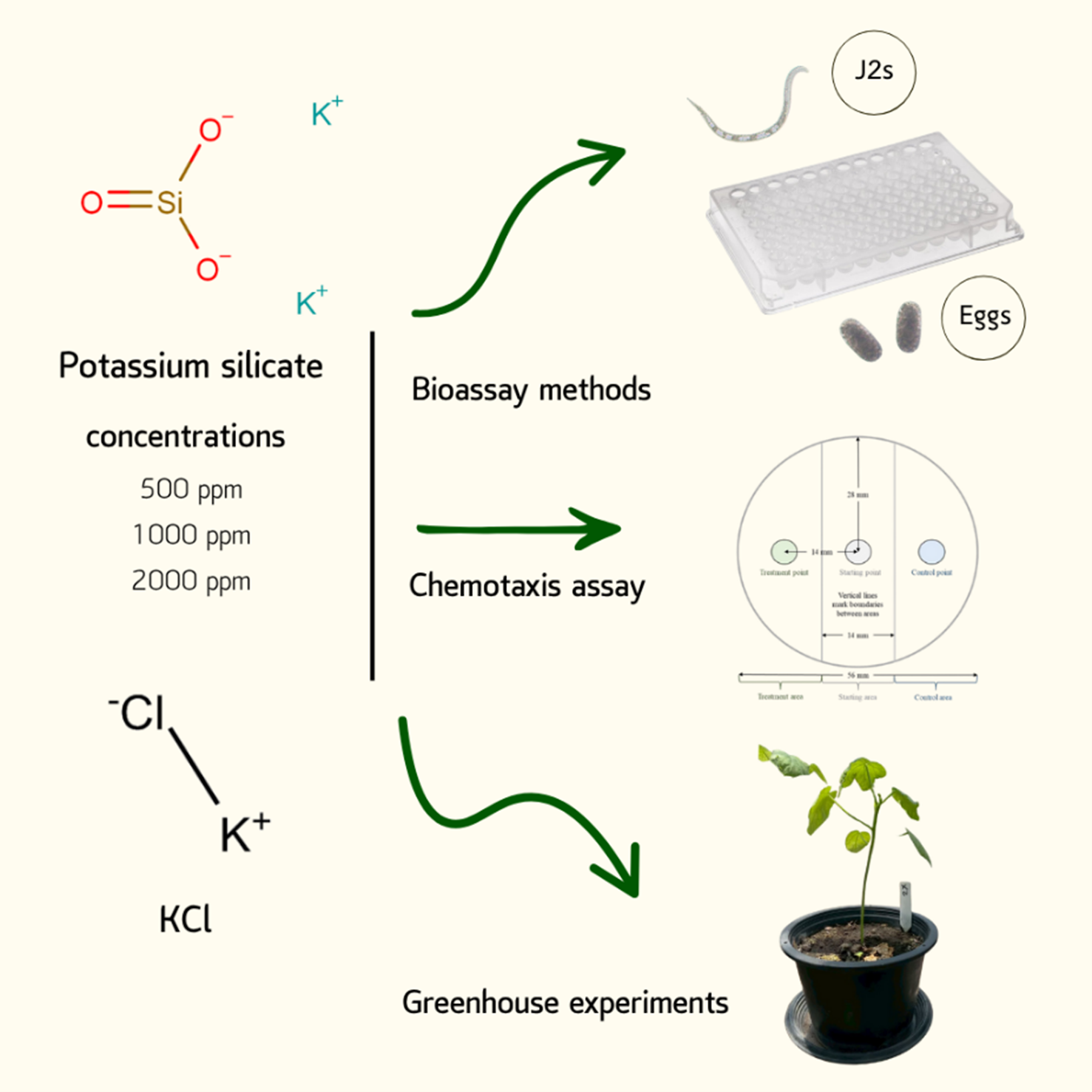
Potassium silicate (PSi) is widely recognized as a biostimulant that enhances plant growth by improving nutrient uptake, promoting root development, and stimulating vegetative growth. In addition, it has been reported to exhibit nematicidal activity against root-knot nematodes (RKN). However, information regarding its application for the control of Meloidogyne enterolobii in Thailand remains limited. Therefore, the objective of this study was to evaluate the potential of PSi in suppressing M. enterolobii under both controlled laboratory and greenhouse conditions. Second-stage juveniles (J2s) and eggs of the nematode were exposed to PSi at concentrations of 250, 500, 1,000, and 2,000 ppm, and nematode mortality and hatching were assessed after 2 and 7 days of incubation, respectively. The highest J2 mortality was observed at 2,000 ppm (56.0 ± 5.8%), showing an effect comparable to KCl (positive control). Inhibition of nematode hatching was most pronounced at 500 - 2,000 ppm, resulting in reductions of 58.4 ± 3.7% to 64.5 ± 2.8% relative to the distilled water control. Moreover, PSi concentrations above 500 ppm exhibited nematicidal activity by directly attracting J2 movement. Under greenhouse conditions, okra plants treated with 1,000 and 2,000 ppm prior to nematode inoculation showed the greatest suppression of M. enterolobii, reducing galls, egg masses, and eggs by 42.1% - 74.4%, 62.8% - 78.9%, and 54.2% - 80.6%, respectively, and lowering the nematode reproduction factor (Rf) by 3- to 4-fold compared with the RKN-inoculated control. Plant growth, particularly plant height, was also markedly enhanced. Overall, PSi application as a soil drench not only promotes plant development but also exhibits strong potential for suppressing
M. enterolobii on okra, highlighting its value as a promising component of Integrated Pest Management (IPM) strategies for sustainable RKN control.
HIGHLIGHTS
- This study was conducted to evaluate the effectiveness of potassium silicate (PSi) in controlling Meloidogyne enterolobii parasitizing okra plants under both laboratory and greenhouse conditions.
- In laboratory assays, 2,000 ppm PSi exhibited the strongest nematicidal effect on juvenile mortality, while nematode hatching was most strongly suppressed at 500 ppm. Additionally, PSi concentrations above 500 ppm attracted nematode movement.
- Greenhouse experiments consistently confirmed the nematicidal effects against enterolobii, reducing the number of galls, egg masses, and eggs, as well as Rf values, while promoting plant growth.
- PSi demonstrates considerable potential against enterolobii and could be incorporated into IPM programs to reduce the damage caused by this nematode.
GRAPHICAL ABSTRACT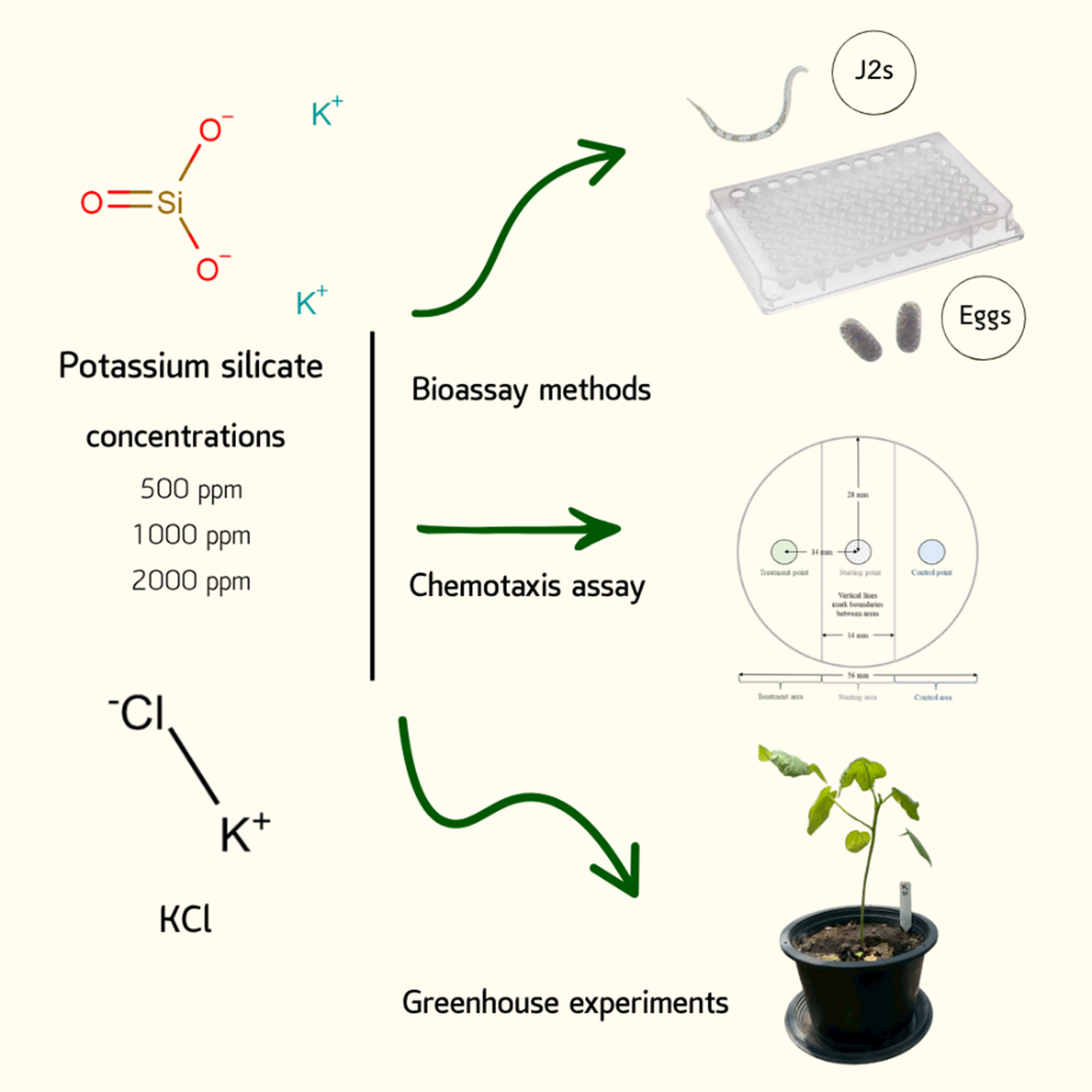
Downloads
References
F Forghani and A Hajihassani. Recent advances in the development of environmentally benign treatments to control root-knot nematodes. Frontiers in Plant Science 2020; 11, 1125.
A Afzal and T Mukhtar. Revolutionizing nematode management to achieve global food security goals: An overview. Heliyon 2024; 10(3), e25325.
K Jindapunnapat, B Chinnasri, N Beesa and N Chomphuphuang. Molecular phylogeny and morphological studies reveal a 30-year-oldrain tree (Samanea saman) maintains populations of Meloidogyne enterolobii, a new host plant in Thailand. Journal of Phytopathology 2023; 171(9), 409-420.
N Beesa, N Kuncharoen, T Dethoup, K Jindapunnapat, A Sasnarukkit and B Chinnasri. Prevalence and characterization of plant-parasitic nematodes existing in RD41 rice fields in Pathum Thani Province, Thailand, with emphasis on Hirschmanniella mucronata and Meloidogyne graminicola. Trends in Sciences 2024; 21(12), 8952.
C Boonrin, N Beesa, K Jindapunnapat, A Sasnarukkit, PJ Chen and B Chinnasri. Morphological and molecular identification of Meloidogyne enterolobii populations from different chili-cultivated areas in Ubon Ratchathani province, Thailand. Trends in Science 2024; 21(8), 7816.
J Tangkam, N Beesa, A Suwanngam, K Puttawong, P Phanbut, T Korsrisakul, A Sasnarukkit and B Chinnasri. First report of Meloidogyne enterolobii infecting mulberry cv. Chiang Mai 80 (Morus alba) in Thailand. New Disease Reports 2024; 50(1), e12296.
R Cetintas, J Brito and D Dickson. Virulence of four Florida isolates of Meloidogyne mayaguensis to selected soybean genotypes. Nematropica 2008; 38(2), 127-136.
S Kiewnick, G Karssen, J Brito, M Oggenfuss and JE Frey. First report of root-knot nematode Meloidogyne enterolobii on tomato and cucumber in Switzerland. Plant Disease 2008; 92(9), 1370.
JA Velloso, MAD Maquilan, VP Campos, JA Brito and DW Dickson. Temperature effects on development of Meloidogyne enterolobii and M. floridensis. Journal of Nematology 2022; 54(1), e20220013.
M Shi, R Liu, DUN Madhusanka, Y Liu, N Luo, W Guo, J Zhao, H Li and Z Mao. Emerging threat of Meloidogyne enterolobii: Pathogenicity mechanisms and sustainable management strategies in the context of global change. Microbiological Research 2025; 16(8), 165.
JB Pinheiro, LS Boiteux, MRA Almeida, RB Pereira, LCS Galhardo and RMDG Carneiro. First report of Meloidogyne enterolobii in capsicum rootstocks carrying the Me1 and Me3/Me7 genes in central Brazil. Nematropica 2015; 45(2), 184-188.
WB Rutter, PA Wadl, JD Mueller and P Agudelo. Identification of sweet potato germplasm resistant to pathotypically distinct isolates of Meloidogyne enterolobii from the Carolinas. Plant Disease 2021; 105(10), 3147-3153.
K Puttawong, N Beesa, S Kasem, K Jindapunnapat, B Chinnasri and A Sasnarukkit. Potential of Bacillus spp. against root-knot nematode, Meloidogyne enterolobii, parasitizing chili (Capsicum annuum L.). Crop Protection 2024; 184, 106780.
K Jindapunnapat, B Chinnasri and S Kwankuae. Biological control of root-knot nematodes (Meloidogyne enterolobii) in guava by the fungus Trichoderma harzianum. Journal of Developments in Sustainable Agriculture 2013; 8(2), 110-118.
P Sakorn and P Ruanpanun. Potential of combination of Trichoderma asperellum strain Cb-pin01 and Streptomyces sp. strain KPS-E004 in controlling M. enterolobii of chili. Journal of Agricultural Science and Management 2023; 6, 15-22.
M Phonginsee, N Beesa, A Suwanngam, K Jindapunnapat, S Chowpongpang, B Chinnasri and A Sasnarukkit. Characterization and biocontrol potential of predatory nematodes (Mononchida and Dorylaimida) against Meloidogyne enterolobii in Thailand. International Journal of Agriculture and Biosciences 2025; 14(5), 862-871.
P Phanbut, K Puttawong, A Suwanngam, N Beesa, K Jindapunnapat, A Sasnarukkit and B Chinnasri. Nematicidal potential of green chiretta extracts against Meloidogyne enterolobii and M. incognita: In vitro assessment for agricultural application. Trends in Sciences 2024; 21(10), 8151.
TS Devi, HS Behera, A Madhu, Samreen, S Chaudhary, S Koushal and P Reddypriya. A comprehensive review on integrated pest management in nematode. International Journal of Research in Agronomy 2024; 7(12), 760-765.
M Luyckx, JF Hausman, S Lutts and G Guerriero. Silicon and plants: Current knowledge and technological perspectives. Frontiers in Plant Science 2017; 8, 411.
J Zhang, J Ding, M Ibrahim, X Jiao, X Song, P Bai and J Li. Effects of the interaction between vapor-pressure deficit and potassium on the photosynthesis system of tomato seedlings under low temperature. Scientia Horticulturae 2021; 283, 110089.
A Oraee and A Tehranifar. Relationship between silicon through potassium silicate and salinity tolerance in Bellis perennis L. Silicon 2023; 15, 93-107.
SR Kumar, AC Jnanesha, S Bharathkumar, K Sravya, S Venugopal and RK Lal. Unveiling the effect of foliar applied siliceous compounds on reducing seed shattering for the conservation of endangered kalmegh (Andrographis paniculata (Burm. f.) wall. ex nees). Biocatalysis and Agricultural Biotechnology 2025; 68, 103700.
S Saher. Potassium silicate: A better approach to flight root-knot nematode (Meloidogyne incognita). Asian Journal of Emerging Research 2020; 2, 3-4.
D Khairy. Nematicidal activity of jojoba oil, potassium silicate and bio-nematon singly or integrated against Meloidogyne incognita, in vitro, and in vivo. Journal of Plant Protection and Pathology 2025; 16(1), 7-12.
AG El-Sherif, SB Gad and SM Saadoon. Impact of potassium silicate application on Meloidogyne incognita infecting cucumber plants under greenhouse conditions. Asian Journal of Nematology 2016; 5(1), 1-7.
O Filialuna, C Wram and I Zasada. What is the optimal way to assess Meloidogyne spp. reproduction in greenhouse pot experiments? Journal of Nematology 2022; 54(1), e20220012.
ACG Bicalho, SA da Silva and ZAC Machado. Control of Meloidogyne paranaensis mediated by silicon. Scientia Agricola 2021; 78(3), e20190039.
AS Ardakani. Toxicity of silver, titanium, and silicon nanoparticles on the root-knot nematode, Meloidogyne incognita, and growth parameters of tomato. Nematology 2013; 15(6), 671-677.
H Hida, H Nishiyama, S Sawa, T Higashiyama and H Arata. Chemotaxis assay of plant-parasitic nematodes on a gel-filled microchannel device. Sensors and Actuators B: Chemical 2015; 221, 1483-1491.
R Čepulytė, WB Danquah, G Bruening and VM Williamson. Potent attractant for root-knot nematodes in exudates from seedling root tips of two host species. Scientific Reports 2018; 8, 10847.
K Jindapunnapat, ND Reetz, MH MacDonald, G Bhagavathy, B Chinnasri, N Soonthornchareonnon, A Sasnarukkit, KR Chauhan, DJ Chitwood and SLF Meyer. Activity of vetiver extracts and essential oil against Meloidogyne incognita. Journal of Nematology 2018; 50(2), 147-162.
RV Silva, RDL Oliveira, KJT Nascimento and FA Rodrigues. Biochemical responses of coffee resistance against Meloidogyne exigua mediated by silicon. Plant Pathology 2010; 59, 586-593.
EG Sampaio, FA Almeida, AM Oliveira, WL Fonseca, MLT Leite and LMS Xavier. Control of Meloidogyne javanica in cowpea with silicon application. Revista Brasileira de Ciências Agrárias - Brazilian Journal of Agricultural Sciences 2022; 17(3), 1-7.
MAM El-Saedy, MEA El-Sayed and SE Hammad. Efficacy of boron, silicon, jojoba and four bio-products on controlling Meloidogyne incognita infecting Thompson seedless grapevines. American-Eurasian Journal of Agricultural & Environmental Sciences 2015; 15(9), 1710-1720.
LMP Guimaraes, EMR Pedrosa, RSB Coelho, EF Couto, SRVL Maranhao and A Chaves. Efficiency and enzymatic activity elicited by methyl jasmonate and potassium silicate on sugarcane under Meloidogyne incognita parasitism. Summa Phytopathologica 2010; 36, 11-15.
A Soffan, SS Alghamdi and AS Aldawood. Peroxidase and polyphenol oxidase activity in moderate resistant and susceptible Vicia faba induced by Aphis craccivora (Hemiptera: Aphididae) infestation. Journal of Insect Science 2014; 14, 285.
SF Afzali, H Sadeghi and A Taban. A comprehensive model for predicting the development of defense system of Capparis spinosa L.: A novel approach to assess the physiological indices. Scientific Reports 2023; 13(1), 12413.
LP Zhan, DL Peng, XL Wang, LA Kong, H Peng, SM Liu, Y Liu and WK Huang. Priming effect of root-applied silicon on the enhancement of induced resistance to the root-knot nematode Meloidogyne graminicola in rice. BMC Plant Biology 2018; 18(1), 50.
LB Santos, JPS Júnior, RM Prado, RF Júnior, VF Souza, MMS Sarah and PLM Soares. Silicon allows halving cadusafos dose to control Meloidogyne incognita and increase cotton development. Silicon 2022; 14, 3809-3816.
EHC Silva, RS Soares, HO Borges, CA Franco, LT Braz and PLM Soares. Quantification of the damage caused by Meloidogyne enterolobii in okra. Pesquisa Agropecuária Brasileira 2019; 54, e00050.
M Wang, L Gao, S Dong, Y Sun, Q Shen and S Guo. Role of silicon on plant-pathogen interactions. Frontiers in Plant Science 2017; 8, 701.
B Chinnasri, N Beesa, A Suwanngam, K Puttawong, T Korsrisakul, P Phanbut, K Jindapunnapat and A Sasnarukkit. Efficacy of fosthiazate (Nemathorin 10% GR) in managing Meloidogyne enterolobii in chili crop in Thailand. Crop Protection 2026; 199, 107425.
KK Verma, XP Song, DD Tian, DJ Guo, ZL Chen, CS Zhong, A Nikpay, M Singh, VD Rajput, RK Singh, T Minika and YR Li. Influence of silicon on biocontrol strategies to manage biotic stress for crop protection, performance, and improvement. Plants 2021; 10, 2163.
VJ Connick. 2011, The impact of silicon fertilisation on the chemical ecology of grapevine, Vitis vinifera; constitutive and induced chemical defences against arthropod pests and their natural enemies. Master’s Thesis. Charles Sturt University, New South Wales, Australia.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






