Morphological and Molecular Characterization of Predominant Plant-Parasitic Nematodes Associated with Pattawia Pineapple-Cultivated Areas in Prachuap Khiri Khan, Thailand
DOI:
https://doi.org/10.48048/tis.2024.8017Keywords:
Plant-parasitic nematodes, Pineapple, Rotylenchulus reniformis, Helicotylenchus dihysteraAbstract
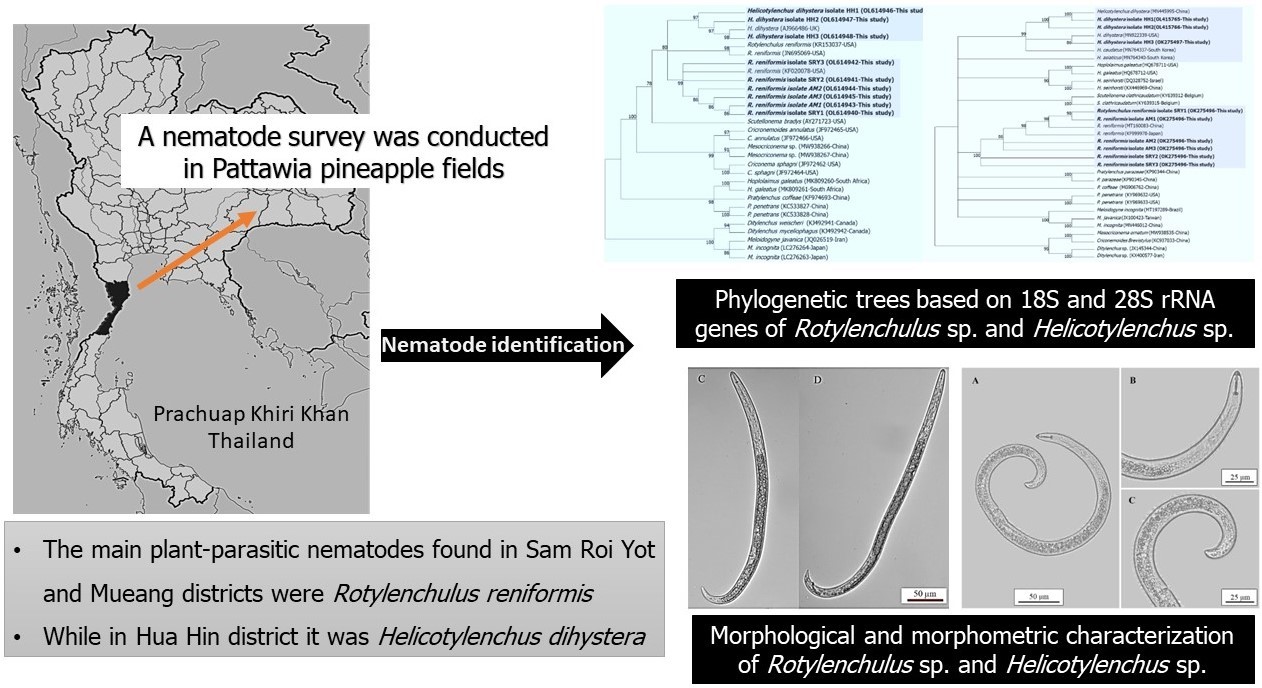
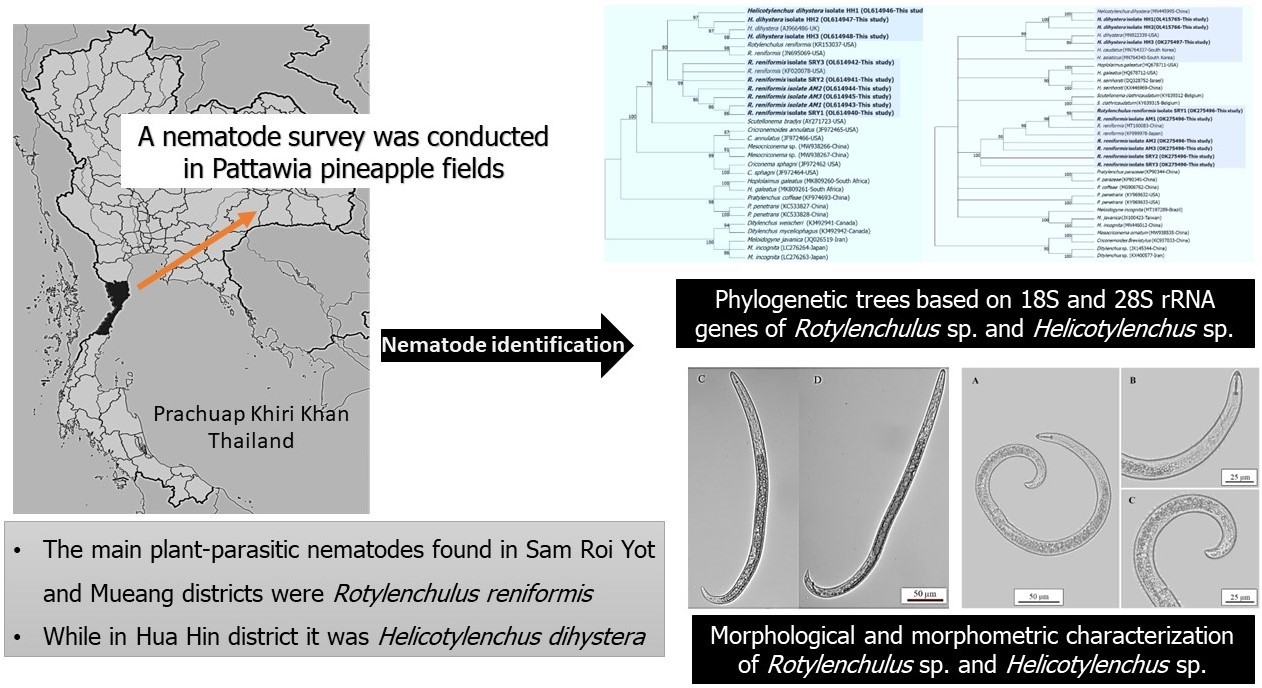
The predominant plant-parasitic nematodes associated with Pattawia pineapple-cultivated areas were investigated using combined molecular and morphological techniques. In total, 134 soil samples were collected for nematode extraction from the main pineapple crops in 3 districts (Sam Roi Yot, Mueang and Hua Hin) of Prachuap Khiri Khan, Thailand. Based on the morphology, 6 plant-parasitic nematode genera were identified: Rotylenchulus sp., Meloidogyne sp., Helicotylenchus sp., Hoplolaimus sp., Mesocriconema sp. and Pratylenchus sp. The main plant-parasitic nematodes that occurred in Sam Roi Yot and Mueang districts were Rotylenchulus sp. (70.27 and 97.01 %, respectively), while in Hua Hin district, Helicotylenchus sp. (37.73 %) was dominant. Based on molecular identification, nucleotide sequence comparisons and the phylogenetic tree analyses based on the 18S and 28S rRNA genes revealed the species of the nematodes were R. reniformis and H. dihystera, showing 99 % identity with the nematode populations from Japan and China, respectively. Notably, the morphometric and morphological characters also identified these 2 species correctly; however, this technique required more time and may be less accurate. While R. reniformis has been identified as a common species in pineapple fields in the USA, India, Kenya, the Philippines and Nigeria, the current study was the 1st to report H. dihystera isolated from soil in Pattawia pineapple crops in Thailand.
HIGHLIGHTS
- Six plant-parasitic nematode genera were associated with Pattawia pineapple fields in Prachuap Khiri Khan, Thailand.
- The main plant-parasitic nematodes in Sam Roi Yot and Mueang districts were Rotylenchulus reniformis, while in Hua Hin district it was Helicotylenchus dihystera.
- This is the 1st description on the molecular characterization of R. reniformis and H. dihystera from pineapple fields in Thailand.
GRAPHICAL ABSTRACT
Downloads
References
Office of Agricultural Economics, Available at: https://www.oae.go.th/assets/portals/1/
fileups/prcaidata/files/Pineapple%2065.pdf, accessed January 2024.
PP Joy and G Sindhu, Available at: http: //www.kau.in/sites/default/files/documents/diseases
_of_pineapple.pdf, accessed January 2023.
EC Rabie. Nematode pests of pineapple. In: H Fourie, V Spaull, R Jones, M Daneel, DD Waele (eds.). Nematology in South Africa: A View from the 21st Century. Springer, Cham, 2017.
B Chinnasri and B Sipes. Effect of a systemic acquired resistance inducer on nematodes infecting pineapple. Acta Hortic. 2005; 666, 213-22.
KG Rohrbach and WJ Apt. Nematode and disease problems of pineapple. Plant Dis. 1986; 70, 81-7.
B Chinnasri, BS Sipes and DP Schmitt. Effects of inducers of systemic acquired resistance on reproduction of Meloidogyne javanica and Rotylenchulus reniformis in pineapple. J. Nematol. 2006; 38, 319-25.
G Devi. Community analysis of plant-parasitic nematodes in pineapple ecosystem in Meghalaya. Indian J. Nematol. 2007; 37, 106-7.
AW Kiriga. 2018, Occurrence of plant parasitic nematodes in commercial pineapple fields and effect of biocontrol agents on Meloidogyne species in Kenya. Master Thesis. Kenyatta University, Kenya.
F Daramola and S Afolami. Studies on the distribution of plant-parasitic nematodes associated with pineapple in Delta, Imo and Cross River states of Nigeria. Aust. J. Basic Appl. Sci. 2014; 8, 248-56.
NC Benzonan, LCS Dalisay, KCC Reponte, CP Mapanao, LV Alvarez, AO Rendon and LY Zurbano. Plant-parasitic nematodes associated with pineapple (Ananas comosus) in selected provinces in Luzon, Philippines. Eur. J. Mol. Clin. Med. 2021; 8, 945-57.
AW Kiriga, D Coyne, JG Atandi, NV Beek and S Haukeland. Diversity and population densities of plant-parasitic nematodes in commercial and smallholder pineapple production in Kenya. Nematology 2021; 23, 837-50.
Norwegian Institute of Bioeconomy Research, Available at: https://phys.org/news/2021-05-parasitic-nematodes-pineapple-crop-yields.html, accessed January 2023.
S Saipetara. 1976, Studies on taxonomy of pineapple nematodes in Chonburi. Master Thesis. Kasetsart University, Bangkok, Thailand.
RMDG Carneiro, FSO Lima and VR Correia. Methods and tools currently used for the identification of plant parasitic nematodes. In: MM Shah and M Mahamood (Eds.). Nematology - Concepts, diagnosis and control. IntechOpen Limited, London, 2017, p. 20-35.
P Castagnone-Sereno. Meloidogyne enterolobii (M. mayaguensis): Profile of an emerging, highly pathogenic, root-knot nematode species. Nematology 2012; 14, 133-8.
EVD Berg, SA Subbotin, ZA Handoo and LR Tiedt. Hirschmanniella kwazuna sp. n. from South Africa with notes on a new record of H. spinicaudata (Schuurmans Stekhoven, 1944) Luc and Goodey, 1964 (Nematoda: Pratylenchidae) and on the molecular phylogeny of Hirschmanniella Luc and Goodey, 1964. Nematology 2009; 11, 523-40.
O Douda, M Zouhar, E Nováková, J Mazáková and P Ryšánek. Variability of D2/D3 segment sequences of several populations and pathotypes of potato cyst nematodes (Globodera rostochiensis, Globodera pallida). Plant Protect. Sci. 2010; 46, 171-80.
ST Nyaku, RV Kantety, E Cebert, KS Lawrence, JO Honger and GC Sharma. Principal component analysis and molecular characterization of reniform nematode populations in Alabama. Plant Pathol. J. 2016; 32, 123-35.
D Riascos-Ortiz, AT Mosquera-Espinosa, FVD Agudelo, CMGD Oliveira and JE Muñoz-Florez. Morpho-molecular characterization of Colombian and Brazilian populations of Rotylenchulus associated with Musa spp. J. Nematol. 2019; 51, e2019-47.
N Beesa, A Sasnarukkit, K Jindapunnapat, B Chinnasri and T Chairin. Incidence and characterization of rice root nematodes, Hirschmanniella mucronata, from rice fields in Pathum Thani province, Thailand. Trends Sci. 2021; 18, 486.
JR Christie and VG Perry. Removing nematodes from soil. Proc. Helminthological Soc. Wash. 1951; 18, 106-8.
M Holterman, AVD Wurff, SVD Elsen, HV Megan, T Bonger, O Holovachov, J Bakker and J Helder. Phylum-wide analysis of SSU rDNA reveals deep phylogenetic relationships among nematodes and accelerated evolution toward crown clades. Mol. Biol. Evol. 2006; 23, 1792-800.
A Suwanngam. 2015, Diversity of predatory nematodes in agricultural area in Southern part of Thailand. Master Thesis. Kasetsart University, Bangkok, Thailand.
N Beesa, A Sasnarukkit, K Jindapunnapat, F Tivet, S Bellafiore and B Chinnasri. Species characterization and population dynamics of Hirschmanniella mucronata in lowland rice fields managed under conservation agriculture in Cambodia. J. Saudi Soc. Agr. Sci. 2021; 20, 137-45.
MF Mai and HH Lyon. Pictorial key to genera of plant-parasitic nematodes. Cornell University Press, London, 1975.
E Abebe, W Traunspurger and I Andrássy. Freshwater nematodes: Ecology and taxonomy. CABI, Wallingford, 2006.
T Mekete, A Dababat, N Sekora, F Akyazi and E Abebe. Identification key for agriculturally important plant-parasitic nematodes: Prepared for the International Nematode Diagnosis and Identification Course 2012 - A manual for nematology. International Maize and Wheat Improvement Center, Texcoco, Mexico, 2012.
G Thorne. On the classification of the Tylenchida, new order (Nematode, Phasmidia). Proc. Helminthological Soc. Wash. 1949; 16, 37-73.
C Chunram. A list of plant parasitic nematodes in Thailand. White Lotus Press, Bangkok, Thailand, 1972.
BS Sipes and DP Schmitt. Rotylenchulus reniformis damage thresholds on pineapple. Acta Horticulturae 2000; 529, 239-46.
JT Jones, A Haegeman, EGJ Danchin, HS Gaur, J Helder, MGK Jones, T Kikuchi, R Manzanilla-López, JE Palomares-Rius, WML Wesemael and RN Perry. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013; 14, 946-61.
CP Peacock. Studies on root-knot nematodes of the genus Meloidogyne in the gold coast. Nematologica 1957; 2, 114-22.
RG Davide. Nematode problems affecting agriculture in the Philippines. J. Nematol. 1988; 20, 214-18.
M Luc, RA Sikora and J Bridge. Plant parasitic nematodes in subtropical and tropical agriculture. CAB International, Wallingford, 2005.
HR Wallace. Abiotic influence in the soil environment. In: BM Zuckerman, WF Mai and RA Rohde (Eds.). Plant parasitic nematodes. Academic Press Inc., New York, 1964, p. 257-80.
NC Burns. Soil pH effects on nematode populations associated with soybeans. J. Nematol. 1971; 3, 238-45.
PR Singh, G Karssen, K Gitau, C Wanjau, M Couvreur, NN Pili, G Gheysen and W Bert. Rotylenchus wimbii n. sp. (Nematoda: Hoplolaimidae) associated with finger millet in Kenya. J. Nematol. 2021; 53, e2021-16.
P Agudelo, RT Robbins, JM Stewart and AL Szalanski. Intraspecific variability of Rotylenchulus reniformis from cotton-growing regions in the United States. J. Nematol. 2005; 37, 105-14.
F Zhang, Y Wang, X Zhan, D Dai, G Guo, S Guo, M Sun and J Zhang. First report of Rotylenchulus reniformis on tomato in Henan, China. Plant Dis. 2019; 103, 1044.
E Brucher, EE Vuletic, FA Guerra, RLD Rossi, MC Plazas, GD Guerra, MG Molina, MM Gabriela and ME Doucet. Characterization of a population of Helicotylenchus dihystera (Cobb, 1893) Sher, 1961, parasitizing maize roots, in southern Córdoba, Argentina. Nematropica 2019; 49, 49-58.
D Riascos-Ortiz, AT Mosquera-Espinosa, FVD Agudelo, CMGD Oliveira and JE Muñoz-Florez. An integrative approach to the study of Helicotylenchus (Nematoda: Hoplolaimidae) Colombian and Brazilian populations associated with Musa crop. J. Nematol. 2020; 52, e2020-54.
S So, Y Garan, K Miyahara and Y Ohshima. Body size change in various nematodes depending on bacterial food, sex and growth temperature. Worm 2012; 1, 93-7.
DL Coyne, JM Nicol and B Claudius-Cole. Practical plant nematology: A field and laboratory guide. International Institute of Tropical Agriculture, Oyo State, Nigeria, 2007.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






