Assessment of Solvent Extraction using Sonication to Recover Tryptophan from Kappaphycus alvarezii (Doty) Doty ex Silva: Experimental and Modelling
DOI:
https://doi.org/10.48048/tis.2024.8197Keywords:
COSMOS-RS, Multilevel factorial design, Ultrasound-assisted extraction, Polarity, Affinity, Ethanol, HPLC-DADAbstract
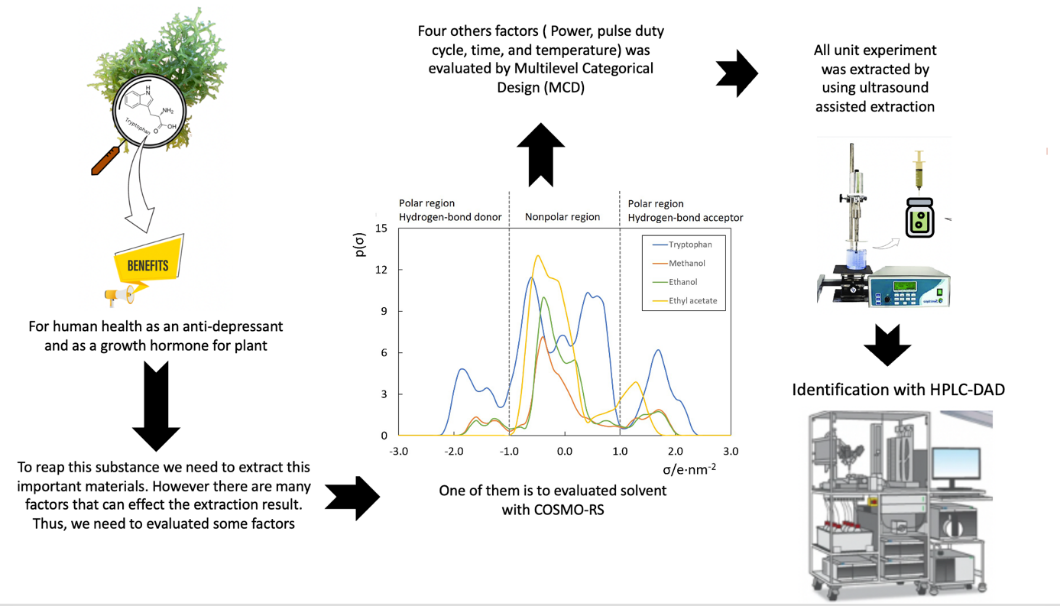
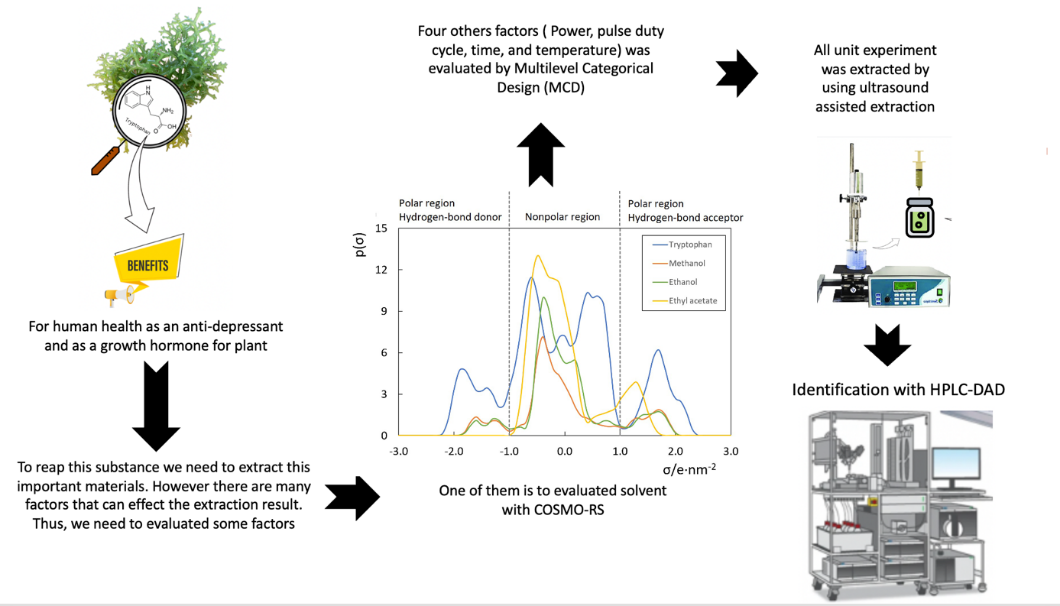
The application of Kappaphycus alvarezii in the food system has attracted researchers due to its bioactive compounds, including tryptophan. The present study was conducted to extract tryptophan from K. alvarezii with the aid of sonication. A reduced multilevel factorial design was conducted to evaluate the effect of solvent, ultrasound power, duty-pulse cycle, time and temperature on the recovery of tryptophan. Analysis of variance suggested that the type of solvent and combination setting of power and duty-pulse cycle significantly influenced the extraction (p < 0.05). In contrast, extraction time and temperature did not alter the extraction (p > 0.05). The optimum was using ethanol, ultrasound power 80 %, pulse duty-cycle 0.8 s–1, extraction time 10 min, and temperature 25 °C and tryptophan concentration was 56.41 ± 2.42 mg L–1. Subsequently, a COnducto-like Screening MOdel for Real Solvent (COSMO-RS) was performed to clarify the impact of solvent affinity and polarity on the tryptophan extraction from K. alvarezii.
HIGHLIGHTS
- Ultrasound-assisted extraction and HPLC-DAD were used to determine tryptophan levels in alvarezii.
- Box-Behnken design, in conjunction with the response surface methodology, effectively optimized the extraction conditions.
- COSMO-RS successfully predicted the solubility of tryptophan extracted from alvarezii using different solvents.
GRAPHICAL ABSTRACT
Downloads
References
Y Astor, G Engineerin, S Program, D Wisayantono, CZ Science and S Hendriatiningsih. Integration construction of marine utilization elements towards Indonesia good ocean governance in marine cadastre perspective. Indonesia J. Geospatial 2015; 4, 1-16.
Erdiwansyah, R Mamat, MSM Sani and K Sudhakar. Renewable energy in Southeast Asia: Policies and recommendations. Sci. Total Environ. 2019; 670, 1095-102.
Maharany F, Nurjanah, Suwandi R, Anwar E, Hidayat T. Bioactive compounds of seaweed Padina australis and Eucheuma cottonii as sunscreen raw materials (in Indonesian). Jurnal Pengolahan Hasil Perikanan Indonesia 2017; 20, 10-7.
F Ferdouse, SL Holdt, R Smith, P Murúa and Z Yang. The global status of seaweed production, trade and utilization. FAO Globefish Research Programme, Rome, Italy, 2018.
L Wang and CL Weller. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Tech. 2006; 17, 300-12.
G Suantika, ML Situmorang, A Khakim, I Wibowo, P Aditiawati, S Suryanarayan, SS Nori, S Kumar and F Putri. Effect of red seaweed Kappaphycus alvarezii on growth, survival, and disease resistance of pacific white shrimp Litopenaeus vannamei against Vibrio harveyi in the nursery phase. J. Aquaculture Res. Dev. 2018; 9, 1000523.
P Rajasulochana, P Krishnamoorthy and R Dhamotharan. Amino acids, fatty acids and minerals in Kappaphycus sps. ARPN J. Agr. Biol. Sci. 2010; 5, 1-12.
J Jamaluddin, YF Kum and A Khumaidi. Comparative study on the amino acid profile of Eucheuma. Tadulako Sci. Tech. J. 2019; 1, 1-11.
L Huang, JE Hogewind-Schoonenboom, L Zhu, JVS Kraaijenga, NPCV Haren, GJ Voortman, H Schierbeek, JWR Twisk, Y Huang, C Chen and JBV Goudoever. Tryptophan requirement of the enterally fed term infant in the first month of life. J. Pediatr. Gastroenterol. Nutr. 2014; 59, 374-9.
C Amaya-Guerra, SOS Saldívar and MG Alanis-Guzman. Soyabean fortification and enrichment of regular and quality protein maize tortillas affects brain development and maze performance of rats. Br. J. Nutr. 2006; 96, 161-8.
G Lindseth, B Helland and J Caspers. The effects of dietary tryptophan on affective disorders. Arch. Psychiatr. Nurs. 2016; 29, 102-7.
S Nikolaus, B Schulte, N Al-Massad, F Thieme, DM Schulte, J Bethge, A Rehman, F Tran, K Aden, R Häsler, N Moll, G Schütze, MJ Schwarz, GH Waetzig, P Rosenstiel, M Krawczak, S Szymczak and S Schreiber. Increased tryptophan metabolism is associated with activity of inflammatory bowel diseases. Gastroenterology 2017; 153, 1504-1516.e2.
L Steenbergen, BJ Jongkees, R Sellaro and LS Colzato. Tryptophan supplementation modulates social behavior: A review. Neurosci. Biobehav. Rev. 2016; 64, 346-58.
EJ Park and WY Lee. Tryptophan enhanced accumulation of phenolic compounds via chorismate mutase activation in the Ganoderma neo-japonicum mycelia. J. Kor. Soc. Appl. Biol. Chem. 2010; 53, 364-70.
S Catena, N Rakotomanomana, P Zunin, R Boggia, F Turrini and F Chemat. Solubility study and intensification of extraction of phenolic and anthocyanin compounds from Oryza sativa L. ‘Violet Nori.’ Ultrason. Sonochem. 2020; 68, 105231.
QW Zhang, LG Lin and WC Ye. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018; 13, 20.
K Duarte, CIL Justino, R Pereira, AC Freitas, AM Gomes, AC Duarte and TAP Rocha-Santos. Green analytical methodologies for the discovery of bioactive compounds from marine sources. Trends Environ. Anal. Chem. 2014; 3, 43-52.
J Cotas, A Leandro, P Monteiro, D Pacheco, A Figueirinha, AMM Goncąlves, GJD Silva and L Pereira. Seaweed phenolics: From extraction to applications. Mar. Drugs 2020; 18, 384.
W Setyaningsih, IE Saputro, M Palma and CG Barroso. Optimization of the ultrasound-assisted extraction of tryptophan and its derivatives from rice (Oryza sativa) grains through a response surface methodology. J. Cereal Sci. 2017; 75, 192-7.
D Gao, VL Ba, R Rustam, CW Cho, SY Yang, XD Su, YH Kim and JS Kang. Isolation of bioactive components with soluble epoxide hydrolase inhibitory activity from Stachys sieboldii MiQ. by ultrasonic-assisted extraction optimized using response surface methodology. Prep. Biochem. Biotechnol. 2021; 51, 395-404.
F Chemat, N Rombaut, AG Sicaire, A Meullemiestre, AS Fabiano-Tixier and M Abert-Vian. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017; 34, 540-60.
SU Kadam, BK Tiwari and CP O’Donnell. Application of novel extraction technologies for bioactives from marine algae. J. Agr. Food Chem. 2013; 61, 4667-75.
AM Cikoš, S Jokić, D Šubarić and I Jerković. Overview on the application of modern methods for the extraction of bioactive compounds from marine macroalgae. Mar. Drugs 2018; 16, 348.
Z Piñeiro, RF Guerrero, MI Fernández-Marin, E Cantos-Villar and M Palma. Ultrasound-assisted extraction of stilbenoids from grape stems. J. Agr. Food Chem. 2013; 61, 12549-56.
OK Topuz, N Gokoglu, P Yerlikaya, I Ucak and B Gumus. Optimization of antioxidant activity and phenolic compound extraction conditions from red seaweed (Laurencia obtuse). J. Aquat. Food Prod. Tech. 2016; 25, 414-22.
A Tomšik, B Pavlić, J Vladić, M Ramić, J Brindza and S Vidović. Optimization of ultrasound-assisted extraction of bioactive compounds from wild garlic (Allium ursinum L.). Ultrason. Sonochem. 2016; 29, 502-11.
Y Jin, CY Wang, W Hu, Y Huang, ML Xu, H Wang, X Kong, Y Chen, TT Don, Q Qin and KWK Tsim. An optimization of ultra-sonication- 62 assisted extraction from flowers of Apocynum venetum in targeting to amount of free amino acids determined by UPLC-MS/MS. Food Qual. Saf. 2019; 3, 52-60.
M Oroian, F Ursachi and F Dranca. Ultrasound-assisted extraction of polyphenols from crude pollen. Antioxidants 2020; 9, 322.
EM Garcia-Castello, AD Rodriguez-Lopez, L Mayor, R Ballesteros, C Conidi and A Cassano. Optimization of conventional and ultrasound assisted extraction of flavonoids from grapefruit (Citrus paradisi L.) solid wastes. LWT Food Sci. Tech. 2015; 64, 1114-22.
O Horovitz and RD Paşca. Classification of amino acids by multivariate data analysis, based on thermodynamic and structural characteristics. Studia UBB Chemia 2017; 62, 19-31.
DO Abranches, M Larriba, LP Silva, M Melle-Franco, JF Palomar, SP Pinho and JAP Coutinho. Using COSMO-RS to design choline chloride pharmaceutical eutectic solvents. Fluid Phase Equilibria 2019; 497, 71-8.
A Klamt. The COSMO and COSMO-RS solvation models. WIREs Comput. Mol. Sci. 2018; 8, e1338.
A Klamt, F Eckert and W Arlt. COSMO-RS: An alternative to simulation for calculating thermodynamic properties of liquid mixtures. Annu. Rev. Chem. Biomol. Eng. 2010; 1, 101-22.
AG Sicaire, MA Vian, F Fine, P Carré, S Tostain and F Chemat. Experimental approach versus COSMO-RS assisted solvent screening for predicting the solubility of rapeseed oil. Oilseeds Fats Crops Lipids 2015; 22, D404.
Z Guo, BM Lue, K Thomasen, AS Meyer and X Xu. Predictions of flavonoid solubility in ionic liquids by COSMO-RS: Experimental verification, structural elucidation, and solvation characterization. Green Chem. 2007; 9, 1362-73.
CU Mussagy, C Valéria, Santos-Ebinuma, KA Kurnia, CRV Dias, P Carvalho, AP Joaõ, Coutinho, F Jorge, B Pereira. Integrative platform for the selective recovery of intracellular carotenoids and lipids from Rhodotorula glutinis CCT-2186 yeast using mixtures of bio-based solvents. Green chem. 2020; 22; 8478- 8494.
GV Subbaraju, S Kannababu, K Vijayakumar, PBS Murthy, M Vanisree and HS Tsay. Spectrophotometric estimation of L-5-hydroxytryptophan in Griffonia simplicifolia extracts and dosage forms. Int. J. Appl. Sci. Eng. 2005; 3, 111-6.
SH Oh, J Ahn, DH Kang and HY Lee. The effect of ultrasonificated extracts of Spirulina maxima on the anticancer activity. Mar. Biotechnol. 2011; 13, 205-14.
W Setyaningsih, IE Saputro, CA Carrera, M Palma and CG Barroso. Multiresponse optimization of a UPLC method for the simultaneous determination of tryptophan and 15 tryptophan-derived compounds using a Box-Behnken design with a desirability function. Food Chem. 2017; 225, 1-9.
FD Vargas, AR Jiménez, OP López and F Francis. Natural pigments: Carotenoids, anthocyanins, and betalains--characteristics, biosynthesis, processing, and stability. J. Crit. Rev. Food Sci. Nutr. 2000; 40, 173-289.
G Adriano, C Ferreira, R Martins and A Quintas. Evaluating polar and nonpolar solvents extraction efficiency of gunpowder components by FTIR. Ann. Med. 2019; 51, 181.
N Medina-Torres, T Ayora-Talavera, H Espinosa-Andrews, A Sánchez-Contreras and N Pacheco. Ultrasound assisted extraction for the recovery of phenolic compounds from vegetable sources. Agronomy 2017; 7, 47.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






