Decreasing of Magnetic Saturation of Yttrium Doped Cobalt Ferrite Prepared by the Sol-Gel Auto-Combustion
DOI:
https://doi.org/10.48048/tis.2024.7320Keywords:
Sol-gel auto-combustion, Yttrium-doped, Cobalt ferrite, Annealing, Saturation magnetization, Photocatalytic, Congo redAbstract
In this research, a nano-sized cobalt ferrite material doped with Yttrium has been successfully fabricated using the sol-gel method of automatic combustion with variations in low sintering temperatures of 200, 300 and 400 °C. The results of refinement of XRD data using the Rietveld method show that the Yttrium-doped cobalt ferrite compound with a Yttrium concentration molarity of 0.1 possess a cubic crystal system and Fd-3m space group. The increasing annealing temperatures also increase the crystallite size of Yttrium-doped cobalt ferrite with the highest magnitude of 16.05 nm. The FTIR results of the samples indicated the presence of Co-O bonds around wave number 385 cm−1 and Fe-O bonds around wave number 582 cm−1 which are characteristic of the presence of cobalt ferrite compounds. From the VSM measurement results, it can be seen that there is a decrease in magnetic saturation with an increase in annealing temperature. The presence of Yttrium substitution, which takes the place of Fe3+ in cobalt ferrite material, indicates lower saturation magnetization. Image from SEM results showed samples have nanoparticle crystallite size. Evaluation of potential photocatalyst applications using a UV-Visible Spectrophotometer (UV-Vis). For Yttrium doped cobalt ferrite at 200 °C, the best degradation efficiency of Congo Red findings showed a magnitude of 76.10 %; the results are confirmed by the occurrence of the smallest crystallite size (15.11 nm).
HIGHLIGHTS
- Preparation of yttrium-doped cobalt ferrite by the sol-gel auto-combustion method with various annealing temperatures
- The presence of Yttrium substitution, which takes the place of Fe3+ in cobalt ferrite material, indicates lower saturation magnetization
- Yttrium-doped cobalt is applied for photocatalytic Congo Red dye in water with the best performance according to the smallest crystallite size
GRAPHICAL ABSTRACT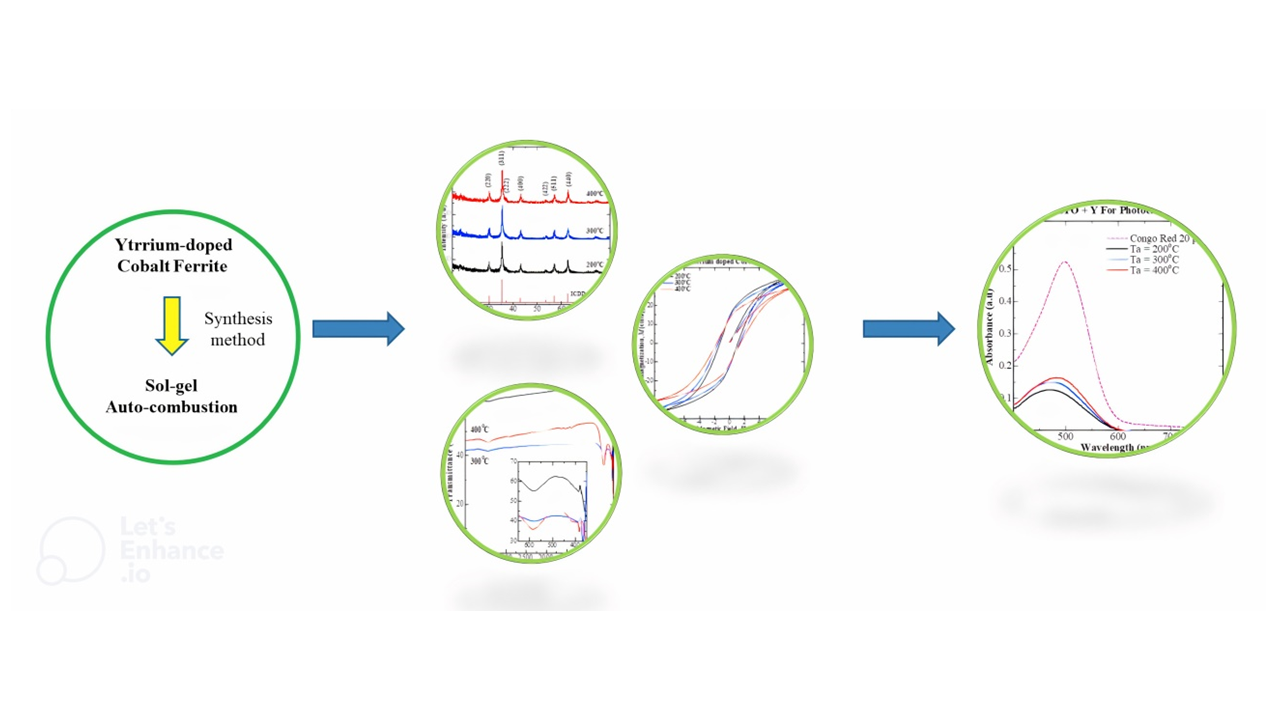
Downloads
References
KM Srinivasamurthy, VJ Angadi, SP Kubrin, S Matteppanavar, DA Sarychev, PM Kumar, HW Azale and B Rudraswamy. Tuning of ferrimagnetic nature and hyperfine interaction of Ni2+ doped cobalt ferrite nanoparticles for power transformer applications. Ceram. Int. 2018; 44, 9194-203.
PD Prasad and J Hemalatha. Enhanced magnetic properties of highly crystalline cobalt ferrite fibres and their application as gas sensors. J. Magn. Magn. Mater. 2019; 484, 225-33.
S Joshi, VB Kamble, M Kumar, AM Umarji and G Srivastava. Nickel substitution induced effects on gas sensing properties of cobalt ferrite nanoparticles. J. Alloy. Comp. 2016; 654, 460-6.
N Gill, AL Sharma, V Gupta, M Tomar, OP Pandey and DP Singh. Enhanced microwave absorption and suppressed reflection of polypyrrole-cobalt ferrite-graphene nanocomposite in X-band. J. Alloy. Comp. 2019; 797, 1190-7.
MD Ali, A Aslam, T Zeeshan, R Mubaraka, SA Bukhari, M Shoaib, M Amami, I ben Farhat, S ben Ahmed, J Abdelhak and S Waseem. Robust effectiveness behavior of synthesized cobalt doped Prussian blue graphene oxide ferrite against EMI shielding. Inorg. Chem. Comm. 2022; 137, 109204.
VS Kumbhar, AD Jagadale, NM Shinde and CD Lokhande. Chemical synthesis of spinel cobalt ferrite (CoFe2O4) nano-flakes for supercapacitor application. Appl. Surf. Sci. 2012; 259, 39-43.
M Ghanbari, F Davar and AE Shalan. Effect of rosemary extract on the microstructure, phase evolution, and magnetic behavior of cobalt ferrite nanoparticles and its application on anti-cancer drug delivery. Ceram. Int. 2021; 47, 9409-17.
Z Shi, Y Zeng, X Chen, F Zhou, L Zheng, G Wang, J Gao, Y Ma, L Zheng, B Fu and R Yu. Mesoporous superparamagnetic cobalt ferrite nanoclusters: Synthesis, characterization and application in drug delivery. J. Magn. Magn. Mater. 2020; 498, 166222.
Y Zhang, H Fang, Y Zhang, M Wen, D Wu and Q Wu. Active cobalt induced high catalytic performances of cobalt ferrite nanobrushes for the reduction of p-nitrophenol. J. Colloid Interface Sci. 2019; 535, 499-504.
Y Wang and X Liu. Enhanced catalytic performance of cobalt ferrite by a facile reductive treatment for H2 release from ammonia borane. J. Mol. Liq. 2021; 343, 117697.
B Purnama, AT Wijayanta and Suharyana. Effect of calcination temperature on structural and magnetic properties in cobalt ferrite nano particles. J. King Saud Univ. Sci. 2019; 31, 956-60.
M Hashim, A Ahmed, SA Ali, SE Shirsath, MM Ismail, R Kumar, S Kumar, SS Meena and D Ravinder. Structural, optical, elastic and magnetic properties of Ce and Dy doped cobalt ferrites. J. Alloy. Comp. 2020; 834, 155089.
X Wu, Z Ding, N Song, L Li and W Wang. Effect of the rare-earth substitution on the structural, magnetic and adsorption properties in cobalt ferrite nanoparticles. Ceram. Int. 2016; 42, 4246-55.
G Kumar, RK Singh, SS Kumar, A Manash, A Prabha, U Shankar and O Priya. Low temperature synthesis and influence of Ce3+ substitution on structural and magnetic properties of nanocrystalline cobalt ferrite using citrate precursor sol-gel method. Mater. Today Proc. 2022; 49, 1664-9, 2022.
SG Kakade, RC Kambale, YD Kolekar and CV Ramana. Dielectric, electrical transport and magnetic properties of Er3+substituted nanocrystalline cobalt ferrite. J. Phys. Chem. Solid. 2016; 98, 20-7.
A Tijerina-Rosa, JM Greneche, AF Fuentes, J Rodriguez-Hernandez, JL Menéndez, FJ Rodríguez-González and SM Montemayor. Partial substitution of cobalt by rare-earths (Gd or Sm) in cobalt ferrite: Effect on its microstructure and magnetic properties. Ceram. Int. 2019; 45, 22920-9.
M Kamran and M Anis-ur-Rehman. Enhanced transport properties in Ce doped cobalt ferrites nanoparticles for resistive RAM applications. J. Alloy. Comp. 2020; 822, 153583.
M Dhiman and S Singhal. Enhanced catalytic properties of rare-earth substituted cobalt ferrites fabricated by sol-gel auto-combustion route. Mater. Today Proc. 2019; 14, 435-44.
SB Das, RK Singh, V Kumar, N Kumar, P Singh and NK Naik. Structural, magnetic, optical and ferroelectric properties of Y3+ substituted cobalt ferrite nanomaterials prepared by a cost-effective sol-gel route. Mater. Sci. Semicond. Proc. 2022; 145, 106632.
DM Ghone, KK Patankar, VL Mathe and SD Kaushik. Influence of substitution of yttrium in cobalt ferrite on the structural, magnetic and magnetostrictive properties. AIP Conf. Proc. 2018; 1942, 130026.
DM Ghone, VL Mathe, KK Patankar and SD Kaushik. Magnetic and magnetostrictive properties of sol-gel prepared Y substituted cobalt ferrite. AIP Conf. Proc. 2019; 2115, 030503.
SS Satpute, SR Wadgane, K Desai, DR Mane and RH Kadam. Substitution effect of Y3+ ions on the structural, magnetic and electrical properties of cobalt ferrite nanoparticles. Cerâmica 2020; 66, 43-9.
IH Dunn, SE Jacobo and PG Bercoff. Structural and magnetic influence of yttrium-for-iron substitution in cobalt ferrite. J. Alloy. Comp. 2017; 691, 130-7.
TEP Alves, HVS Pessoni and AF Jr. The effect of Y3+ substitution on the structural, optical band-gap, and magnetic properties of cobalt ferrite nanoparticles. Phys. Chem. Chem. Phys. 2017; 19, 16395-405.
A Franco, HVS Pessoni and TEP Alves. Enhanced dielectric permittivity on yttrium doped cobalt ferrite nanoparticles. Mater. Lett. 2017; 208, 115-7.
MK Shobana, W Nam and H Choe. Yttrium-doped cobalt nanoferrites prepared by sol-gel combustion method and its characterization. J. Nanoscience Nanotechnology 2013; 13, 3535-8.
M Ishaque, MA Khan, I Ali, HM Khan, MA Iqbal, MU Islam and MF Warsi. Study on the electromagnetic behavior evaluation of Y3+ doped cobalt nanocrystals synthesized via co-precipitation route. J. Magn. Magn. Mater. 2014; 372, 68-73.
M Houshiar, F Zebhi, ZJ Razi, A Alidoust and Z Askari. Synthesis of cobalt ferrite (CoFe2O4) nanoparticles using combustion, coprecipitation, and precipitation methods: A comparison study of size, structural, and magnetic properties. J. Magn. Magn. Mater. 2014; 371, 43-8.
MK Shobana, H Kwon and H Choe. Structural studies on the yttrium-doped cobalt ferrite powders synthesized by sol-gel combustion method. J. Magn. Magn. Mater. 2012; 324, 2245-8.
X Wu, H Yu and H Dong. Enhanced infrared radiation properties of CoFe2O4 by doping with Y3+ via sol-gel auto-combustion. Ceram. Int. 2014; 40, 12883-9.
MSA Maashani, KA Khalaf, AM Gismelseed and IA Al-Omari. The structural and magnetic properties of the nano-CoFe2O4 ferrite prepared by sol-gel auto-combustion technique. J. Alloy. Comp. 2020; 817, 152786.
L Kumar, P Kumar, A Narayan and M Kar. Rietveld analysis of XRD patterns of different sizes of nanocrystalline cobalt ferrite. Int. Nano Lett. 2013; 3, 8.
LE Caldeira, CS Erhardt, FR Mariosi, J Venturini, RYS Zampiva, ORK Montedo, S Arcaro, CP Bergmann and SR Bragança. Correlation of synthesis parameters to the structural and magnetic properties of spinel cobalt ferrites (CoFe2O4) - an experimental and statistical study. J. Magn. Magn. Mater. 2022; 550, 169128.
NA Rana, V Kumar and AM Awasthi. Effect of dopant concentration and annealing temperature on electric and magnetic properties of lanthanum substituted CoFe2O4 nanoparticles for potential use in 5G wireless communication systems. Ceram. Int. 2021; 47, 20669-77.
E Hutamaningtyas, Utari, Suharyana, AT Wijayanta and B Purnama. FTIR and structural properties of co-precipitated cobalt ferrite nano particles. J. Phys. Conf. Ser. 2016; 776, 012023.
NP Prasetya, RI Setiyani, Utari, Kusumandari, Y Iriani, J Safani, A Taufiq, NA Wibowo, Suharno and B Purnama. Cation trivalent tune of crystalline structure and magnetic properties in coprecipitated cobalt ferrite nanoparticles. Mater. Res. Exp. 2023; 10, 036102.
Riyatun, T Kusumaningsih, A Supriyanto, HB Akmal, FM Zulhaina, NP Prasetya and B Purnama. Nanoparticle-preparation-procedure tune of physical, antibacterial, and photocatalyst properties on silver substituted cobalt ferrite. Results Eng. 2023; 18, 101085.
OK Mmelesi, N Masunga, A Kuvarega, TT Nkambule, BB Mamba and KK Kefeni. Cobalt ferrite nanoparticles and nanocomposites: Photocatalytic, antimicrobial activity and toxicity in water treatment. Mater. Sci. Semicond. Proc. 2021; 123, 105523.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






