Phytochemicals, Cytotoxicity, Inhibitory Effect on Nitric Oxide Production and Alpha-Glucosidase Activity, and DPPH Scavenging Activity of Leaf Extract from Gymnema inodorum (Lour.) Decne
DOI:
https://doi.org/10.48048/tis.2023.6548Keywords:
Gymnema inodorum, Chemical components, α-glucosidase, DPPH, NO production, Phytochemicals, Inflammatory mediatorAbstract
Investigation of phytochemicals, cyto-toxicity, inhibitory effect on nitric oxide (NO) production and α-glucosidase activity, and DPPH scavenging activity of leaf extract from Gymnema inodorum (Lour.) Decne (LEGI) revealed that GC-MS analysis demonstrated the presence of 83 phytochemicals with 5 main compounds i.e. Phytol (18.14 %), n-Hexadecanoic acid (12.45 %), 9,12-Octadecadienoic acid (Z,Z)-(3.92 %), 9,12,15-Octadecatrienoic acid, (Z,Z,Z)-(10.39 %) and Betulin (8.56 %), and 78 trace compounds in LEGI. MTT assay to investigate cytotoxicity showed that LEGI exhibited low cytotoxicity to the RAW 264.7 macrophages with IC50 of 128.77±2.82 µg/mL. Treatment with the LEGI significantly inhibited nitric oxide (NO) production, an inflammatory mediator, in the LPS-stimulated RAW 264.7 cells. At the concentration of 50 mg/mL, LEGI inhibited NO production by 16.73±1.52 % which was less than Diclofenac did (27.07±1.81 %). The α-glucosidase inhibitory activity to determine the antidiabetic activity showed that LEGI exhibited the inhibition on α-glucosidase activity with IC50 of 0.34 mg/mL, which was less potent than Acarbose (IC50 of 0.20 mg/mL). Assay of 2,2-diphenyl-2-picrylhydrazyl- hydrate (DPPH) radical scavenging to examine antioxidant activity revealed that LEGI demonstrated less potent DPPH scavenging activity compared to Butylated hydroxytoluene, BHT with IC50 of 0.43 vs 0.36 mg/mL, respectively. These results indicate that the leaf extract from G. inodorum possesses anti-inflamatory, antidiabetic and antioxidant activities that can be developed and used as natural agents for the tréamatent of diseases related to these activities.
HIGHLIGHTS
- Using GC-MS analysis, there are 83 phytochemicals identified with 5 main compounds i.e. Phytol, n-Hexadecanoic acid, 9,12-Octadecadienoic acid (Z,Z)-, 9,12,15-Octadecatrienoic acid, (Z,Z,Z)- and Betulin, and 78 trace compounds in the ethanol leaf extract from Gymnema inodorum, and Phytol is the most predominant compound
- The extract can inhibit nitric oxide (NO) production, indicating its anti-inflammatory property
- The extract displays the inhibitory effect on α-Glucosidase activity, suggesting that the extract can be applied for antidiabetic management
- The extract possesses antioxidant property as evidenced by scavenging DPPH free radicals
- The extract shows low cytotoxicity to RAW 264.7 macrophages with IC50 of 128.77±2.82 µg/ml, by MTT assay
GRAPHICAL ABSTRACT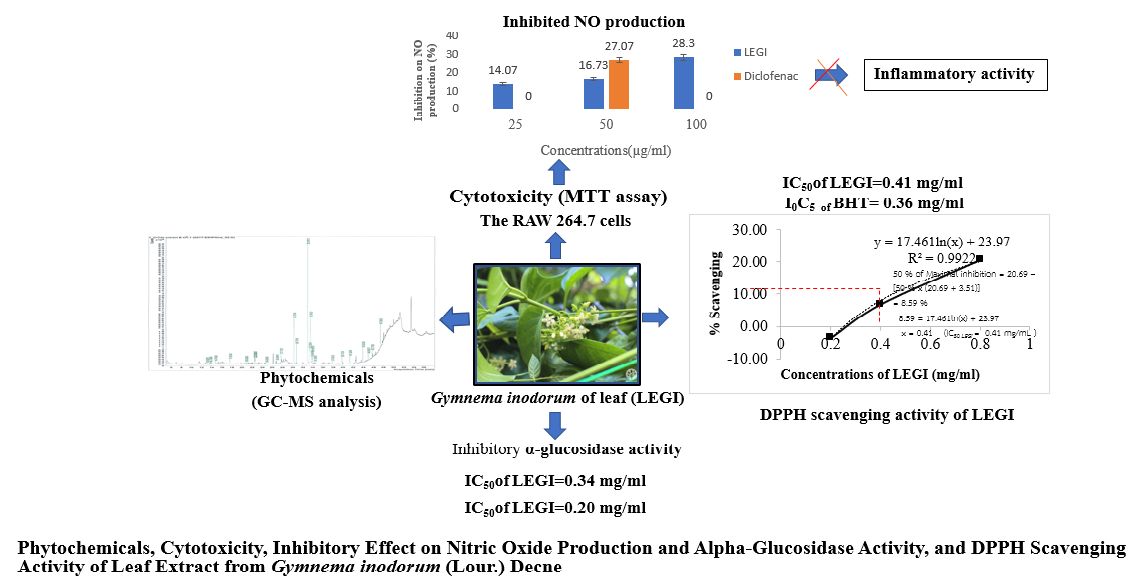
Downloads
References
K Tiamyom, K Sirichaiwetchakoon, T Hengpratom, S Kuppitayanant, R Srisawat, A Thaeomor and G Eumkeb. The effects of Cordyceps sinensis (Berk.) Sacc. and Gymnema inodorum (Lour.) Decne. Extracts on adipogenesis and lipase activity in vitro. Evid. Based Complement. Altern. Med. 2019; 2019, 5370473.
N Chongviriyaphan, T Muangman, C Pisalphong, NP Molarese, P Klungsupya, U Suthutvoravut. Antioxidant activity of Gymnema inodorum Decne. (Pak chiang daa) and its effect on DNA damage. FASEB J. 2006; 20, LB90-LB90.
A Chiabchalard, T Tencomnao and R Santiyanont. Effect of Gymnema inodorum on postprandial peak plasma glucose levels in healthy human. Afr. J. Biotechnol. 2010; 9, 1079-85.
B Dunkhunthod, C Talabnin, M Murphy, K Thumanu, P Sittisart and G Eumkeb. Gymnema inodorum (Lour.) Decne. Extract alleviates oxidative stress and inflammatory mediators produced by RAW264.7 macrophages. Oxid. Med. Cell. Longev. 2021; 2021, 8658314.
MPT Maw and N Piyapolrungroj. Effect of Gymnema inodorum on P-glycoprotein function: An in vitro study. In: Proceedings of the 5th International Conference on Pharmaceuticals, Nutraceuticals and Cosmetic Science, Bangkok, Thailand. 2017, p. 153-6.
A Saneja, C Sharma, K Aneja and R Pahwa. Gymnema sylvestre (Gurmar): A review. Pharm. Lett. 2010; 2, 275-84.
P Yingthongchai, P Sirikhum, S Nilsamranchit and S Tateing. Comparison of nutritional value and bioactive compounds in Gymnema inodorum Decne. leaves. J. Agr. 2018; 34, 363-72.
A Chanwitheesuk, A Teerawutgulrag and N Rakariyatham. Screening of antioxidant activity and antioxidant compounds of some edible plants of Thailand. Food Chem. 2005, 92, 491-7.
DT Trang, DTH Yen, NT Cuong, LT Anh, NT Hoai, BH Tai, VV Doan, PH Yen, TH Quang, NX Nhiem, CV Minh and PV Kiem. Pregnane glycosides from Gymnema inodorum and their α-glucosidase inhibitory activity. Nat. Prod. Res. 2019; 35, 2157-63.
T Muangman, N Chongviriyaphan, C Pisalphong, NP Molarese, P Klungsupya. Free radical scavenging activity of Gymnema inodorum Decne extracts. J. Nutr. Assoc. Thai. 2007; 42,19-28.
W Srinuanchai, R Nooin, P Pitchakarn, J Karinchai, U Suttisansanee, C Chansriniyom, S Jarussophon, P Onanong and Nuchuchua. Inhibitory effects of Gymnema inodorum (Lour.) Decne. leaf extracts and its triterpene saponin on carbohydrate digestion and intestinal glucose absorption. J. Ethnopharmacol. 2021; 10, 113-398.
A Wongnoppavich, A Bonness and N Waloh. 2014, Effects of ethanolic extracts from Azadirachta indica and Gymnema inodorum leaves on anti-inflammation in macrophage and anti-insulin resistance in adipocyte. Master’s thesis. Chiang Mai University, Chiang Mai, Thailand.
W Srinuanchai, P Pitchakarn, S Jarussophon and O Nuchuchua. Antidiabetic phytoconstituents and cytotoxicity of Gymnema inodorum (Lour.) Decne leaf extract, Available at: https://www.semanticscholar.org/paper/Antidiabetic-phytoconstituents-and-cytotoxicity-of-Srinuanchai-Pitchakarn/509d9f0a30e427281a13c19862879f1dc4e8685c#paper-header.
R Bespinyowong, S Pongthananikorn and A Chiabchalard. Efficacy and safety of Gymnema inodorum tea consumption in type 2 diabetic patients. Chula. Med. J. 2013, 57, 587-99.
JP An, EJ Park, B Ryu, BW Lee, HM Cho, TP Doan, HTT Pham and WK Oh. Triterpenoids from the leaves of Gymnema inodorum and their insulin mimetic activities. J. Nat. Prod. 2020, 83, 1265-74.
P Klungsupya, T Muangman, N Theangtrong, A Khayungarnnawee, W Phatvej, K thisayakorn, U Rerk-Am, T Sematong, S Trangvacharakul and V Arunpairojana. Antioxidant and antihyperglycemic activities of Gymnema inodorum Dence. In: Proceedings of the 8th NRCT-JSPS Joint Seminar WInnovative Research in Natural Products for Substainable Development, Chulalongkorn University. 2008, p. 1-3.
P Klungsupya, T Muangman, N Theangtrong, A Khayungarnnawee, W Phatvej, K Thisayakorn, U Rerk-Am, T Sematong, S Trangvacharakul, V Arunpairojana. Antioxidant and antihyperglycemic activities of Gymnema inodorum Dence. In: Proceedings of the 9th NRCT-JSPS Joint Seminar Innovative Research in Natural Products for Sustainable Development, Bangkok, Thailand, 2009, p. 207-9.
W Keapai, P Toejing, J Intakhad, P Saengratwatchara, T Tangjai and N Lailerd. Anti-diabetic activity of Gymnema inodorum extract in high-fat diet and streptozotocin-induced diabetic rats: Preliminary study. In: Proceedings of the 1st International Conference on Innovation of Functional Foods in Asia. University of Phayao, Phayao, Thailand. 2018, p. 1-19.
J Thongdon-A, P Klungsupya, T Muangman, A Tantrawong and S Trangvacharakul. Use of three Thai indigenous vegetables as potential dietary fiber sources for health food product. Thai. J. Pharm. Sci. 2013; 38, 48-51.
N Buddhakala and C Talubmook. Toxicity and antidiabetic activity of ethanolic extract of Sphagneticola trilobata (L.) Pruski flower in rats. J. Ethnopharmacol. 2020, 262, 113-28.
JV Meerloo, GJL Kaspers and J Cloos. Cell sensitivity assays: The MTT assay. Meth. Mol. Bio. 2011; 731, 237-45.
HQ Dong, M Li, F Zhu, FL Liu and JB Huang. Inhibitory potential of trilobatin from Lithocarpus polystachyus Rehd against α-glucosidase and α-amylase linked to type 2 diabetes. Food Chem. 2012; 30, 261-6.
V Aparna, V Dileep, M Pradeep, K Ponnuraj, S Chittalakkottu and M Haridas. Anti-Inflammatory property of n-Hexadecanoic acid: Structural evidence and kinetic assessment. Chem. Biol. Drug Des. 2012; 80, 434-9.
SU Ponnamma and K Manjunath. GC-MS analysis of phytocomponents in the methanolic extract of Justicia wyaadensis (Nees) T. Anders. Int. J. Pharm. Bio Sci. 2012; 3, P570-P576.
PN Ruvanthika, S Manikandan and S Lalitha. A comparative study on phytochemical screening of aerial parts of Nelumbo nucifera Gaertn by gas chromatographic mass spectrometry. Int. J. Pharm. Sci. Res. 2016; 8, 2258-66.
S Chitra and J Karthikeyan. Phytochemical profiling of cat whisker’s (Orthosiphon stamineus) tea leaves extract. J. Pharmacogn. Phytochem. 2018; 7, 1396-402.
CCMP Santos, MS Salvadori, VG Mota, LM Costa, AACO Almeida GALD Oliveira, JP Costa, DPD Sousa, RMD Freitas and RND Almeida. Antinociceptive and antioxidant activities of phytol in vivo and in vitro models. Neurosci J. 2013; 2013, 949452.
JP Costa, MT Islam, PS Santos, PB Ferreira, GL Oliveira, MV Alencar, MF Paz, ÉL Ferreira, CM Feitosa, AM Citó, DP Sousa, AA Melo-Cavalcante. Evaluation of antioxidant activity of phytol using non- and pre-clinical models. Curr. Pharm. Biotechnol. 2016; 17, 1278-84.
JD Moraes, RND Oliveira, JP Costa, ALG Junior, DPD Sousa, RM Freitas, SM Allegretti, PLS Pinto. Phytol, a diterpene alcohol from chlorophyll, as a drug against neglected tropical disease Schistosomiasis mansoni. PLoS Negl. Trop. Dis. 2014; 8, e2617.
KR Ryu, JY Choi, S Chung and DH Kim. Anti-scratching behavioral effect of the essential oil and phytol isolated from Artemisia princeps Pamp. in mice. Planta Med. 2011; 77, 22-6.
MT Islam, SA Ayatollahi, SM Neamul, K Zihad, N Sifat, MdR Khan, A Paul, B Salehi, T Islam, MS Mubarak, N Martins and J Sharifi-Rad. Phytol anti-inflammatory activity: Pre-clinical assessment and possible mechanism of action elucidation. Cell. Mol. Biol. 2020; 66, 264-9.
RO Silva, FB Sousa, SR Damasceno, NS Carvalho, VG Silva, FR Oliveira, DP Sousa, KS Aragão, AL Barbosa, RM Freitas and JV Medeiros. Phytol, a diterpene alcohol, inhibits the inflammatory response by reducing cytokine production and oxidative stress. Fundam. Clin. Pharmacol. 2014; 28, 455-64.
HC Upadhyay, A Mishra, J Pandey, P Sharma, AK Tamrakar, AK Srivastava, F Khan and SK Srivastava. In vitro, in vivo and in silico antihyperglycemic activity of some semi-synthetic phytol derivatives. Med. Chem. 2022; 18, 115-21.
S Gunasekarana, T Vijay, K Sarumathyd, S Palanie, RPS Panneerselvamb and V Srinivasanba. Phytoconstituents evaluation by GC-MS and therapeutic efficacy of Grewia umbellifera on Streptozotocin (STZ) induced diabetic. Int. J. Pharm. Sci. 2013; 4, 2380-6.
DE Barre. The role of consumption of alpha-linolenic, eicosapentaenoic and docosahexaenoic acids in human metabolic syndrome and type 2 diabetes - a mini-review. J. Oleo Sci. 2007; 56, 319-25.
N Blondeau, RH Lipsky, M Bourourou, MW Duncan, PB Gorelick and AM Marini. Alpha-linolenic acid: An omega-3 fatty acid with neuroprotective properties - Ready for use in the stroke clinic. BioMed Res. Int. 2015; 2015, 519830.
X Ci, J Zhou, Lv Hongming, Q Yu, L Peng and S Hua. Betulin exhibits anti-inflammatory activity in LPS-stimulated macrophages and endotoxin-shocked mice through an AMPK/AKT/Nrf2-dependent mechanism. Cell Death Dis. 2017; 8, e2798.
A Boparai, J Niazi, N Bajwa and PA Singh. Betulin a pentacyclic tri-terpenoid: An hour to rethink the compound. J. Transl. Med. Res. 2017; 1, 53-9.
GS Thakur, R Sharma, BS Sanodiya, M Pandey, G Prasad and PS Bisen. Gymnema sylvestre: An alternative therapeutic agent for management of diabetes. J. Appl. Pharm. Sci. 2012; 2, 1-6.
E Shanmugasundaram, KL Gopinath, KR Shanmugasundaram and V Rajendran. Possible regeneration of the islets of Langerhans in streptozotocin-diabetic rats given Gymnema sylvestre leaf extracts. J. Ethnopharmacol. 1990; 30, 265-79.
HG Preuss, ST Jarrell, R Scheckenbach, S Lieberman and RA Anderson. Comparative effects of chromium, vanadium and Gymnema sylvestre on sugar-induced blood pressure elevations in SHR. J. Am. Coll. Nutr. 1998; 17, 116-23.
R Chattopadhyay. A comparative evaluation of some blood sugar lowering agents of plant origin. J. Ethnopharmacol. 1999; 67, 367-72.
Y Ogawa, K Sekita, T Umemura, M Saito, A Ono, Y Kawasaki, O Uchida, Y Matsushima, T Inoue and J Kanno. Gymnema sylvestre leaf extract: A 52-week dietary toxicity study in Wistar rats. Shokuhin Eiseigaku zasshi. 2004; 45, 8-18.
J Wungrath. Antidiabetic activity of Chiang Da (Gymnema inodorum) local plant of Northern Thailand. Eur. J. Mol. Clin. Med. 2020; 7, 4710-17.
S Nair and R Keshavachandran. Genetic variability of chakkarakolli (Gymnena sylvestre R. Br.) in Kerala assessed using morphological and biochemical markers. J. Trop. Agr. 2006; 44, 64-6.
N Sandech. 2019, Efficiency of Gymnemic acid from Gymnema sylvestre for restoration and improvement of brain vascular characteristic in diabetic rat. Master’s thesis. Prince of Songkla University, Songkla, Thailand.
K Shimizu, M Ozeki, K Tanaka, K Itoh, S Nakjyo, N Urakawa and M Atsuchi. Suppression of glucose absorption by extracts from the leaves of Gymnema inodorum. J. Vet. Med. Sci. 1997; 59, 753-7.
K Shimizu, M Ozeki, A Iino, S Nakajyo, N Urakawa and M Atsuchi. Structure-activity relationship of triterpenoid derivatives extracted from Gymnema inodorum leaves on glucose absorption. Jpn. J. Pharmacol. 2002; 86, 223-9.
P Kanetkar, K Laddha and M Kamat. Gymnemic acids: A molecular perspective of its action on carbohydrate metabolism. In: Proceedings of the 16th Indian Convention of Food Scientists and Technologists, Mysore, India. 2004.
N Taokaenchan, K Areesrisom, P Areesrisom, V Maifaey and S Sateinperakul. Total phenolic compound, flavonoid content and antioxidant activity of Gymnema inodorum tea from difference process. J. Agr. Prod. 2020; 2, 51-61.
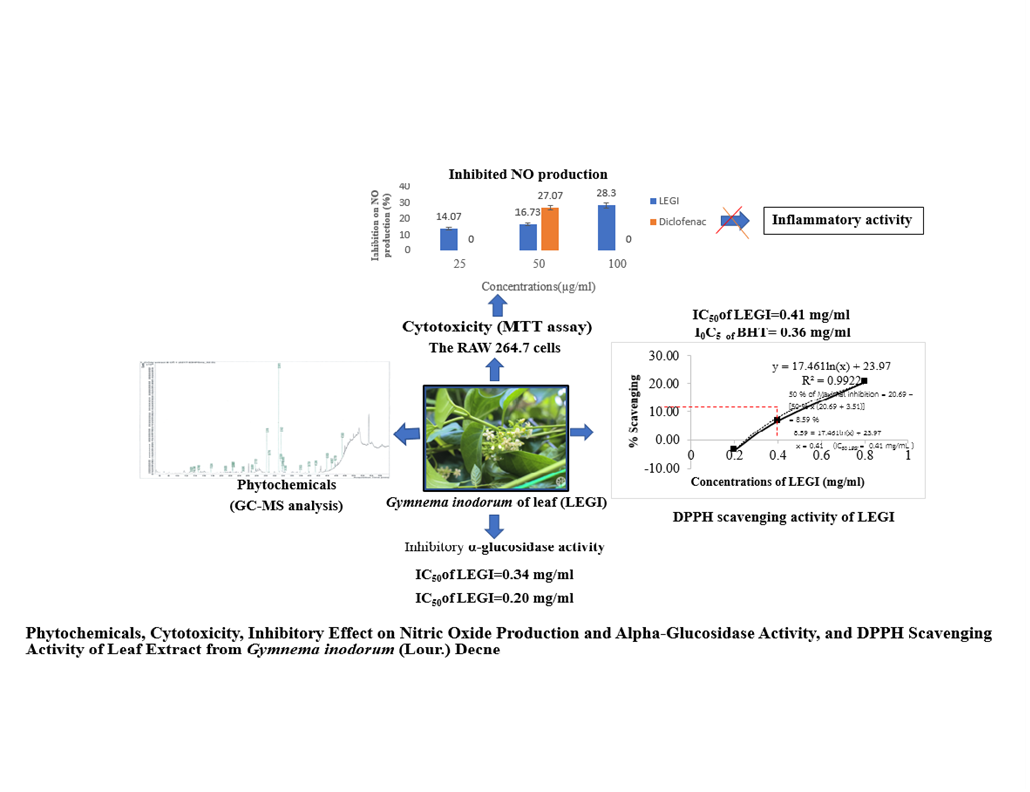
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






