Caffeic Acid Phenethyl Ester from NanoPropolis Inhibits Peptide Deformylase in Staphylococcus aureus and Pseudomonas aeruginosa: A Novel Strategy Against Endocarditis
DOI:
https://doi.org/10.48048/tis.2026.11774Keywords:
Caffeic Acid Phenethyl Ester (CAPE), Peptide Deformylase (PDF), Staphylococcus aureus, Pseudomonas aeruginosa, Molecular Dynamics, Nanopropolis, Antibacterial, Endocarditis, Caffeic Acid Phenethyl Ester (CAPE), Peptide Deformylase (PDF), Staphylococcus sureus, Pseudomonas aeruginosa, Molecular dynamics, Nanopropolis, Antibacterial, EndocarditisAbstract
Bacterial infections, particularly bacteremia and bacterial endocarditis caused by Staphylococcus aureus and Pseudomonas aeruginosa, pose significant global health challenges. Peptide Deformylase (PDF), a metalloenzyme essential for bacterial viability and absent in eukaryotic cells, is a promising target for novel antibacterial drug development. Caffeic Acid Phenethyl Ester (CAPE), a major active component of propolis, exhibits potent antimicrobial properties. This study investigates the potential of nanoencapsulated CAPE derived from Apis trigona propolis as a therapeutic agent against S. aureus and P. aeruginosa by inhibiting bacterial PDF activity. Physicochemical characterization confirmed the successful formation of stable nanoencapsulates with an average particle size of 109 ± 15 nm and good colloidal stability. Molecular docking studies revealed that CAPE exhibits strong binding affinity to the active sites of S. aureus PDF (PDB ID: 1Q1Y) and P. aeruginosa PDF (PDB ID: 1LRY), comparable to or exceeding that of the reference inhibitor, actinonin. Detailed analysis of docking poses indicated crucial interactions with key amino acid residues within the PDF active site. Furthermore, 20 ns molecular dynamics simulations demonstrated that the CAPE-PDF complexes remained stable, maintaining key hydrogen bonds and hydrophobic interactions, indicating robust and persistent binding. These findings suggest that nanoencapsulated CAPE holds significant promise as a novel antibacterial strategy by targeting essential bacterial PDF activity, potentially mitigating the risk of severe systemic infections like bacterial endocarditis.
HIGHLIGHTS
- Bacteremia and bacterial endocarditis caused by S. aureus and P. aeruginosa remain critical global health concerns.
- Nanoencapsulated CAPE from Apis trigona propolis was successfully formulated with stable physicochemical properties (109 ± 15 nm).
- CAPE exhibited strong binding affinity to bacterial peptide deformylase (PDF), comparable to actinonin.
- Molecular dynamics (20 ns) confirmed stable CAPE-PDF complexes with persistent key interactions.
- CAPE from NanoPropolis shows promise as a novel antibacterial candidate for mitigating systemic infections via PDF inhibition.
GRAPHICAL ABSTRACT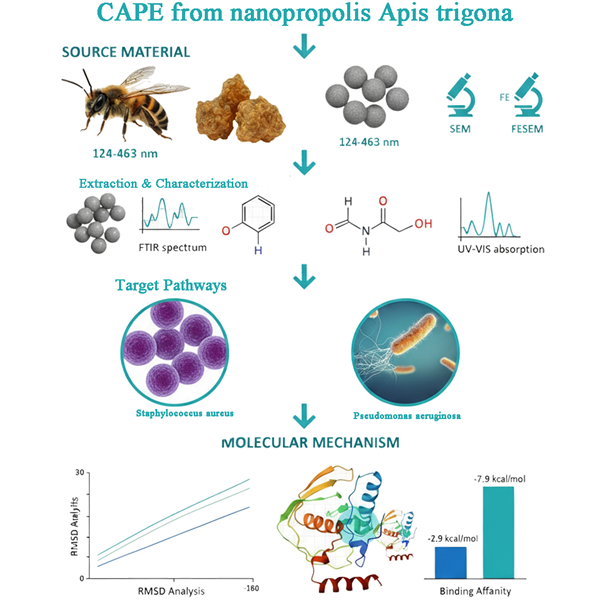
Downloads
References
JL Falconer, R Rajani, V Androshchuk, A Yogarajah, RA Greenbury, A Ismail, N Oh, L Nibali, EM D’Agostino and V Sousa. Exploring links between oral health and infective endocarditis. Frontiers in Oral Health 2024; 5, 1426903.
F Nappi. Staphylococcus aureus endocarditis immunothrombosis. Metabolites 2025; 15(5), 328.
AS Lee, H De Lencastre, J Garau, J Kluytmans, S Malhotra-Kumar, A Peschel and S Harbarth. Methicillin-resistant Staphylococcus aureus. Nature Reviews Disease Primers 2018; 4(1), 18033.
W Zhang, GE Margarita, D Wu, W Yuan, S Yan, S Qi, X Xue, K Wang and L Wu. Antibacterial activity of Chinese red propolis against Staphylococcus aureus and MRSA. Molecules 2022; 27(5), 1693.
KM Alarjani, HM Yehia, AN Badr, HS Ali, AH Al-Masoud, SM Alhaqbani, SA Alkhatib and AM Rady. Anti-MRSA and biological activities of propolis concentrations loaded to chitosan nanoemulsion for pharmaceutics applications. Pharmaceutics 2023; 15(10), 2386.
AH Siddiqui and J Koirala. Methicillin-resistant Staphylococcus aureus. The Handbook of Zoonotic Diseases of Goats 2023; 4(1), 202-207.
V Rapti, E Giannitsioti, N Spernovasilis, AP Magiorakos and G Poulakou. The evolving landscape of infective endocarditis: Difficult-to-treat resistance bacteria and novel diagnostics at the foreground. Journal of Clinical Medicine 2025; 14(6), 2087.
X Li, KM Kolltveit, L Tronstad and I Olsen. Systemic diseases caused by oral infection. Clinical Microbiology Reviews 2000; 13(4), 547-558.
D Mader, MJ Rabiet, F Boulay and A Peschel. Formyl peptide receptor-mediated proinflammatory consequences of peptide deformylase inhibition in Staphylococcus aureus. Microbes and Infection 2010; 12(5), 415-419.
W Wang, R White and Z Yuan. Proteomic study of peptide deformylase inhibition in Streptococcus pneumoniae and Staphylococcus aureus. Antimicrobial Agents and Chemotherapy 2006; 50(5), 1656-1663.
K Cui, W Lu, L Zhu, X Shen and J Huang. Caffeic acid phenethyl ester (CAPE), an active component of propolis, inhibits Helicobacter pylori peptide deformylase activity. Biochemical and Biophysical Research Communications 2013; 435(2), 289-294.
M Cahyati, ZD Salsabila, A Susilo, D Pranowo and Nurjannah. Effect of propolis and liquid smoke nanogel on TGF-β and macrophage activity in rattus norvegicus with traumatic ulcer. International Journal of Design and Nature and Ecodynamics 2024; 19(6), 1963-1969.
A Susilo, M Cahyati, Nurjannah, D Pranowo, FE Hermanto and EP Primandasari. Chrysin inhibits Indonesian serotype foot-and-mouth-disease virus replication: Insights from DFT, molecular docking and dynamics analyses. Journal of Tropical Biodiversity and Biotechnology 2024; 9(1), 83140.
SA Hidayat, A Susilo, KUA Awwaly, D Masyithoh and M Cahyati. Molecular docking study of east java propolis compounds as ACE-2 inhibitors for covid-19. AIP Conference Proceedings 2024; 3132, 040021.
EP Primandasari, A Susilo, KUA Awwaly, M Cahyati and D Masyithoh. Evaluation on antioxidant activity and active components of Trigona itama propolis extract and its potential as SARS-CoV2 infection inhibitors. Jurnal Teknologi Pertanian 2023; 24(2), 127-136.
A Barış, EA Abdik and H Abdik. Caffeic acid phenethyl ester (CAPE): An active component of propolis: A review on its therapeutic potentials. Studies in Natural Products Chemistry 2024; 83, 183-205.
Y Niu, K Wang, S Zheng, Y Wang, Q Ren, H Li, L Ding, W Li and L Zhang. Antibacterial effect of caffeic acid phenethyl ester on cariogenic bacteria and Streptococcus mutans biofilms. Antimicrob Agents Chemother 2020; 69(9), 1110-1128.
S Meyuhas, M Assali, M Huleihil and M Huleihel. Antimicrobial activities of caffeic acid phenethyl ester. Journal of Molecular Biochemistry 2015; 4(2), 143.
AA Amin, KF Mahmoud, MF Salama, V Longo, L Pozzo, EI Seliem and MA Ibrahim. Characterization and stability evaluation of Egyptian propolis extract nano-capsules and their application. Scientific Reports 2023; 13(1), 16065.
M Soleimanifard, J Feizy and F Maestrelli. Nanoencapsulation of propolis extract by sodium caseinate-maltodextrin complexes. Food and Bioproducts Processing 2021; 128, 177-185.
J Mehta, K Pathania and SV Pawar. Recent overview of nanotechnology based approaches for targeted delivery of nutraceuticals. Sustainable Food Technology 2025; 3(4), 947-978.
B Venkidasamy, A Shelar, AR Dhanapal, AS Nile, R Patil, Y Zhang, K Kuksal and SH Nile. Emerging biopolymer nanocarriers for controlled and protective delivery of food bioactive compounds - current status and future perspective. Food Hydrocolloids 2025; 160, 110769.
IA Justino, JPR Furlan, IRS Ferreira, A Marincek, JA Aldana-Mejía, LFF Tucci, JK Bastos, EG Stehling, CM Marzocchi-Machado and PD Marcato. Antimicrobial, antioxidant, and anticancer effects of nanoencapsulated Brazilian red propolis extract: Applications in cancer therapy. Processes 2024; 12(12), 2856.
TM Younus, MA Abed, MA Hamood and GG Ali. Study the optical and electrical properties of gold nanoparticles deposited on porous silicon. Trends in Sciences 2025; 22(10), 10404.
IF Pangesti, A Susilo, KU Awwaly, M Cahyati, Nurjannah and D Pranowo. Physical quality of halal propolis extract using the ultrasonic as an active drug ingredient. In: Proceedings of the 3rd International Conference on Environmentally Sustainable Animal Industry 2022 (ICESAI 2022), Malang, Indonesia. 2023, p. 361-370.
S Rampogu, A Zeb, A Baek, C Park, M Son and KW Lee. Discovery of potential plant-derived peptide deformylase (PDF) inhibitors for multidrug-resistant bacteria using computational studies. Journal of Clinical Medicine 2018; 7(12), 563.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






