Green Tea and Green Coffee Therapy for Aortic Calcification Prevention in Metabolic Syndrome Model Rats: Effects on Expression of AKT, mTOR, RUNX2, and Osteopontin Levels
DOI:
https://doi.org/10.48048/tis.2024.8204Keywords:
Cardiovascular disease, Green tea, Green coffee, Metabolic syndrome, Vascular calcificationAbstract
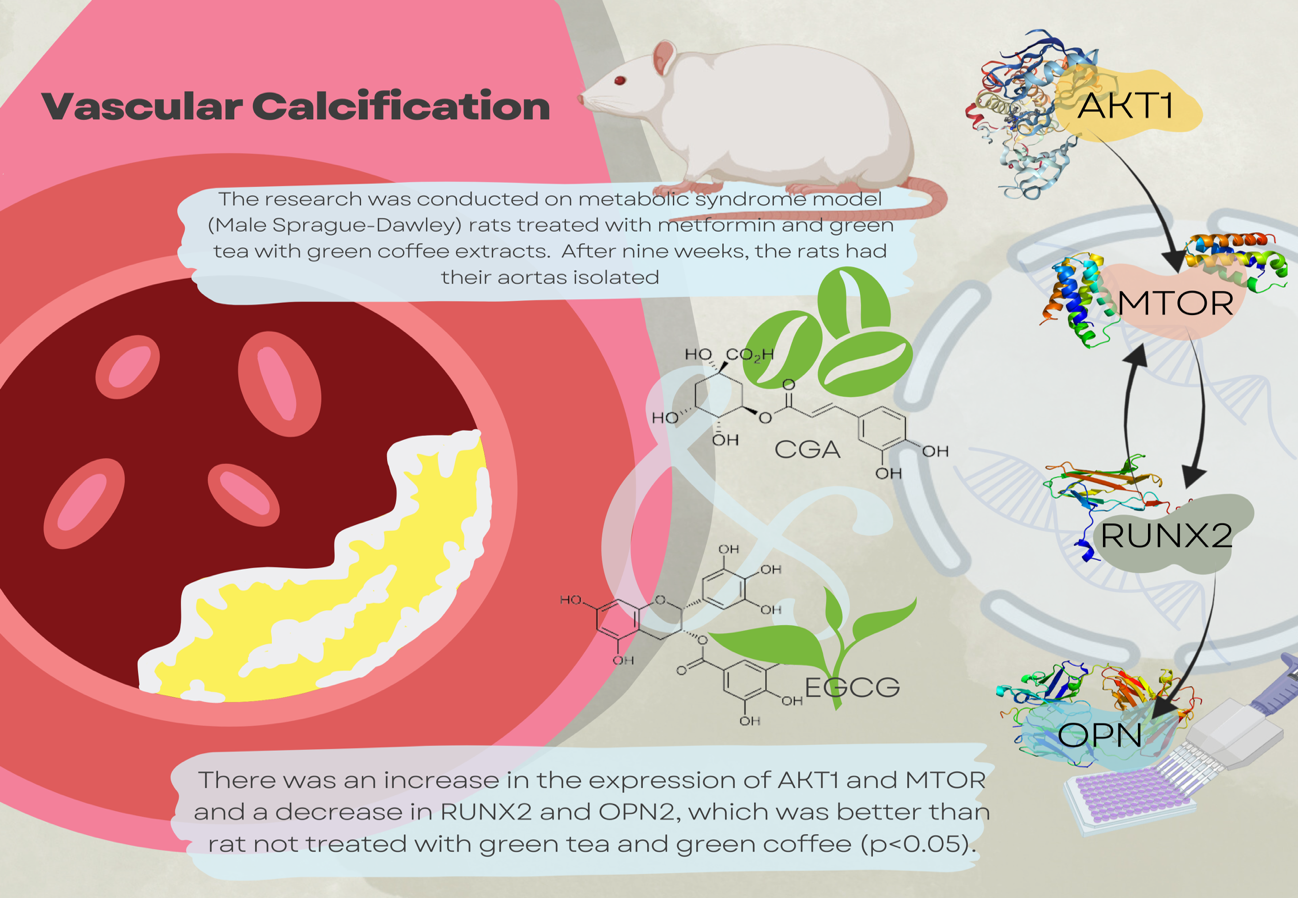
Metabolic syndrome (METS) consists of several independent risk factors for cardiovascular disease (CVD), one of which is vascular calcification (VC). Increased oxidative stress is essential in the pathogenesis of CVD in METS. One of the pathways involved in the pathogenesis of VC is the AKT pathway. Green tea and green coffee have many health benefits, including treating METS risk factors. Although several benefits of green tea and green coffee are known, there is not much information regarding the effects of these 2 extracts for treating heart disease, which is often found in METS sufferers. This research explores the benefits of green tea and green coffee extracts in preventing VC in METS through the AKT/MTOR mechanism. This research focuses on the mechanism of CVD in the AKT-mTOR, RUNX activity, and osteopontin (OPN) expression as one of the downstream pathogenesis of CVD in METS. The research was conducted on METS model rat treated with metformin and green tea with green coffee extracts. Male Sprague-Dawley rats were given a diet high in fat and sugar until they met METS for 4 months, then given treatment for 9 weeks. After 9 weeks, the rat had their aortas isolated for staining with specific antibodies to AKT1, MTOR, RUNX2, and serum OPN levels were measured using ELISA-sandwich methods. There was an increase in the expression of AKT1 and MTOR and a decrease in RUNX2 and OPN2, which was better than rat not treated with green tea and green coffee (p < 0.05). A significant reduction was found in the green tea and green coffee therapy group, which was better than when given metformin alone (p < 0.05). The results of this research showed that there is good potential for therapy using green tea and green coffee extracts to prevent VC in metabolic syndrome models.
HIGHLIGHTS
- Metabolic syndrome consists of several independent risk factors for cardiovascular disease, one of which is vascular calcification. Increased oxidative stress is essential in the pathogenesis of cardiovascular disease in metabolic syndrome.
- This study explores the benefits of green tea and green coffee extracts in preventing vascular calcification in the metabolic syndrome model through the AKT/MTOR mechanism.
- This research focuses on the mechanism of CVD in the AKT-mTOR, RUNX activity, and osteopontin (OPN) expression as one of the downstream patogenesis of CVD in METS. The research was conducted on METS model rats treated with metformin and green tea with green coffee extracts.
- In conclusion, this study showed the potential for therapeutic use of green tea and green coffee extracts to prevent vascular calcification in rat models of metabolic syndrome by increasing AKT1 expression, decreasing MTOR and RUNX2, with the main implication of reducing the circular protein product, osteopontin.
GRAPHICAL ABSTRACT
Downloads
References
P Ranasinghe, Y Mathangasinghe, R Jayawardena, AP Hills and A Misra. Prevalence and trends of metabolic syndrome among adults in the asia-pacific region: A systematic review. BMC Public Health 2017; 17, 101.
NCDV Moreira, A Hussain, B Bhowmik, I Mdala, T siddiquee, VO Fernandes, RMM Junior and HE Meyer. Prevalence of metabolic syndrome by different definitions, and its association with type 2 diabetes, pre-diabetes, and cardiovascular disease risk in Brazil. Diabetes Metab. Syndr. Clin. Res. Rev. 2020; 14, 1217-24.
R Belete, Z Ataro, A Abdu and M Sheleme. Global prevalence of metabolic syndrome among patients with type I diabetes mellitus: A systematic review and meta-analysis. Diabetol. Metab. Syndr. 2021; 13, 25.
A Friend, L Craig and S Turner. The prevalence of metabolic syndrome in children: A systematic review of the literature. Metab. Syndr. Relat. Disord. 2013; 11, 71-80.
EH Herningtyas and TS Ng. Prevalence and distribution of metabolic syndrome and its components among provinces and ethnic groups in Indonesia. BMC Public Health 2019; 19, 377.
T Skurk, C Alberti-Huber, C Herder and H Hauner. Relationship between adipocyte size and adipokine expression and secretion. J. Clin. Endocrinol. Metab. 2007; 92, 1023-33.
JD Tune, AG Goodwill, DJ Sassoon and KJ Mather. Cardiovascular consequences of metabolic syndrome. Transl. Res. 2017; 183, 57-70
D Moris, M Spartalis, E Spartalis, G Karaaachaliou, GI Karaolanis, G Tsourrouflis, DI Tsilimigras, E Tzatzaki and S Theocharis. The role of reactive oxygen species in the pathophysiology of cardiovascular diseases and the clinical significance of myocardial redox. Ann. Transl. Med. 2017; 5, 326.
R D’Oria, R Schipani, A Leonardini, A Natalicchio, S Perrini, A Cignarelli, L Laviola and F Giorgino. The role of oxidative stress in cardiac disease, from physiological response to injury factor. Oxid. Med. Cell. Longev. 2020; 2020, 5732956.
ND Wong, JC Nelson, T Granston, AG Bertoni, RS Blumenthal, JJ Carr, A Guerci, DR Jacobs, R Kronmal, K Liu, M Saad, E Selvin, R Traacy and R Detrano. Metabolic syndrome, diabetes, and incidence and progression of coronary calcium: The multiethnic study of atherosclerosis study. JACC Cardiovasc. Imag. 2012; 5, 358-66.
CM Shanahan, MH Crouthamel, A Kapustin and CM Giachelli. Arterial calcification in chronic kidney disease: Key roles for calcium and phosphate. Circ. Res. 2011; 109, 697-711.
F Rutsch, Y Nitschke and R Terkeltaub. Genetics in arterial calcification: Pieces of a puzzle and cogs in a wheel. Circ. Res. 2011; 109, 578-92.
J Shiau, Y Chuang, J Tang, K Yang, F Chang, M Hou, C Yen and H Chang. The impact of oxidative stress and AKT pathway on cancer cell functions and its application to natural products. Antioxidants 2022; 11, 1845.
JM Heat, Y Sun, K Yuan, WE Bradley, S Litovsky, LJ Dell’ltalia, JC Chatham, H Wu and Y Chen. Activation of AKT by O-linked N-acetylglucosamine induces vascular calcification in diabetes mellitus. Circ. Res. 2014; 114, 1094-102.
L Deng, L Huang, Y Sun JM Heath, H Wu and Y Chen. Inhibition of FOXO1/3 promotes vascular calcification. Arterioscler. Thromb. Vasc. Biol. 2015; 35, 175-83.
MP Reill and DJ Rader. The metabolic syndrome: More than the sum of its parts? Circulation 2003; 108, 1546-51.
P Libby, PM Ridker and GK Hansson. Progress and challenges in translating the biology of atherosclerosis. Nature 2011; 473, 317-25.
M Lukitasari, DA Nugroho, MS Rohman, NIP Nugrahini and TW Sardjono. Light-roasted green coffee extract improved adiponectin, insulin resistance, and metabolic profile of metabolic syndrome rat model. Asian J. Pharmaceut. Clin. Res. 2017; 10, 279-83.
M Lukitasari, DA Nugroho and MS Rohman. 28 Green tea extract administration had a beneficial effect on ppar alpha and ppar gamma gene expression in metabolic syndrome rat model. J. Hypertens. 2018; 36, e9.
DA Nugroho, M Lukitasari, MS Rohman. 29 Dose-dependent effects of green tea extract on adiponectin level and adiponectin receptor gene expression in metabolic syndrome rat models. J. Hypertens. 2018; 36, e9.
MS Rohman, M Lukitasari, DA Nugroho, N Widodo, NIP Nugraheini and TW Sardjono. Development of an experimental model of metabolic syndrome in sprague dawley rat. Res. J. Life Sci. 2017; 4, 76-86.
IN Chomsy, MS Rohman, M Lukitasari, DA Nugroho and H Khotimah. cardiac fibrosis attenuation by chlorogenic acid and epigallocatechin-gallate mediated by suppression of Galectin-3 gene expression and collagen deposition in rat metabolic syndrome model. Indian J. Forensic Med. Toxicol. 2021; 15, 2567-74.
IN Chomsy, MS Rohman, H Khotimah, N Widodo and NIP Nugrahini. Decaffeinated green tea and green coffee extracts as metformin’s add-on enhance metabolic syndrome risk factors and improve the cardiac insulin-gene-related pathway. J. Pharm. Pharmacogn. Res. 2023; 11, 414-25.
C Xue, G Li, J Lu and L Li. Crosstalk between circRNAs and the PI3K/AKT signaling pathway in cancer progression. Signal Transduction Targeted Ther. 2021; 6, 400.
L Wu, Y Xu, K Pantopoulos, X Tan, X Wei, H Zheng and Z Luo. Glycophagy mediated glucose-induced changes of hepatic glycogen metabolism via OGT1-AKT1-FOXO1Ser238 pathway. J. Nutr. Biochem. 2023; 117, 109337.
Z Zhang, H Liu and J Liu. Akt activation: A potential strategy to ameliorate insulin resistance. Diabetes Res. Clin. Pract. 2019, 156, 107092.
T Yuan, B Lupse, K Maedler and A Ardestani. mTORC2 signaling: A path for pancreatic β cell’s growth and function. J. Mol. Biol. 2018; 430, 904-18.
Q Jia, R Yang, S Mehmood and Y Li. Epigallocatechin-3-gallate attenuates myocardial fibrosis in diabetic rats by activating autophagy. Exp. Biol. Med. 2022; 247, 1591-600.
P Shokouh, PB Jeppesen, CB Christiansen, FB Mellbye, K Hermansen and S Gregersen. Efficacy of arabica versus robusta coffee in improving weight, insulin resistance, and liver steatosis in a rat model of type-2 diabetes. Nutrients 2019; 11, 2074.
B Vali, LG Rao and A El-Sohemy. Epigallocatechin-3-gallate increases the formation of mineralized bone nodules by human osteoblast-like cells. J. Nutr. Biochem. 2007; 18, 341-7.
GJV Woudenbergh, R Vliegenthart, FJAV Rooij, A Hofman, JCM Wittemen and JM Geleijnse. Coffee consumption and coronary calcification: The rotterdam coronary calcification study. Arterioscler. Thromb. Vasc. Biol. 2008; 28, 1018-23.
Q Ge, C Ruan, Y Ma, X Tang, Q Wu, J Wang, D Zhu and P Gao. Osteopontin regulates macrophage activation and osteoclast formation in hypertensive patients with vascular calcification. Sci. Rep. 2017; 7, 40253.
Y Yang, Y Wang and P Gao. Osteopontin associated with left ventricular hypertrophy and diastolic dysfunction in essential hypertension. J. Hum. Hypertens. 2020; 34, 388-96.
ZS Lok and AN Lyle. Osteopontin in vascular disease: Friend or foe? Arterioscler. Thromb. Vasc. Biol. 2019; 39, 613-22.
Q Ge, C Ruan, Y Ma, X Tang, Q Wu, J Wang, L Zhu and P Gao. Osteopontin regulates macrophage activation and osteoclast formation in hypertensive patients with vascular calcification. Sci. Rep. 2017; 7, 40253.
Z Xie, M Singh and K Singh. Osteopontin modulates myocardial hypertrophy in response to chronic pressure overload in mice. Hypertension 2004; 44, 826-31.
SA Steitz, MY Speer, MD McKee, L Liaw, M Almeida, H Yang and CM Giachelli. Osteopontin inhibits mineral deposition and promotes regression of ectopic calcification. Am. J. Pathol. 2002; 161, 2035-46.

Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






