Activated Carbon from Waste Coffee Grounds for Effective Methylene Blue Removal in Textile Wastewater Treatment
DOI:
https://doi.org/10.48048/tis.2025.10925Keywords:
Activated carbon, Adsorption, Hexane, Hot water, Methylene blue, Reusability, WCGsAbstract
Utilizing waste coffee grounds (WCGs) to produce activated carbon (WCGs-AC) supports sustainable waste management by converting organic waste into value-added materials. This study investigates the optimization of the production process, physicochemical properties, and adsorption performance of activated carbon derived from WCGs. This study pretreated WCGs using hexane and hot water washing methods, followed by carbonization under oxygen-limited conditions. The resulting WCGs-AC exhibited grain sizes ranging from 1.34 to 1,376.00 nm, high specific surface areas (1,304 - 3,405 m²/g), pore volumes of 1.262 - 4.079 cm³/g, and average pore radii between 1.936 and 2.397 nm. Functional groups such as amine (N–H), hydroxyl (–OH), and C–O were identified on the WCGs-AC surface. All WCGs-AC samples achieved 100% Methylene Blue (MB) removal, with similar adsorption capacities ranging from 10.20 ± 0.00 to 10.74 ± 0.02 mg/g. The highest-performing adsorbent was obtained using the hot water washing method and carbonization at 550 °C (WZ550), which exhibited an effectively infinite adsorption rate and sustained 100% removal efficiency over 6 reuse cycles. Various kinetic models, including pseudo-1st-order (PFO), pseudo-2nd-order (PSO), Elovich, and Weber-Morris, were applied to describe the adsorption behavior. These findings demonstrate that low-cost and efficient adsorbents can be derived from coffee waste, offering promising potential for dye removal in textile wastewater treatment.
HIGHLIGHTS
- WCGs are extracted by the hot water and hexane washing method, then carbonized without a gas flow.
- The activated carbon showed large surface areas, 1,300 - 3,400 m²/g
- The WZ550 demonstrated sustained 100% removal efficiency across 6 reuse cycles
- Adsorption kinetics fixed to the PSO model with R2 = 1
GRAPHICAL ABSTRACT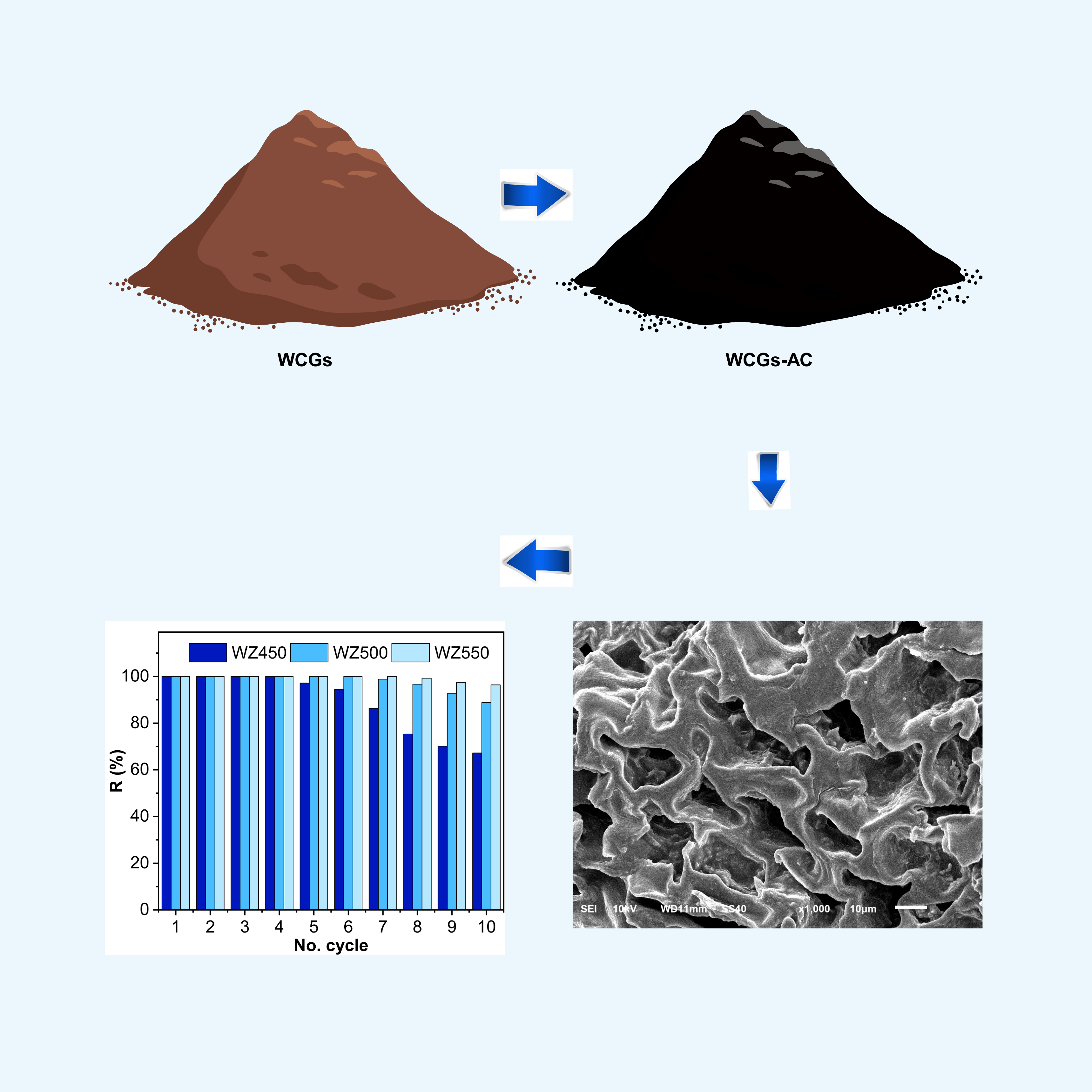
Downloads
References
I Ali, P Singh, HYA -Enein and B Sharma. Chiral analysis of ibuprofen residues in water and sediment. Analytical Letters 2009; 42(2), 1747-1760.
AA Basheer. Chemical chiral pollution: Impact on the society and science and need of the regulations in the 21st century. Chirality 2018; 30(4), 402-406.
AA Basheer and I Ali. Stereoselective uptake and degradation of (±)-o,p-DDD pesticide stereomers in water-sediment system. Chirality 2018; 30(9), 1088-1095.
S Karoui, RB Arfi, K Mougin, A Ghorbal, AA Assadi and A Amrane. Synthesis of novel biocomposite powder for simultaneous removal of hazardous ciprofloxacin and methylene blue: Central composite design, kinetic and isotherm studies using Brouers-Sotolongo family models. Journal of Hazardous Materials 2020; 387, 121675.
ZH Mussa, LR Al-Ameer, FF Al-Qaim, IF Deyab, H Kamyab and S Chelliapan. A comprehensive review on adsorption of methylene blue dye using leaf waste as a bio-sorbent: Isotherm adsorption, kinetics, and thermodynamics studies. Environmental Monitoring and Assessment 2023; 195(8), 940.
RS Bangari, A Yadav, J Bharadwaj and N Sinha. Boron nitride nanosheets incorporated polyvinylidene fluoride mixed matrix membranes for removal of methylene blue from aqueous stream. Journal of Environmental Chemical Engineering 2022; 10(1), 107052.
A Ulu, M Alpaslan, A Gultek and B Ates. Eco-friendly chitosan/κ-carrageenan membranes reinforced with activated bentonite for adsorption of methylene blue. Materials Chemistry and Physics 2022; 278, 125611.
SS Vedula and GD Yadav. Wastewater treatment containing methylene blue dye as pollutant using adsorption by chitosan lignin membrane: Development of membrane, characterization and kinetics of adsorption. Journal of the Indian Chemical Society 2022; 99(1), 100263.
H Raval, R Sharma and A Srivastava. Novel protocol for fouling detection of reverse osmosis membrane based on methylene blue colorimetric method by image processing technique. Water Science and Technology 2024; 89(3), 513-528.
A Hasnaoui, M Chikhi, F Balaska, W Seraghni, M Boussemghoune and N Dizge. Electrocoagulation employing recycled aluminum electrodes for methylene blue remediation. Desalination and Water Treatment 2024; 319(42), 100453.
G Somasundaram, T Thavamani and S Thangavelu. Integration of sequential electrocoagulation and adsorption for effective removal of colour and total organic carbon in textile effluents and its utilization for seed germination and irrigation. Environmental Science and Pollution Research 2024; 31(21), 30716-30734.
IM Minisy, NA Salahuddin and MM Ayad. Adsorption of methylene blue onto chitosan-montmorillonite/polyaniline nanocomposite. Applied Clay Science 2021; 203(22), 105993.
Y Guo, X Fu, R Liu, M Chu and W Tian. Efficient green photocatalyst of Ag/ZnO nanoparticles for methylene blue photodegradation. Journal of Materials Science: Materials in Electronics 2022; 33(5), 1-13.
EHA Zaid, JC Sin, SM Lam and AR Mohamed. Fabrication of La, Ce co-doped ZnO nanorods for improving photodegradation of methylene blue. Journal of Rare Earths 2024; 42(1), 76-83.
M Aravind, A Ahmad, I Ahmad, M Amalanathan, K Naseem, SMM Mary, C Parvathiraja, S Hussain, TS Algarni, M Pervaiz and M Zuber. Critical green routing synthesis of silver NPs using jasmine flower extract for biological activities and photocatalytical degradation of methylene blue. Journal of Environmental Chemical Engineering 2021; 9(1), 104877.
FE Koc and TG Altıncekic. Investigation of gelatin/chitosan as potential biodegradable polymer films on swelling behavior and methylene blue release kinetics. Polymer Bulletin 2021; 78(2), 3383-3398.
K Wu, M Shi, X Pan, J Zhang, X Zhang, T Shen and Y Tian. Decolourization and biodegradation of methylene blue dye by a ligninolytic enzyme-producing Bacillus thuringiensis: Degradation products and pathway. Enzyme and Microbial Technology 2022; 156, 109999.
KE Abbadi, F Echabbi, Y Ouzidan, AK Idrissi, R Lakhmiri and M Safi. The study on the use of a biocatalyst based on Calcined Cow Teeth-TiO2 composite in degrading the methylene blue dye. Results in Chemistry 2023; 6(1), 101027.
X Dong, Y Lin, G Ren, Y Ma and L Zhao. Catalytic degradation of methylene blue by fenton-like oxidation of ce-doped MOF. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2021; 608, 125578.
M Babar, HMS Munir, A Nawaz, N Ramzan, U Azhar, M Sagir, MS Tahir, A Ikhlaq, SNHM Azmin, M Mubashir, KS Khoo and KW Chew. Comparative study of ozonation and ozonation catalyzed by Fe-loaded biochar as catalyst to remove methylene blue from aqueous solution. Chemosphere 2022; 307(P1), 135738.
F Javed, A Tariq, A Ikhlaq, OS Rizvi, U Ikhlaq, UY Qazi and F Qi. Application of laboratory-grade recycled borosilicate glass coated with iron and cobalt for the removal of methylene blue by catalytic ozonation process. Arabian Journal for Science and Engineering 2023; 48, 8753-8768.
KO Iwuozor, JO Ighalo, LA Ogunfowora, AG Adeniyi and CA Igwegbe. An empirical literature analysis of adsorbent performance for methylene blue uptake from aqueous media. Journal of Environmental Chemical Engineering 2021; 9(4), 105658.
P Kaur, Kalpana, S Kumar, A Kumar and A Kumar. Effect of various sodium hydroxide treatment parameters on the adsorption efficiency of rice husk for removal of methylene blue from water. Emergent Materials 2023; 6(10), 1809-1824.
R Coşkun, S Savcı and A Delibaş. Fast removal of methylene blue (MB) with functionalized resin. Journal of Macromolecular Science Part A 2019; 56(7), 1-11.
EC Peres, JC Slaviero, AM Cunha, AH Bandegharaei and GL Dotto. Microwave synthesis of silica nanoparticles and its application for methylene blue adsorption. Journal of Environmental Chemical Engineering 2018; 6(1), 649-659.
PO Oladoye, TO Ajiboye, EO Omotola and OJ Oyewola. Methylene blue dye: Toxicity and potential elimination technology from wastewater. Results in Engineering 2022; 16(6), 100678.
ZA ALOthman, AY Badjah and I Ali. Facile synthesis and characterization of multi walled carbon nanotubes for fast and effective removal of 4‑tert‑octylphenol endocrine disruptor in water. Journal of Molecular Liquids 2019; 275(12), 41-48.
T Liu, Y Li, Q Du, J Sun, Y Jiao, G Yang, Z Wang, Y Xia, W Zhang, K Wang, H Zhu and D Wu. Adsorption of methylene blue from aqueous solution by graphene. Colloids and Surfaces B: Biointerfaces 2012; 90(1), 197-203.
I Ali, AV Babkin, IV Burakova, AE Burakov, EA Neskoromnaya, AG Tkachev, S Panglisch, N AlMasoud and TS Alomar. Fast removal of samarium ions in water on highly efficient nanocomposite-based graphene oxide modified with polyhydroquinone: Isotherms, kinetics, thermodynamics and desorption. Journal of Molecular Liquids 2021; 329(2), 115584.
S Ali, Y Abbas, Z Zuhra and IS Butler. Synthesis of γ-alumina (Al2O3) nanoparticles and their potential for use as an adsorbent in the removal of methylene blue dye from industrial wastewater. Nanoscale Advances 2019; 1(1), 213-218.
X Pan, M Zhang, H Liu, S Ouyang, N Ding and P Zhang. Adsorption behavior and mechanism of acid orange 7 and methylene blue on self-assembled three-dimensional MgAl layered double hydroxide: Experimental and DFT investigation. Applied Surface Science 2020; 522, 146370.
M Aaddouz, K Azzaoui, N Akartasse, E Mejdoubi, B Hammouti, M Taleb, R Sabbahi and SF Alshahateet. Removal of methylene blue from aqueous solution by adsorption onto hydroxyapatite nanoparticles. Journal of Molecular Structure 2023; 1288, 135807.
S Husien, RM El-taweel, AI Salim, IS Fahim, LA Said and AG Radwan. Review of activated carbon adsorbent material for textile dyes removal: Preparation, and modelling. Current Research in Green and Sustainable Chemistry 2022; 5(1), 100325.
MRD Silva, FS Bragagnolo, RL Carneiro, IOC Pereira, JAA Ribeiro, CM Rodrigues, RE Jelley, B Fedrizzi and CS Funari. Metabolite characterization of fifteen by-products of the coffee production chain: From farm to factory. Food Chemistry 2022; 369, 130753.
KT Lee, YT Shih, S Rajendran, YK Park and WH Chen. Spent coffee ground torrefaction for waste remediation and valorization. Environmental Pollution 2023; 324, 121330.
C Setter, FA Borges, CR Cardoso, RF Mendes and TJP Oliveira. Energy quality of pellets produced from coffee residue: Characterization of the products obtained via slow pyrolysis. Industrial Crops and Products 2020; 154, 112731.
LL Kang, YN Zeng, YT Wang, JG Li, FP Wang, YJ Wang, Q Yu, XM Wang, R Ji, D Gao and Z Fang. Removal of pollutants from wastewater using coffee waste as adsorbent: A review. Journal of Water Process Engineering 2022; 49, 103178.
JG Kim, HB Kim and K Baek. Novel electrochemical method to activate biochar derived from spent coffee grounds for enhanced adsorption of lead (Pb). Science of The Total Environment 2023; 886, 163891.
SS Choi, TR Choi and HJ Choi. Surface modification of phosphoric acid-activated carbon in spent coffee grounds to enhance Cu (II) adsorption from aqueous solutions. Applied Chemistry for Engineering 2021; 85(4), 1218-1234.
R Campbell, B Xiao and C Mangwandi. Production of activated carbon from spent coffee grounds (SCG) for removal of hexavalent chromium from synthetic wastewater solutions. Journal of Environmental Management 2024; 366(7), 121682.
I Loulidi, F Boukhlifi, M Ouchabi, A Amar, M Jabri, A Kali and F Aziz. Kinetic. Isotherm and mechanism investigations of the removal of basic violet 3 from water by raw spent coffee grounds. Physical Chemistry Research 2020; 8(3), 869-884.
OU Trakoolsa and W Yoochatchaval. Adsorption efficiency of copper and nickel by activated carbon from coffee ground. EnvironmentAsia 2020; 13(1), 46-53.
DW Cho, K Yoon, EE Kwon, JK Biswas and H Song. Fabrication of magnetic biochar as a treatment medium for As (V) via pyrolysis of FeCl3-pretreated spent coffee ground. Environmental Pollution 2017; 229, 942-949.
L Giraldo and JCM Piraján. Synthesis of activated carbon mesoporous from coffee waste and its application in adsorption zinc and mercury ions from aqueous solution. Journal of Chemistry 2012; 9(2), 938-948.
D Azzouni, EMS Hassani, Z Rais and M Taleb. An excellent alternative to industrial activated carbons for the purification of textile water elaborated from waste coffee grounds. International Journal of Environmental Research 2022; 16(5), 89.
T Wirawan, S Koesnarpadi and NT Widodo. Study of rhodamine B adsorption onto activated carbon from spent coffee grounds. AIP Conference Proceedings 2020; 2237(1), 020007.
JW Lim, KY Lam, MJK Bashir, YF Yeong, MK Lam and YC Ho. Spent coffee grounds-based activated carbon preparation for sequestering of malachite green. AIP Conference Proceedings 2016; 1787(1), 040008.
R Lafi, I Montasser and A Hafiane. Adsorption of congo red dye from aqueous solutions by prepared activated carbon with oxygen-containing functional groups and its regeneration. Adsorption Science & Technology 2018; 37(1-2), 160-181.
I Loulidi, M Jabri, A Amar, A Kali, A Alrashdi, C Hadey, M Ouchabi, PS Abdullah, H Lgaz, Y Cho and F Boukhlifi. Comparative study on adsorption of crystal violet and chromium (VI) by activated carbon derived from spent coffee grounds. Applied Sciences 2023; 13(2), 985.
E Pagalan Jr, M Sebron, S Gomez, SJ Salva, R Ampusta, AJ Macarayo, C Joyno, A Ido and R Arazo. Activated carbon from spent coffee grounds as an adsorbent for treatment of water contaminated by aniline yellow dye. Industrial Crops and Products 2020; 145(25004), 111953.
H Laksaci, A Khelifi, B Belhamdi and M Trari. The use of prepared activated carbon as adsorbent for the removal of orange G from aqueous solution. Microchemical Journal 2019; 145, 908-913.
I Block, C Günter, AD Rodrigues, S Paasch, P Hesemann and A Taubert. Carbon adsorbents from spent coffee for removal of methylene blue and methyl orange from water. Materials 2021; 14(14), 3996.
VT Nguyen, TB Nguyen, CP Huang, CW Chen, XT Bui and CD Dong. Alkaline modified biochar derived from spent coffee ground for removal of tetracycline from aqueous solutions. Journal of Water Process Engineering 2021; 40, 101908.
M El-Azazy, AS El-Shafie and H Morsy. Biochar of spent coffee grounds as per Se and impregnated with TiO2: Promising waste-derived adsorbents for Balofloxacin. Molecules 2021; 26(8), 2295.
A Lykoudi, Z Frontistis, J Vakros, ID Manariotis and D Mantzavinos. Degradation of sulfamethoxazole with persulfate using spent coffee grounds biochar as activator. Journal of Environmental Management 2020; 271, 111022.
A Hgeig, M Novaković and I Mihajlović. Sorption of carbendazim and linuron from aqueous solutions with activated carbon produced from spent coffee grounds: Equilibrium, kinetic and thermodynamic approach. Journal of Environmental Science and Health, Part B 2019; 54(4), 226-236.
MHL Bergamini, SBD Oliveira and PS Scalize. Production of activated carbon from exhausted coffee grounds chemically modified with natural eucalyptus ash lye and its use in the fluoride adsorption process. Environmental Science and Pollution Research 2023; 30(39), 91276-91291.
S Zhang, X Huo, S Xu, Y Zhang, B Zhang, M Wang, Q Wang and J. Zhang. Original sulfur-doped carbon materials synthesized by coffee grounds for activating persulfate to BPA degradation: The key role of electron transfer. Process Safety and Environmental Protection 2022; 168, 1219-1234.
N Patel, AL Srivastav, A Patel, A Singh, SK Singh, K Chaudhary, PK Singh and B Bhunia. Nitrate contamination in water resources, human health risks and its remediation through adsorption: A focused review. Environmental Science and Pollution Research 2022; 29(4), 69137-69152.
MJ Ahmed, BH Hameed and MA Khan. Recent advances on activated carbon-based materials for nitrate adsorption: A review. Journal of Analytical and Applied Pyrolysis 2023; 169(7), 105856.
H Zhao, H Zhong, Y Jiang, H Li, P Tang, D Li and Y Feng. Porous ZnCl2-activated carbon from shaddock peel: Methylene blue adsorption behavior. Materials 2022; 15(3), 895.
M Lauberts, I Mierina, M Pals, MAA Latheef and A Shishkin. Spent coffee grounds valorization in biorefinery context to obtain valuable products using different extraction approaches and solvents. Plants 2023; 12(1), 30.
A Namane, A Mekarzia, K Benrachedi, N Belhaneche-Bensemra and A Hellal. Determination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl2 and H3PO4. Journal of Hazardous Materials 2005; 119(1-3), 189-194.
VT Le, TM Pham, VD Doan, OE Lebedeva and HT Nguyen. Removal of Pb(ii) ions from aqueous solution using a novel composite adsorbent of Fe3O4/PVA/spent coffee grounds. Separation Science and Technology 2019; 54(18), 1-12.
DN Mengesha, MW Abebe, RA Ntiamoah and H Kim. Ground coffee waste-derived carbon for adsorptive removal of caffeine: Effect of surface chemistry and porous structure. Science of The Total Environment 2022; 818, 151669.
LM Madikizela and VE Pakade. Trends in removal of pharmaceuticals in contaminated water using waste coffee and tea-based materials with their derivatives. Water Environment Research 2023; 95(4), e10857.
VT Le, TKN Tran, NK Dang, VD Doan, VA Tran, Y Vasseghian and TM Aminabhavi. Mn3O4/activated carbon nanocomposites for adsorptive removal of methylene blue. Chemical Engineering Journal 2023; 474, 145903.
HM Alraddadi, TM Fagieh, EM Bakhsh, K Akhtar, SB Khan, SA Khan, EA Bahaidarah and TA Homdi. Adsorptive removal of heavy metals and organic dyes by sodium alginate/coffee waste composite hydrogel. International Journal of Biological Macromolecules 2023; 274, 125708.
D Chen, Y Yin, Y Xu and C Liu. Adsorptive recycle of phosphate by MgO-biochar from wastewater: Adsorbent fabrication, adsorption site energy analysis and long-term column experiments. Journal of Water Process Engineering 2023; 51, 103445.
H Laksaci, B Belhamdi, O Khelifi, A Khelifi and M Trari. Elimination of amoxicillin by adsorption on coffee waste based activated carbon. Journal of Molecular Structure 2023; 1274(P1), 134500.
W Ma, J Fan, X Cui, Y Wang, Y Yan, Z Meng, H Gao, R Lu and W Zhou. Pyrolyzing spent coffee ground to biochar treated with H3PO4 for the efficient removal of 2,4-dichlorophenoxyacetic acid herbicide: Adsorptive behaviors and mechanism. Journal of Environmental Chemical Engineering 2023; 11(1), 109165.
Nurmayasari, SA Kurniasari, S Sholihun and AD Nugraheni. The effectiveness of coffee waste ground by simple washing on the adsorption of methylene blue. Key Engineering Materials 2023; 949, 103-109.
J Wang and X Guo. Adsorption kinetic models: Physical meanings, applications, and solving methods. Journal of Hazardous Materials 2020; 390, 122156.
NNR do Nascimento, ALM da T Silva, WL Silva and MGF Rodrigues. Valorization of coffee agro-industrial residue for biochar production: Use as adsorbent for methylene blue removal. Desalination and Water Treatment 2024; 320, 100767.
DI Çifçi and N Aydın. Comparison of H3PO4 and ZnCl2 activated filtered coffee waste carbon-based adsorbents in methylene blue removal by using ultrasonic-assisted adsorption. Arabian Journal for Science and Engineering 2023; 48(73), 8641-8653.
M Cuccarese, S Brutti, A De Bonis, R Teghil, FD Capua, IM Mancini, S Masi and D Caniani. Sustainable adsorbent material prepared by soft alkaline activation of spent coffee grounds: Characterisation and adsorption mechanism of methylene blue from aqueous solutions. Sustainability 2023; 15(3), 2454.
MS Akindolie and HJ Choi. Surface modification of spent coffee grounds using phosphoric acid for enhancement of methylene blue adsorption from aqueous solution. Water Science & Technology 2022; 85(4), 1218-1234.
A Bouzidi, M Djedid, CAdM Benalia, B Hafez and H Elmsellem. Biosorption of Co (II) ions from aqueous solutions using selected local luffa cylindrica: Adsorption and characterization studies. Moroccan Journal of Chemistry 2021; 9(1), 156-167.
J Hou, C Cao, F Idrees and X Ma. Hierarchical porous nitrogen-doped carbon nanosheets derived from silk for ultrahigh-capacity battery anodes and supercapacitors. ACS Nano 2015; 9(3), 2556–2564.
N Kumar, A Pandey, Rosy and YC Sharma. A review on sustainable mesoporous activated carbon as adsorbent for efficient removal of hazardous dyes from industrial wastewater. Journal of Water Process Engineering 2023; 54, 104054.
H Zhao, H Zhong, Y Jiang, H Li, P Tang, D Li and Y Feng. Porous ZnCl2-activated carbon from shaddock peel: Methylene blue adsorption behavior. Materials 2022; 15(3), 895.
A Ahmadpour and DD Do. The preparation of activated carbon from macadamia nutshell by chemical activation. Carbon 1997; 32(12), 1723-1732.
MA Hubbe, S Azizian and S Douven. Implications of apparent pseudo-second-order adsorption kinetics onto cellulosic materials: A review. BioResources 2019; 14(3), 7582-7626.
Q Zhang, Y Wu, S Dong, J Zhuo, X Sun, and Q Yao. Development of activated carbon/CaCl2 composites for seasonal thermochemical energy storage: Effect of pore structure. Journal of Energy Storage 2024; 97(A), 112697.
M Fan, Y. Shao, Y Wang, J Sun, H He, Y Jiang, S Zhang, Y Wang and X Hu. Preparation of activated carbon with recycled ZnCl2 for maximizing utilization efficiency of the activating agent and minimizing generation of liquid waste. Chemical Engineering Journal 2024; 500, 157278.
S Kundu, T Khandaker, MdAM Anik, MdK Hasan, PK Dhar, SK Dutta, MA Latif and MS Hossain. A comprehensive review of enhanced CO2 capture using activated carbon derived from biomass feedstock. RSC Advances 2024; 40(40), 29693-29736.
T Fischer, H Kungl, H Tempel, A Kretzschmar, V Selmert and RA Eichelab. Post-treatment strategies for pyrophoric KOH activated carbon nanofibers. RSC Advances 2024; 14(6), 3845-3856.
I Pet, MN Sanad, M Farouz, MM ElFaham, AE -Hussein, MSA El-sadek, RA Althobiti, A Ioanid. Review: Recent developments in the implementation of activated carbon as heavy metal removal management. Water Conservation Science and Engineering 2024; 9, 62.
GC Hernández, EP Melián and DE Santiago. Synthesis of activated carbon from sewage sludge using phosphoric acid as activating agent. Environmental Science and Pollution Research 2025. https://doi.org/10.1007/s11356-025-36516-y
YAB Neolaka, ZS Ngara, Y Lawa, JN Naat, DP Benu, A Chetouani, H Elmsellem, H Darmokoesoemo and HS Kusuma. Simple design and preliminary evaluation of continuous submerged solid small-scale laboratory photoreactor (CS4PR) using TiO2/NO3-@TC for dye degradation. Journal of Environmental Chemical Engineering 2019; 7(6),103482.
AT Hoang, S Kumar, E Lichtfouse, CK Cheng, RS Varma, N Senthilkumar, PQP Nguyen and XP Nguyen. Remediation of heavy metal polluted waters using activated carbon from lignocellulosic biomass: An update of recent trends. Chemosphere 2022; 302, 134825.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






