Potential of Syzygium cumini L. Seed as Cosmetic Bioactive Compounds: Matrix Metalloproteinases Inhibition, Antioxidant Activities, Cytotoxicity, and Safety Assessment
DOI:
https://doi.org/10.48048/tis.2025.10833Keywords:
Syzygium cumini L., Antioxidant activity, Matrix metalloproteinase inhibition, CytotoxicityAbstract
This study systematically evaluated the extraction efficiency, phytochemical content, antioxidant activity, matrix metalloproteinase inhibition, cytotoxicity, stability, and safety of Syzygium cumini L. seed extracts using various solvents. Extraction yields were highest with 50 %v/v ethanol (19.19% ± 0.44%), while the 70 %v/v ethanol extract exhibited the greatest total phenolic (488.51 ± 3.60 mg GAE/g) and flavonoid (88.45 ± 1.61 mg QE/g) contents. Antioxidant assays revealed that the water extract (SC100W) had the strongest DPPH and ABTS radical scavenging activities. In contrast, the 70 %v/v ethanol extract (SC70ET) demonstrated superior ferric reducing antioxidant power, correlating with higher phenolic content. Matrix metalloproteinase inhibition assays identified the 50 %v/v ethanol extract (SC50ET) as the most effective, showing the lowest IC₅₀ for strong collagenase, elastase, and tyrosinase inhibition. All Syzygium cumini L. seed extracts showed over 90% cell viability within a concentration range of 1 - 250 µg/mL, demonstrating that it is safe for skincare application. The active ingredients and efficacy have not significantly decreased, even when stored at high temperatures under accelerated conditions. Microbiological and heavy metal analyses confirmed the extract’s safety for use as an active ingredient in cosmetic products. Collectively, these results highlight the influence of extraction solvent on bioactivity and identify optimal conditions for maximizing cosmeceutical properties of Syzygium cumini L. seed extracts.
HIGHLIGHTS
- Extraction yield was highest with 50 %v/v ethanol (19.19%) and 30 %v/v ethanol (18.40%), while water yielded the lowest, confirming that the solvent significantly affects yield.
- Antioxidant activity varied by assay; 70 %v/v ethanol extract showed the highest phenolic/flavonoid content and FRAP reducing power, but water extract had superior DPPH and ABTS radical scavenging activity.
- Matrix metalloproteinase inhibition was optimal with 50 %v/v ethanol extract, which showed the strongest collagenase, competitive tyrosinase, and elastase inhibition among the tested solvents.
- Syzygium cumini L. seed extract showed cell viability of over 90% with a concentration range of 1-250 micrograms/milliliter.
- The optimal extraction condition overall was 50% v/v ethanol, balancing high yield, potent matrix metalloproteinase inhibition, and strong antioxidant activity, making it the most promising for bioactive applications.
GRAPHICAL ABSTRACT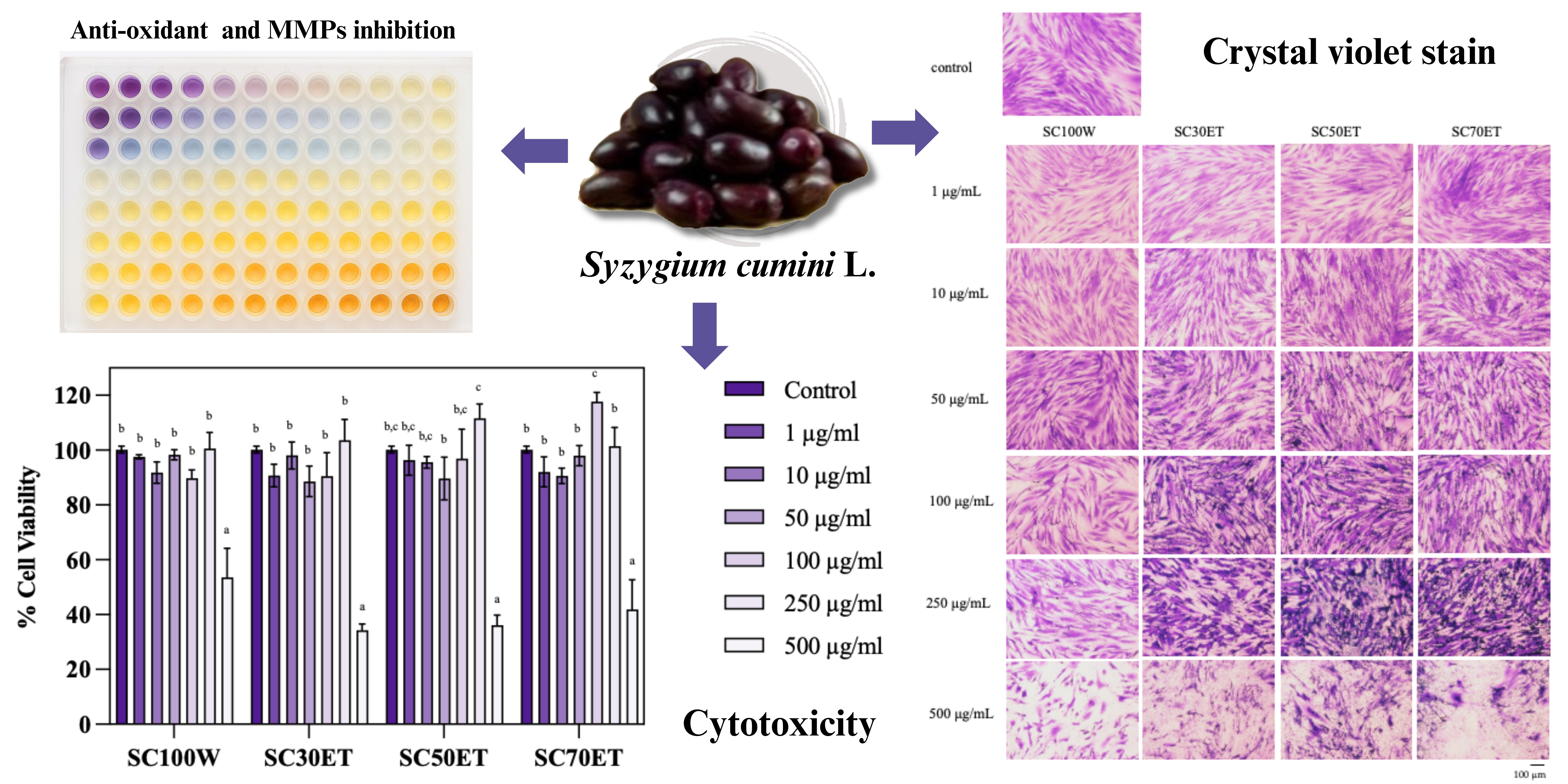
Downloads
References
SB Swami, NSJ Thakor, MM Patil and PM Haldankar. Jamun (Syzygium cumini (L.)): A review of its food and medicinal uses. Food and Nutrition Sciences 2012; 3(8), 1100-1117.
M Qamar, S Akhtar, T Ismail, M Wahid, MW Abbas, MS Mubarak, Y Yuan, R T Barnard, ZM Ziora and T Esatbeyoglu. Phytochemical profile, biological properties, and food applications of the medicinal plant syzygium cumini. Foods 2022; 11(3), 378.
Y Tak, M Kaur, MC Jain, MK Samota, NK Meena, G Kaur, R Kumar, D Sharma, JM Lorenzo and R Amarowicz. Jamun seed: A review on bioactive constituents, nutritional value and health benefits. Polish Journal of Food and Nutrition Sciences 2022; 72(3), 211-228.
S Ahmed, KS Ahmed, MS Hossain, MS Azam, M Rahman and MM Hoque. Proximate composition and antioxidant activity of Syzygium cumini fruit grown at different regions in Bangladesh. Food Research 2020; 4(5), 1693-1699.
ZP Ruan, LL Zhang and YM Lin. Evaluation of the antioxidant activity of Syzygium cumini leaves. Molecules 2008; 13(10), 2545-2556.
T Grimm, A Schafer and P Hogger. Antioxidant activity and inhibition of matrix metalloproteinases by metabolites of maritime pine bark extract (pycnogenol). Free Radical Biology and Medicine 2004; 36(6), 811-822.
SM Kotakadi, MJ Bangarupeta, K Kandati, DPR Borelli, JA Sayyed, MI Shaik and JS Nannepaga. Biosynthesized MgONPs using Syzygium cumini seed extract: Characterization, In vitro anti-oxidant and anti-microbial activity. Biotechnology Reports 2024; 43, e00846.
M Srisayam, C Puengtang, A Srisopa and A Chodnakarin. Biological activities, phenolic and vitamin C contents from Syzygium cumini (L.) extract. PSRU Journal of Science and Technology 2022; 7(2), 103-113.
AK Yadav, S Saraswat, P Sirohi, M Rani, S Srivastava, MP Singh and NK Singh. Antimicrobial action of methanolic seed extracts of Syzygium cumini Linn. on Bacillus subtilis. AMB Express 2017; 7(1), 196.
R Srimoon and S Niyomwan. Development and validation of the folin-ciocalteu semi-micro method for total phenolic compounds determination in rangdaeng (Ventilago denticulata Willd.) extract. Burapha Science Journal 2021; 26(1), 378-398.
M Chatatikun and A Chiabchalard. Phytochemical screening, total flavonoid and total phenolic contents and antioxidant activity of moringa oleifera leaf extracts. Current Topics in Pharmacology 2020; 18(1), 93-99.
S Kamtekar, V Keer and V Patil. Estimation of phenolic content, flavonoid content, antioxidant and alpha amylase inhibitory activity of marketed polyherbal formulation. Journal of Applied Pharmaceutical Science 2014; 4(9), 061-065.
M Minarti, N Idiawati and MA Wibowo. Potential antioxidant activity methods DPPH, ABTS, FRAP, total phenol and total flavonoid of methanol extract and its fractions from macaranga hypoleuca leaves. E3S Web of Conferences 2024; 371, 07005.
R Re, N Pellegrini, A Proteggente, A Pannala, M Yang and C Rice-Evans. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine 1999; 26(9-10), 1231-1237.
O Bulut, IE Kose, C Sonmez and HA Oktem. Phenolic compounds, carotenoids, and antioxidant capacities of a thermo-tolerant Scenedesmus sp. (Chlorophyta) extracted with different solvents. Journal of Applied Phycology 2019; 31(3), 1675-1683.
O Bulut, IE Kose, C Sonmez and HA Oktem. Antioxidant activity of Micractinium sp. (Chlorophyta) extracts against H2O2 induced oxidative stress in human breast adenocarcinoma cells. Scientific Reports 2024; 14(1), 27593.
MM Younis, IM Ayoub, NM Mostafa, MAE Hassab, WM Eldehna, ST Al-Rashood and OA Eldahshan. GC/MS profiling, anti-collagenase, anti-elastase, anti-tyrosinase and anti-hyaluronidase activities of a stenocarpus sinuatus leaves extract. Plants 2022; 11(7), 918.
N Laothaweerungsawat, J Sirithunyalug, W Chaiyana, C Saenjum, N Waranuch, C Chaiyasut, K Pattamapun and S Saokaew. Chemical compositions and anti-skin-ageing activities of Origanum vulgare L. essential oil from tropical and Mediterranean region. Molecules 2020; 25(5), 1101.
W Wang, H Lin, W Shen, X Qin, J Gao, W Cao, H Zheng, Z Chen and Z Zhang. Optimization of a novel tyrosinase inhibitory peptide from atrina pectinata: Identification, molecular docking and enzyme kinetics. Foods 2023; 12(21), 3884.
AA Ala, BB Olotu and CMD Ohia. Assessment of cytotoxicity of leaf extracts of andrographis paniculata and aspilia africana on murine cells in vitro. Archives of Basic and Applied Medicine 2018; 6(1), 61-65.
R Asada, K Kageyama, H Tanaka, M Kimura, Y Saitoh and N Miwa. Carcinostatic effects of diverse ascorbate derivatives in comparison with aliphatic chain moiety structures: Promotion by combined hyperthermia and reduced cytotoxicity to normal cells. Oncology Letters 2012; 3(5), 1042-1046.
S Garg, H Huifu, SC Kaul and R Wadhwa. Integration of conventional cell viability assays for reliable and reproducible read-outs: Experimental evidence. BMC Research Notes 2018; 11(1), 403.
Thai Industrial Standards Institute Ministry of Industry. Thai industrial standard for cosmetics: General requirements (TIS 152-2555). Ministry of Industry, Bangkok, Thailand, 2012.
United States Pharmacopeial Convention. United states pharmacopeia 41–national formulary 36 (USP41/NF36). United States Pharmacopeial Convention, Maryland, 2018.
CAF Artilha-Mesquita, AP Stafussa, PDSD Santos, OO Santos, SCD Costa and GS Madrona. Extraction of bioactive compounds from the fruits of jambolan (Syzygium cumini (L.)) using alternative solvents. Plants 2024; 13(15), 2065.
M Faradilla, I Fidrianny and MI Iwo. Antioxidant and immunomodulatory activities of ethanol extracts from Syzygium cumini L. skeels and pogostemon cablin Benth. Narra Journal 2024; 4(3), e918.
R Lodhi, M Yadav, S Jain and S Nayak. Phytochemical investigation and comparative study on percentage yield with different solvent of “syzygium cumini” leaf. Journal of Emerging Technologies and Innovative Research 2023; 10(8), f587-f597.
SH Priya, N Prakasan and J Purushothaman. Antioxidant activity, phenolic-flavonoid content and high-performance liquid chromatography profiling of three different variants of Syzygium cumini seeds: A comparative study. Journal of Intercultural Ethnopharmacology 2017; 6(1), 107-114.
M Platzer, S Kiese, T Herfellner, U Schweiggert-Weisz, O Miesbauer and P Eisner. Common trends and differences in antioxidant activity analysis of phenolic substances using single electron transfer based assays. Molecules 2021; 26(5), 1244.
C Vieito, E Fernandes, MV Velho and P Pires. The effect of different solvents on extraction yield, total phenolic content and antioxidant activity of extracts from pine bark (Pinus pinaster subsp. atlantica). Chemical Engineering Transactions 2018; 64, 127-132.
TV Tung, NH Duy, PLK My, NTT Thuy, TPM Nam, LT Hue and TQ Minh. Optimization of phenolic and flavonoid extraction from durian peel: Effects of solvent type, extraction parameters, and antioxidant activity evaluation. Journal of Xi’an Shiyou University, Natural Science Edition 2025; 21(1), 161-179.
QD Do, AE Angkawijaya, PL Tran-Nguyen, LH Huynh, FE Soetaredjo, S Ismadji and H Ju. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. Journal of Food and Drug Analysis 2014; 22(3), 296-302.
J Chen, J Yang, L Ma, J Li, N Shahzad and CK Kim. Structure-antioxidant activity relationship of methoxy, phenolic hydroxyl, and carboxylic acid groups of phenolic acids. Scientific Reports 2020; 10(1), 2611.
S Baliyan, R Mukherjee, A Priyadarshini, A Vibhuti, A Gupta, RP Pandey and CM Chang. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of ficus religiosa. Molecules 2022; 27(4), 1326.
SB Kedare and RP Singh. Genesis and development of DPPH method of antioxidant assay. Journal of Food Science and Technology 2011; 48(4), 412-422.
S Rana, NMA Rayhana, SH Emon, T Islam, K Rathry, M Hasana, MI Mansura, BC Srijona, S Islamb, A Raya, A Rakib, A Islamc, KE Zahana, F Hossena and A Asraf. Antioxidant activity of Schiff base ligands using the DPPH scavenging assay: An updated review. RSC Advances 2024; 14(45), 33094-33123.
IR Ilyasov, VL Beloborodov, IA Selivanova and RP Terekhov. ABTS/PP decolorization assay of antioxidant capacity reaction pathways. International Journal of Molecular Sciences 2020; 21(3), 1131.
A Kiss, VA Papp, A Pal, J Prokisch, S Mirani, BE Toth and T Alshaal. Comparative study on antioxidant capacity of diverse food matrices: Applicability, suitability and inter-correlation of multiple assays to assess polyphenol and antioxidant status. Antioxidants 2025; 14(3), 317.
RPP Fernandes, MA Trindade, FG Tonin, CG Lima, SM Pugine, PE Munekata, JM Lorenzo and MPD Melo. Evaluation of antioxidant capacity of 13 plant extracts by three different methods: cluster analyses applied for selection of the natural extracts with higher antioxidant capacity to replace synthetic antioxidant in lamb burgers. Journal of Food Science and Technology 2016; 53(1), 451-460.
NS Rajurkar and SM Hande. Estimation of phytochemical content and antioxidant activity of some selected traditional Indian medicinal plants. Indian Journal of Pharmaceutical Sciences 2011; 73(2), 146-151.
SJ Hwang and JH Lee. Comparison of antioxidant activities expressed as equivalents of standard antioxidant. Food Science and Technology, Campinas 2023; 43, e121522.
J Junlatat, N Fangkrathok and B Sripanidkulchai. Antioxidative and melanin production inhibitory effects of Syzygium cumini (L.) Skeels extracts. Songklanakarin Journal of Science and Technology 2018; 40(5), 1136-1143.
RM Lima, HC Polonini, MAF Brandao, FJ Raposo, RC Dutra and NRB Raposo. In vitro assessment of anti-aging properties of Syzygium cumini (L.) leaves extract. International Journal of Pharmaceutical Sciences and Research 2022; 13(4), 10185-10191.
B Deri, M Kanteev, M Goldfeder, D Lecina, V Guallar, N Adir and A Fishman. The unravelling of the complex pattern of tyrosinase inhibition. Scientific Reports 2016; 6, 34993.
S Zolghadri, A Bahrami, MTH Khan, J Munoz-Munoz, F Garcia-Molina, F Garcia-Canovas and AA Saboury. A comprehensive review on tyrosinase inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry 2019; 34(1), 279-309.
F Liang. Inhibition mechanism investigation of quercetagetin as a potential tyrosinase inhibitor. Frontiers in Chemistry 2024; 12, 1411801.
MN Masum, K Yamauchi and T Mitsunaga. Tyrosinase inhibitors from natural and synthetic sources as skin-lightening agents. Reviews in Agricultural Science 2019; 7, 41-58.
NS Ashmawy, HA Gad and HAS El-Nashar. Comparative study of essential oils from different organs of Syzygium cumini (Pamposia) based on GC/MS chemical profiling and in vitro antiaging activity. Molecules 2023; 28(23), 7861.
N Atale, CB Mishra, S Kohli, RK Mongre, A Prakash, S Kumari, UCS Yadav, R Jeon and V Rani. Anti-inflammatory effects of S. cumini seed extract on gelatinase-B (MMP-9) regulation against hyperglycemic cardiomyocyte stress. Oxidative Medicine and Cellular Longevity 2021; 2021, 8839479.
Q Lei, SH Alotaibi and D Yang. Anti-small cell lung cancer and collagenase inhibition properties of hydroxysafflor yellow A. Archives of Medical Science 2021. https://doi.org/10.5114/aoms/138833
S Ahmad, M Saleem, N Riaz, Y S Lee, R Diri, A Noor, D Almasri, A Bagalagel and MF Elsebai. The natural polypeptides as significant elastase inhibitors. Frontiers in Pharmacology 2020; 11, 688.
BD Vanjare, YES Eom, H Raza, M Hassan, KH Lee and SJ Kim. Elastase inhibitory activity of quinoline Analogues: Synthesis, kinetic mechanism, cytotoxicity, chemoinformatics and molecular docking studies. Bioorganic & Medicinal Chemistry 2022; 63, 116745.
RJRS Thanapaul, CK Nambur and K Giriraj. Development of multi-herbal formulation with enhanced antimicrobial, antioxidant, cytotoxic, and antiaging properties. Journal of the Indian Chemical Society 2024; 101(11), 101402.
M Prasathkumar, S Anisha, A Khusro, MM Essa, SB Chidambaram, MW Qoronfleh, S Sadhasivam, MUK Sahibzada, S Alghamdi, M Almehmadi, O Abdulaziz, MU Khandaker, MRI Faruque and TB Emran. Anti-pathogenic, anti-diabetic, anti-inflammatory, antioxidant, and wound healing efficacy of datura metel L. leaves. Arabian Journal of Chemistry 2022; 15(9), 104112.
SN Silva, IC Abreu, GFC Silva, RM Ribeiro, AS Lopes, MSS Cartagenes, SMF Freire, ACR Borges and MOR Borges. The toxicity evaluation of Syzygium cumini leaves in rodents. Revista Brasileira de Farmacognosia 2012; 22(1), 102-108.
T Alide, P Wangila and A Kiprop. Effect of cooking temperature and time on total phenolic content, total flavonoid content and total in vitro antioxidant activity of garlic. BMC Research Notes 2020; 13(1), 564.
I Esparza, MJ Cimminelli, JA Moler, N Jimenez-Moreno and C Ancin-Azpilicueta. Stability of phenolic compounds in grape stem extracts. Antioxidants 2020; 9(8), 720.
C Wan, Y Yu, S Zhou, W Liu, S Tian and S Cao. Antioxidant activity and free radical-scavenging capacity of Gynura divaricata leaf extracts at different temperatures. Pharmacognosy Magazine 2011; 7(25), 40-45.

Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






