Tacca chantrieri Rhizome Extract Induces Apoptosis and Inhibits Colony Formation and Migration in Triple-Negative, HER2+, and ER+ Breast Cancer Cell Lines
DOI:
https://doi.org/10.48048/tis.2025.10525Keywords:
Tacca chantrieri, Breast cancer, Anti-cancer agent, Apoptosis, Cell migration inhibitionAbstract
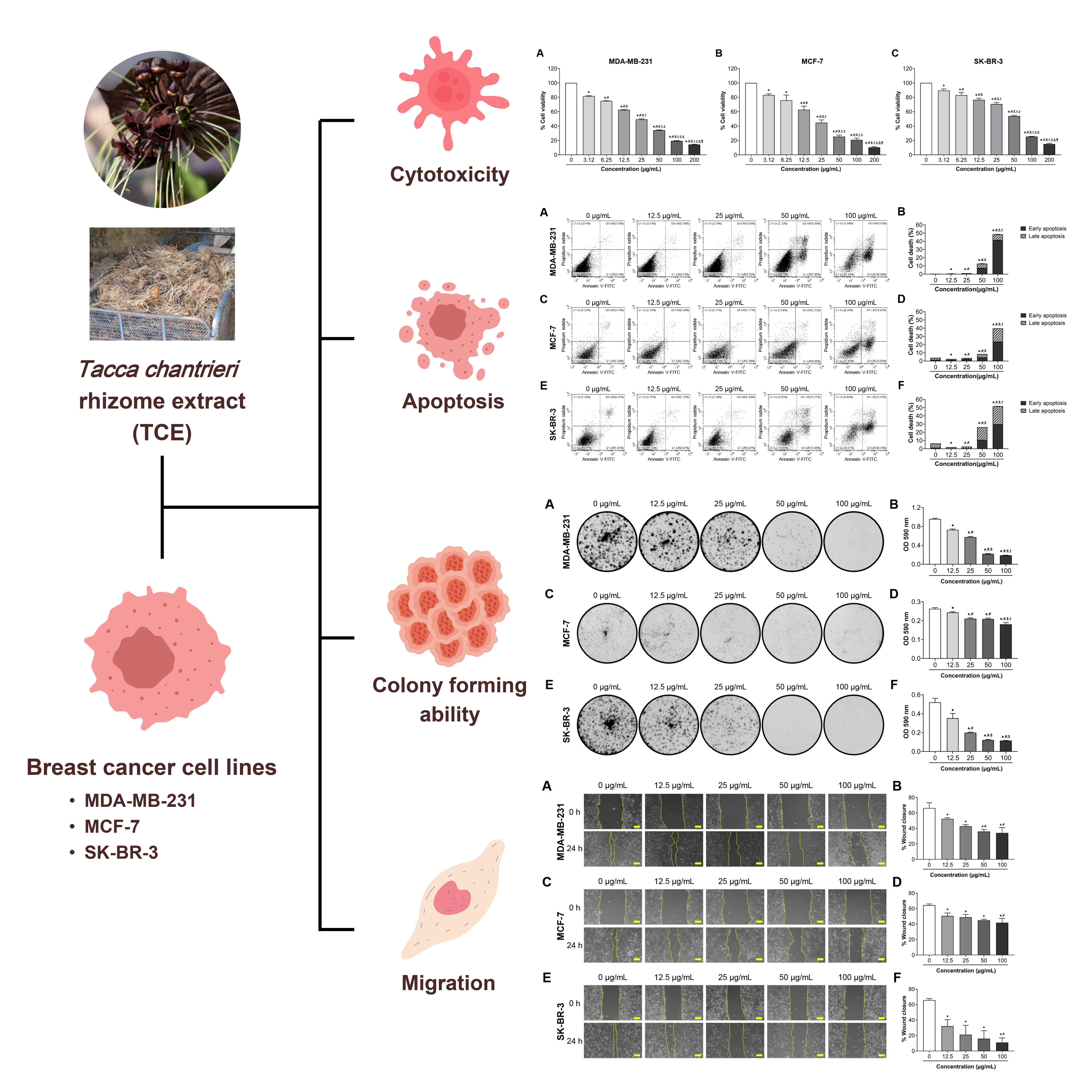
Breast cancer is the most common cancer in females worldwide, and standard chemotherapy often causes significant side effects. Tacca chantrieri, a plant used in traditional medicine, contains bioactive compounds with anti-cancer properties. This study aimed to evaluate the anti-cancer activity of T. chantrieri rhizome extract (TCE) on breast cancer cell lines. The rhizomes of T. chantrieri were extracted with 95% ethanol and used to treat MDA-MB-231, MCF-7, and SK-BR-3 breast cancer cell lines. Cell viability was assessed using the MTT assay. Apoptosis was evaluated by annexin V and propidium iodide staining, while colony formation and cell migration were assessed using clonogenic and wound healing-scratch assays, respectively. The results showed that TCE significantly reduced cell viability, induced apoptosis, and inhibited colony formation and migration. In conclusion, TCE exhibits anti-cancer activity against breast cancer cells, highlighting its potential for further development as an alternative treatment for breast cancer.
HIGHLIGHTS
- T. chantrieri rhizome extract (TCE) exhibited anti-cancer breast activity.
- TCE reduced the viability of MDA-MB-231, MCF-7, and SK-BR-3 cell lines.
- TCE induced apoptosis of MDA-MB-231, MCF-7, and SK-BR-3 cell lines.
- TCE inhibited colony formation and migration of MDA-MB-231, MCF-7, and SK-BR-3 cell lines.
GRAPHICAL ABSTRACT
Downloads
References
F Bray, M Laversanne, H Sung, J Ferlay, Siegel RL, I Soerjomataram and A Jemal. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a Cancer Journal for Clinicians 2024; 74(3), 229-263.
X Xiong, LW Zheng, Y Ding, YF Chen, YW Cai, LP Wang, L Huang, CC Liu, ZM Shao and KD Yu. Breast cancer: Pathogenesis and treatments. Signal Transduction and Targeted Therapy 2025; 10(1), 49.
J Zhang, Y Wu, Y Li, S Li, J Liu, X Yang, G Xia and G Wang. Natural products and derivatives for breast cancer treatment: From drug discovery to molecular mechanism. Phytomedicine 2024; 129, 155600.
WJJ Chan, J Adiwidjaja, AJ McLachlan, AV Boddy and JE Harnett. Interactions between natural products and cancer treatments: underlying mechanisms and clinical importance. Cancer Chemotherapy and Pharmacology 2023; 91(2), 103-119.
Y Yang, Q Gong, W Wang, YL Mao, XR Wang, S Yao, HY Zhang, C Tang and Y Ye. Neuroprotective and anti-inflammatory ditetrahydrofuran-containing diarylheptanoids from Tacca chantrieri. Journal of Natural Products 2020; 83(12), 3681-3688.
R Charoensub, D Thiantong and S Phansiri. Micropropagation of bat flower plant, Tacca chantrieri Andre. Agriculture and Natural Resources 2008; 42(5), 7-12.
K Keardrit, C Rujjanawate and D Amornlerdpison. Analgesic, antipyretic and anti-inflammatory effects of Tacca chantrieri Andre. Journal of Medicinal Plants Research 2010; 4(19), 1991-1995.
R Tiamjan, A Panthong, T Taesotikul, C Rujjanawate, WC Taylor and D Kanjanapothi. Hypotensive activity of Tacca chantrieri and its hypotensive principles. Pharmaceutical Biology 2007; 45(6), 481-485.
U Kamsrijai, B Thaweethee-Sukjai, N Teerapattarakan, K Wanchai and P Jirarattanawan. Neuroprotective effects of Tacca chantrieri Andre against lipopolysaccharide-induced cognitive impairment and neuroinflammation. Physiology and Pharmacology 2023; 27(3), 296-306.
T Hawiset, N Sriraksa, S Anukanon, U Kamsrijai, S Praman, N Teerapattarakan and P Inkaew. Tacca chantrieri André rhizome extract alleviates scopolamine-induced cognitive impairment and neuroinflammation in rats. Advances in Pharmacological and Pharmaceutical Sciences 2025; 2025(1), 7334303.
A Yokosuka, Y Mimaki and Y Sashida. Spirostanol saponins from the rhizomes of Tacca chantrieri and their cytotoxic activity. Phytochemistry 2002; 61(1), 73-78.
G Ni, HZ Yang, NJ Fu, LL Zhang, MC Wang, J Chen, CL Zhang, Y Li, XG Chen, RY Chen and DQ Yu. Cytotoxic taccalonolides and withanolides from Tacca chantrieri. Planta Medica 2015; 81(3), 247-256.
A Yokosuka, Y Mimaki, H Sakagami and Y Sashida. New diarylheptanoids and diarylheptanoid glucosides from the rhizomes of Tacca chantrieri and their cytotoxic activity. Journal of Natural Products 2002; 65(3), 283-289.
N Armartmuntree, Y Kittirat, B Promraksa, W Loilome, H Dokduang, A Techasen, P Sansomchai, M Thanee,T O’Connor, P Kongthitilerd and S Padthaisong. Antioxidative and anticancer effects of Tacca chantrieri extract enhancing cisplatin sensitivity in cholangiocarcinoma cells. PLoS One 2025; 20(1), e0317111.
S Aluksanasuwan, K Somsuan, J Ngoenkam, W Chiangjong, A Rongjumnong, A Morchang, A Morchang, S Chutipongtanate and S Pongcharoen. Knockdown of heat shock protein family D member 1 (HSPD1) in lung cancer cell altered secretome profile and cancer-associated fibroblast induction. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2024; 1871(5), 119736.
S Aluksanasuwan, K Somsuan, W Chiangjong, A Rongjumnong, W Jaidee, N Rujanapun, S Chutipongtanate, S Laphookhieo and R Charoensup. SWATH-proteomics reveals Mathurameha, a traditional anti-diabetic herbal formula, attenuates high glucose-induced endothelial dysfunction through the EGF/NO/IL-1β regulatory axis. Journal of Proteomics 2024; 306, 105263.
A Morchang, K Somsuan, A Rongjumnong, C Weeraphan, S Pramual, T Ruangjaroon, D Chokchaichamnankit, W Jaidee, P Hankittichai, C Pramvichai, R Charoensup, A Wanta, C Srisomsap, J Svasti and S Aluksanasuwan. Proteomic analysis reveals a potential anticancer mechanism for a novel effervescent curcumin-ascorbic acid-polysaccharide-β-cyclodextrin inclusion complex towards colorectal cancer cells through modulation of ribosome biogenesis. Carbohydrate Polymer Technologies and Applications 2025; 10, 100775.
K Somsuan, A Rongjumnong, A Morchang, P Hankittichai, J Ngoenkam, A Makeudom, K Lirdprapamongkol, S Krisanaprakornkit, S Pongcharoen, J Svasti and S Aluksanasuwan. Heat shock protein family D member 1 mediates lung cancer cell‑induced angiogenesis of endothelial cells. BiomedicalReports 2025; 22(5), 77.
A Suarez-Arnedo, FT Figueroa, C Clavijo, P Arbeláez, JC Cruz and C Muñoz-Camargo. An image J plugin for the high throughput image analysis of in vitro scratch wound healing assays. PLoS One 2020; 15(7), e0232565.
L Xie, Y Chen, J Chen, H Zhang, Y Liao, Y Zhou, L Zhou and C Qing. Anti-tumor effects and mechanism of GA-13315, a novel gibberellin derivative, in human lung adenocarcinoma: An in vitro and in vivo study. Cellular & Molecular Biology Letters 2019; 24, 6.
M Dowsett, PE Goss, TJ Powles, G Hutchinson, AMH Brodie, SL Jeffcoate and RC Coombes. Use of the aromatase inhibitor 4-hydroxyandrostenedione in postmenopausal breast cancer: optimization of therapeutic dose and route. Cancer Research 1987; 47(7), 1957-1961.
M Barman, S Roy, N Singh, D Sarkar, N Barman, A Pal, S Bhattacharyya and S Ray. 3-epicaryoptin induces G2/M phase cell cycle arrest and apoptosis in human breast cancer cells by disrupting the microtubule network, an in vitro and in silico investigation. bioRxiv 2024. https://doi.org/10.1101/2024.01.04.574171
YC Chen, YH Kuo, NC Yang, CW Liu, WT Chang and CL Hsu. Cytotoxic and apoptotic effects of caffeate derivatives on A549 human lung carcinoma cells. Journal of the Chinese Medical Association 2014; 77(10), 535-543.
C Wang, R Zhang, X Chen, M Yuan, J Wu, Q Sun, C Miao and Y Jing. The potential effect and mechanism of Saikosaponin A against gastric cancer. BMC Complementary Medicine and Therapies 2023; 23(1), 295.
X Guo, Y Meng, X Sheng, Y Guan, F Zhang, Z Han, Y Kang, G Tai, Y Zhou and H Cheng. Tunicamycin enhances human colon cancer cells to TRAIL-induced apoptosis by JNK-CHOP-mediated DR5 upregulation and the inhibition of the EGFR pathway. Anticancer Drugs 2017; 28(1), 66-74.
X Wang, W Xiong and Y Tang. Tunicamycin suppresses breast cancer cell growth and metastasis via regulation of the protein kinase B/nuclear factor-κB signaling pathway. Oncology Letters 2018; 15(4), 4137-4142.
J Plati, O Bucur and R Khosravi‐Far. Dysregulation of apoptotic signaling in cancer: Molecular mechanisms and therapeutic opportunities. Journal of Cellular Biochemistry 2008; 104(4), 1124-1149.
L Yuan, Y Cai, L Zhang, S Liu, P Li and X Li. Promoting apoptosis, a promising way to treat breast cancer with natural products: A comprehensive review. Frontiers in Pharmacology 2021; 12, 801662.
J Xu, Z Wang, Y Huang, Y Wang, L Xiang and X He. A spirostanol saponin isolated from Tupistra chinensis Baker simultaneously induces apoptosis and autophagy by regulating the JNK pathway in human gastric cancer cells. Steroids 2020; 164, 108737.
AL Risinger, SM Riffle, M Lopus, MA Jordan, L Wilson and SL Mooberry. The taccalonolides and paclitaxel cause distinct effects on microtubule dynamics and aster formation. Molecular Cancer 2014; 13, 41.
AL Risinger, EM Jackson, LA Polin, GL Helms, DA LeBoeuf, PA Joe, E Hopper-Borge, RF Ludueña, GD Kruh and SL Mooberry. The taccalonolides: Microtubule stabilizers that circumvent clinically relevant taxane resistance mechanisms. Cancer Research 2008; 68(21), 8881-8888.
L Huang, Y Wei, M Ni, H Hu, L Xi, C Wang, Z Zhu, B Yang and H Zhao. Novel withanolides from tubocapsicum anomalum suppress triple-negative breast cancer by triggering apoptosis and p53-ASCT2-SLC7A11-mediated ferroptosis. Molecules 2024; 29(8), 1838.
S Huang, P Huang, H Wu, S Wang and G Liu. Soyasaponin Ag inhibits triple-negative breast cancer progression via targeting the DUSP6/MAPK signaling. Folia Histochemica et Cytobiologica 2021; 59(4), 291-301.
BA Carneiro and WS El-Deiry. Targeting apoptosis in cancer therapy. Nature Reviews Clinical Oncology 2020; 17(7), 395-417.
A Allegra, A Alonci, G Penna, V Innao, D Gerace, F Rotondo and C Musolino. The cancer stem cell hypothesis: A guide to potential molecular targets. Cancer Investigation 2014; 32(9), 470-495.
BA Teicher. In vivo/ex vivo and in situ assays used in cancer research: A brief review. Toxicologic Pathology 2009; 37(1), 114-122.
FM Chaves, ICB Pavan, LGS da Silva, LB de Freitas, MA Rostagno, AEC Antunes, RMN Bezerra and FM Simabuco. Pomegranate juice and peel extracts are able to inhibit proliferation, migration and colony formation of prostate cancer cell lines and modulate the Akt/mTOR/S6K signaling pathway. Plant Foods for Human Nutrition 2020; 75(1), 54-62.
T Zeng, L Zhu, M Liao, W Zhuo, S Yang, W Wu and D Wang. Knockdown of PYCR1 inhibits cell proliferation and colony formation via cell cycle arrest and apoptosis in prostate cancer. Medical Oncology 2017; 34(2), 27.
NS Noor and SBS Hamid. Thymoquinone-PLGA-PF68 nanoparticles induce S phase cell cycle arrest and apoptosis, leading to the inhibition of migration and colony formation in tamoxifen-resistant breast cancer cells. Current Molecular Medicine 2025; 25(6), 760-772.
J Wu, J Jiang, B Chen, K Wang, Y Tang and X Liang. Plasticity of cancer cell invasion: Patterns and mechanisms. Translational Oncology 2021; 14(1), 100899.
SY Lee. Ginsenoside Rg1 drives stimulations of timosaponin AIII-induced anticancer effects in human osteosarcoma cells. Evidence‐Based Complementary and Alternative Medicine 2020; 2020, 8980124.
R Gao, N Shah, JS Lee, SP Katiyar, L Li, E Oh, D Sundar, CO Yun, R Wadhwa and SC Kaul. Withanone-rich combination of Ashwagandha withanolides restricts metastasis and angiogenesis through hnRNP-K. Molecular Cancer Therapeutics 2014; 13(12), 2930-2940.
A Gandalovičová, D Rosel, M Fernandes, P Veselý, V Čermák, L Petruželka, S Kumar, V Sanz-Moreno and J Brábek. Migrastatics-anti-metastatic and anti-invasion drugs: Promises and challenges. Trends Cancer 2017; 3(6), 391-406.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






