Combined Prognostic Significance of ARID1A Expression and Metastatic Lymph Node Ratio in Predicting Outcomes of Colorectal Cancer
DOI:
https://doi.org/10.48048/tis.2026.11895Keywords:
ARID1A, Colorectal cancer, Metastatic lymph node ratio, Poor Prognosis, LNM-associated genes, ARID1A, Colorectal cancer, Metastatic lymph node ratio, Poor Prognosis, LNM-associated genesAbstract
This study investigated the prognostic significance of AT-rich interactive domain 1A (ARID1A) protein expression and the metastatic lymph node ratio (mLNR) in colorectal cancer (CRC). Immunohistochemical analysis of 78 primary resected CRC specimens revealed that 85.9% exhibited low ARID1A expression, as determined by histological (H)-score and quantitative assessment. High mLNR was observed in 17.9% of cases. Notably, low ARID1A expression combined with high mLNR was significantly associated with advanced AJCC stage, increased lymph node involvement (pN stage), lymph node metastasis (LNM), lymphovascular invasion, and comorbidities. Kaplan-Meier survival analysis demonstrated that both low ARID1A expression and high mLNR were significantly associated with reduced progression-free survival (PFS). Univariable Cox proportional hazards regression identified mLNR, AJCC stage, pN stage, distant metastasis (pM stage), and LNM as significant predictors of shorter PFS. Multivariable analysis further identified mLNR and pM stage as independent prognostic factors. In addition, GEPIA2-based transcriptomic analysis revealed significant correlations between ARID1A and LNM-associated genes, such as insulin-like growth factor 1 receptor (IGF1R), heat shock protein 47 (HSP47), and vascular endothelial growth factor C (VEGF-C). These genes also showed significant prognostic relevance for survival outcomes in patients with colon adenocarcinoma (COAD) and rectum adenocarcinoma (READ) cohorts within the TCGA database. In conclusion, diminished ARID1A expression and elevated mLNR are associated with aggressive tumor features and poorer prognosis, suggesting their potential utility as combined prognostic biomarkers in CRC.
HIGHLIGHTS
- Low ARID1A and high mLNR link to aggressive features in colorectal cancer.
- ARID1A loss and mLNR elevation predict poor progression-free survival in CRC.
- mLNR and pM stage emerge as independent prognostic factors in multivariable analysis.
- ARID1A correlates with LNM-associated genes (IGF1R, HSP47, VEGF-C) tied to poor survival in TCGA.
- The combined assessment of ARID1A and mLNR improves outcome prediction in patients with colorectal cancer.
GRAPHICAL ABSTRACT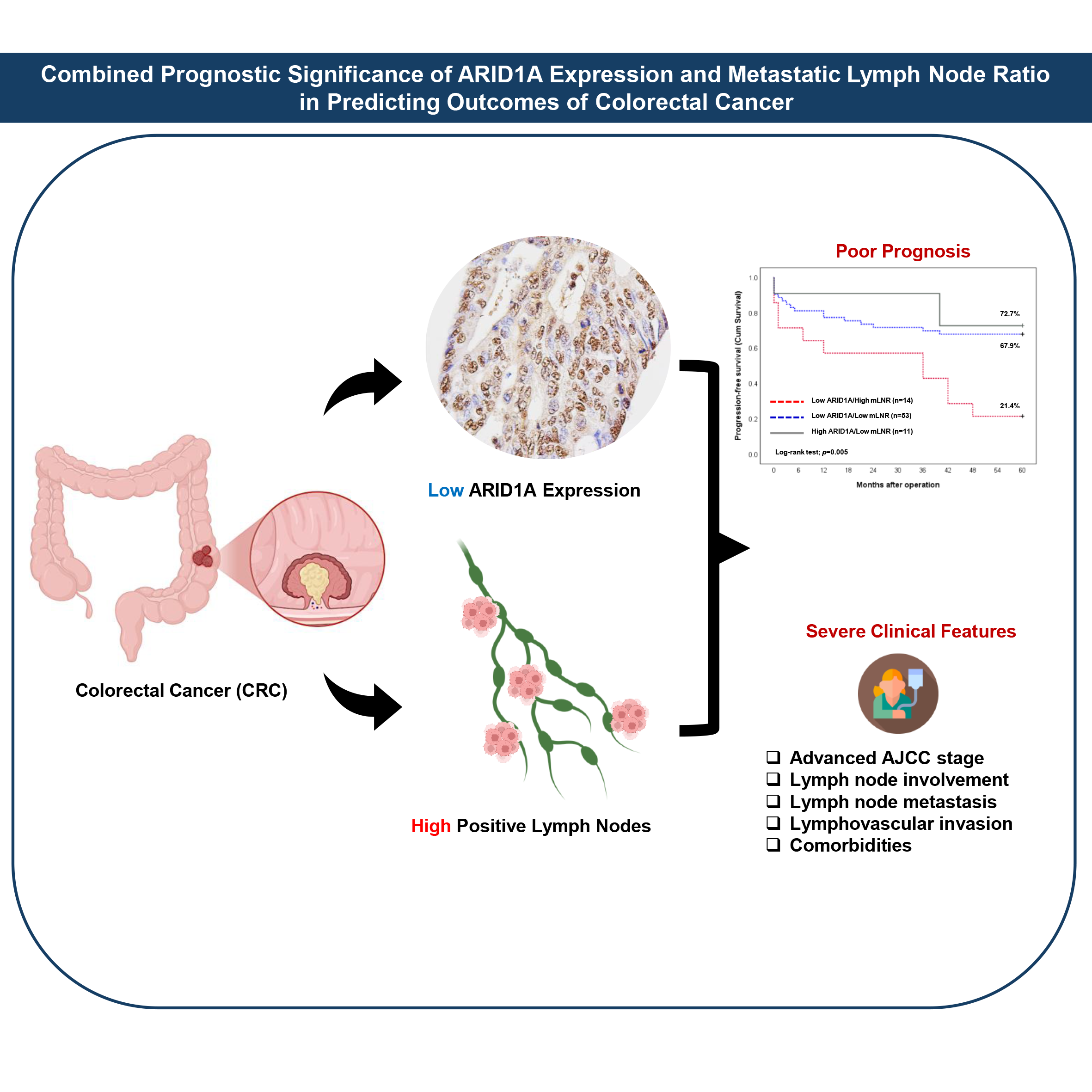
Downloads
References
H Sung, J Ferlay, RL Siegel, M Laversanne, I Soerjomataram, A Jemal and F Bray. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians 2021; 71(3), 209-249.
M Arnold, MS Sierra, M Laversanne, I Soerjomataram, A Jemal and F Bray. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017; 66(4), 683-691.
RL Siegel, KD Miller, AG Sauer, SA Fedewa, LF Butterly, JC Anderson, A Cercek, RA Smith and A Jemal. Colorectal cancer statistics, 2020. CA: A Cancer Journal for Clinicians 2020; 70(3), 145-164.
F Bray, J Ferlay, I Soerjomataram, RL Siegel, LA Torre and A Jemal. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians 2018; 68(6), 394-424.
X Wang, NG Nagl, D Wilsker, MV Scoy, S Pacchione, P Yaciuk, PB Dallas and E Moran. Two related ARID family proteins are alternative subunits of human SWI/SNF complexes. The Biochemical Journal 2004; 383(2), 319-325.
RC Wu, TL Wang and IM Shih. The emerging roles of ARID1A in tumor suppression. Cancer Biology & Therapy 2014; 15(6), 655-664.
R Mathur. ARID1A loss in cancer: Towards a mechanistic understanding. Pharmacology & Therapeutics 2018; 190, 15-23.
KC Wiegand, SP Shah, OM Al-Agha, Y Zhao, K Tse, T Zeng, J Senz, MK McConechy, MS Anglesio, SE Kalloger, W Yang, A Heravi-Moussavi, R Giuliany, C Chow, J Fee, A Zayed, L Prentice, N Melnyk, G Turashvili, AD Delaney, …, DG Huntsman. ARID1A mutations in endometriosis-associated ovarian carcinomas. The New England Journal of Medicine 2010; 363(16), 1532-1543.
Y Kishida, T Oishi, T Sugino, A Shiomi, K Urakami, M Kusuhara, K Yamaguchi, Y Kitagawa and H Ono. Associations between loss of ARID1A expression and clinicopathologic and genetic variables in T1 early colorectal cancer. American Journal of Clinical Pathology 2019; 152(4), 463-470.
M Erfani, SV Hosseini, M Mokhtari, M Zamani, K Tahmasebi, MA Naini, A Taghavi, JM Carethers, M Koi, H Brim, P Mokarram and H Ashktorab. Altered ARID1A expression in colorectal cancer. BMC Cancer 2020; 20(1), 350.
W Mongkolwat, P Sonthi, K Somsuan, S Aluksanasuwan, R Samol, N Sakulsak and S Wanna-Udom. Relationship between the protein expression of ARID1A, ARID1B and ARID2 with the clinicopathological characteristics of colorectal cancer. Biomedical Reports 2025; 23(1), 119.
HJ Kim and GS Choi. Clinical implications of lymph node metastasis in colorectal cancer: Current status and future perspectives. Annals of Coloproctology 2019; 35(3), 109-117.
FL Greene, AK Stewart and HJ Norton. A new TNM staging strategy for node-positive (stage III) colon cancer: An analysis of 50,042 patients. Annals of Surgery 2002; 236(4), 416-421.
LL Gunderson, JM Jessup, DJ Sargent, FL Greene and AK Stewart. Revised TN categorization for colon cancer based on national survival outcomes data. Journal of Clinical Oncology 2010; 28(2), 264-271.
X Zhu, SQ Lin, J Xie, LH Wang, LJ Zhang, LL Xu, JG Xu and YB Lv. Biomarkers of lymph node metastasis in colorectal cancer: Update. Frontiers in Oncology 2024; 14, 1409627.
E Bando, Y Yonemura, K Taniguchi, S Fushida, T Fujimura and K Miwa. Outcome of ratio of lymph node metastasis in gastric carcinoma. Annals of Surgical Oncology 2002; 9(8), 775-784.
AC Berger, JC Watson, EA Ross and JP Hoffman. The metastatic/examined lymph node ratio is an important prognostic factor after pancreaticoduodenectomy for pancreatic adenocarcinoma. The American Surgeon 2004; 70(3), 235-240.
AC Berger, ER Sigurdson, T LeVoyer, A Hanlon, RJ Mayer, JS Macdonald, PJ Catalano and DG Haller. Colon cancer survival is associated with decreasing ratio of metastatic to examined lymph nodes. Journal of Clinical Oncology 2005; 23(34), 8706-8712.
R Greenberg, R Itah, R Ghinea, E Sacham-Shmueli, R Inbar and S Avital. Metastatic lymph node ratio (LNR) as a prognostic variable in colorectal cancer patients undergoing laparoscopic resection. Techniques in Coloproctology 2011; 15(3), 273-279.
JS Pyo, YM Shin and DW Kang. Prognostic implication of metastatic lymph node ratio in colorectal cancers: Comparison depending on tumor location. Journal of Clinical Medicine 2019; 8(11), 1812.
FR Hirsch, M Varella-Garcia, PA Bunn, MVD Maria, R Veve, RM Bremmes, AE Baron, C Zeng and WA Franklin. Epidermal growth factor receptor in non-small-cell lung carcinomas: Correlation between gene copy number and protein expression and impact on prognosis. Journal of Clinical Oncology 2003; 21(20), 3798-3807.
J Schindelin, I Arganda-Carreras, E Frise, V Kaynig, M Longair, T Pietzsch, S Preibisch, C Rueden, S Saalfeld, B Schmid, JY Tinevez, DJ White, V Hartenstein, K Eliceiri, P Tomancak and A Cardona. Fiji: An open-source platform for biological-image analysis. Nature Methods 2012; 9(7), 676-682.
M Numata, S Morinaga, T Watanabe, H Tamagawa, N Yamamoto, M Shiozawa, Y Nakamura, Y Kameda, S Okawa, Y Rino, M Akaike, M Masuda and Y Miyagi. The clinical significance of SWI/SNF complex in pancreatic cancer. International Journal of Oncology 2013; 42(2), 403-410.
K Derwinger, G Carlsson and B Gustavsson. A study of lymph node ratio as a prognostic marker in colon cancer. European Journal of Surgical Oncology 2008; 34(7), 771-775.
CC Chin, JY Wang, CY Yeh, YH Kuo, WS Huang and CH Yeh. Metastatic lymph node ratio is a more precise predictor of prognosis than number of lymph node metastases in stage III colon cancer. International Journal of Colorectal Disease 2009; 24(11), 1297-1302.
Z Tang, B Kang, C Li, T Chen and Z Zhang. GEPIA2: An enhanced web server for large-scale expression profiling and interactive analysis. Nucleic acids research 2019; 47(1), 556-560.
R Wang, M Chen, X Ye and K Poon. Role and potential clinical utility of ARID1A in gastrointestinal malignancy. Mutation Research Reviews in Mutation Research 2021; 787, 108360.
C Xie, L Fu, Y Han, Q Li, E Wang. Decreased ARID1A expression facilitates cell proliferation and inhibits 5-fluorouracil-induced apoptosis in colorectal carcinoma. Tumour biology 2014; 35(8), 7921-7927.
XL Wei, DS Wang, SY Xi, WJ Wu, DL Chen, ZL Zeng, RY Wang, YX Huang, Y Jin, F Wang, MZ Qiu, HY Luo, DS Zhang and RH Xu. Clinicopathologic and prognostic relevance of ARID1A protein loss in colorectal cancer. World Journal of Gastroenterology 2014; 20(48), 18404-18412.
A Chou, CW Toon, A Clarkson, L Sioson, M Houang, N Watson, K DeSilva and AJ Gill. Loss of ARID1A expression in colorectal carcinoma is strongly associated with mismatch repair deficiency. Human Pathology 2014; 45(8), 1697-1703.
S Aluksanasuwan, K Somsuan, S Wanna-Udom, S Roytrakul, A Morchang, A Rongjumnong and N Sakulsak. Proteomic insights into the regulatory function of ARID1A in colon cancer cells. Oncology letters 2024; 28(2), 392.
CC Chen, JC Lin and KW Chen. Lymph node ratio as a prognostic factor in head and neck cancer patients. Radiation Oncology 2015; 10, 181.
Z Feng, QS Xu, C Wang, JZ Li, MH Mao, H Li, LZ Qin and Z Han. Lymph node ratio is associated with adverse clinicopathological features and is a crucial nodal parameter for oral and oropharyngeal cancer. Scientific Reports 2017; 7(1), 6708.
D Ding, W Stokes, M Eguchi, M Hararah, W Sumner, A Amini, J Goddard, H Somerset, C Bradley, J McDermott, D Raben and SD Karam. Association between lymph node ratio and recurrence and survival outcomes in patients with oral cavity cancer. JAMA Otolaryngology- Head & Neck Surgery 2019; 145(1), 53-61.
H Hua, Q Kong, J Yin, J Zhang and Y Jiang. Insulin-like growth factor receptor signaling in tumorigenesis and drug resistance: A challenge for cancer therapy. Journal of Hematology & Oncology 2020; 13(1), 64.
X Ye, WL Tam, T Shibue, Y Kaygusuz, F Reinhardt, EN Eaton and RA Weinberg. Distinct EMT programs control normal mammary stem cells and tumour-initiating cells. Nature 2015; 525(7568), 256-260.
S Jdeed, E Erdos, BL Balint and IP Uray. The Role of ARID1A in the nonestrogenic modulation of IGF-1 signaling. Molecular Cancer Research 2022; 20(7), 1071-1082.
S Poghosyan, N Frenkel, L van den Bent, D Raats, T Spaapen, J Laoukili, IB Rinkes, O Kranenburg and J Hagendoorn. VEGF-C propagates ‘onward’ colorectal cancer metastasis from liver to lung. British Journal of Cancer 2025; 132(1), 69-80.
K Akagi, Y Ikeda, M Miyazaki, T Abe, J Kinoshita, Y Maehara and K Sugimachi. Vascular endothelial growth factor-C (VEGF-C) expression in human colorectal cancer tissues. British Journal of Cancer 2000; 83(7), 887-891.
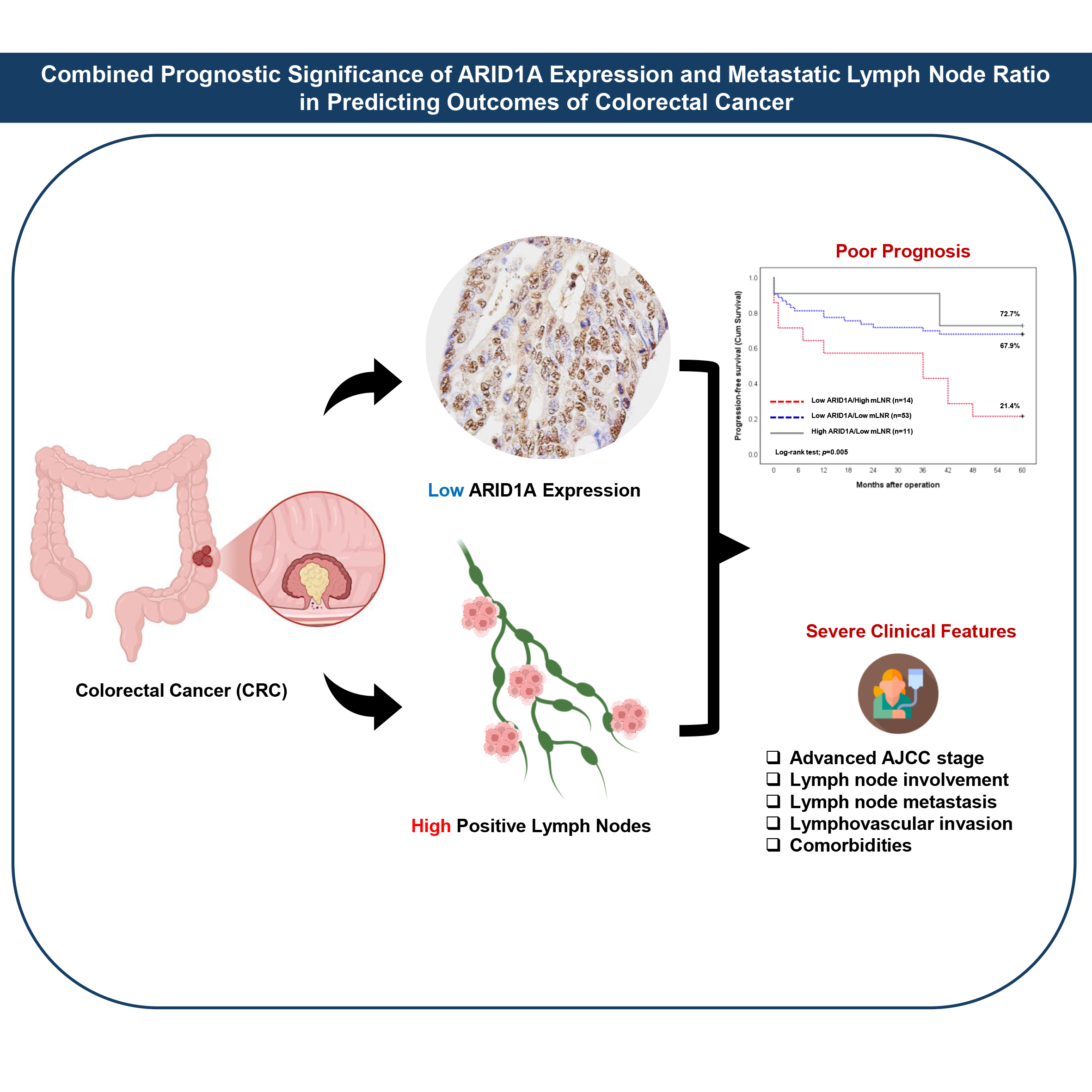
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






