Isolation and Celulolytic Activity Assay of Actinobacteria Isolated from Palm Oil Wastewater
DOI:
https://doi.org/10.48048/tis.2023.6715Keywords:
Actinobacteria, Cellulases, Congo red, Liquid waste, Oil palmAbstract
Cellulose plays an important role in the carbon cycle in nature and is the largest compound. This study aimed to isolate, to characterize and to determine actinobacteria that capable of producing cellulases. The sampling method in this study was carried out by purposive sampling at the outlet point of aeration pond of the wastewater treatment plant (IPAL) from palm oil waste station of PT. Teupin Lada. Isolation of actinobacteria was carried out on Humic Acid Vitamin b Agar (HVA), morphological characterization was carried out on Yeast Malt Agar (YMA), Yeast Starch Agar (YSA), Oatmeal Agar (OA), and microscopic characterization of actinobacteria and measuring the diameter of the clear zone formed on Carboxymethyl Cellulose (CMC) medium using the indicator Congo Red. Eight isolates were obtained from the isolation. Of the 8 isolates obtained, 7 of them were able to produce cellulase enzymes which were measured based on the clear zone formed in the test Congo Red on Carboxymethil Cellulose (CMC), and one isolate did not show any clear zones. The highest value of Cellulolytic Index (IS) was obtained from isolate ATLS-05, namely 8.38 mm.
HIGHLIGHTS
- Cellulose plays an important role in the carbon cycle in nature and is the largest compound
- The high waste load especially palm oil mill effluent (Elaeis guineensis) or known as Palm Oil Mill Effluent(POME) could cause various problems for the environment and society. POME is wastewater from the palm oil industry, which is one of the most polluting agro-industrial wastes
- Actinobacteria are one of the soil microbes which have the greatest abundance and play an important role in the decomposition process. One of the roles of soil microbes is too degrading cellulose
GRAPHICAL ABSTRACT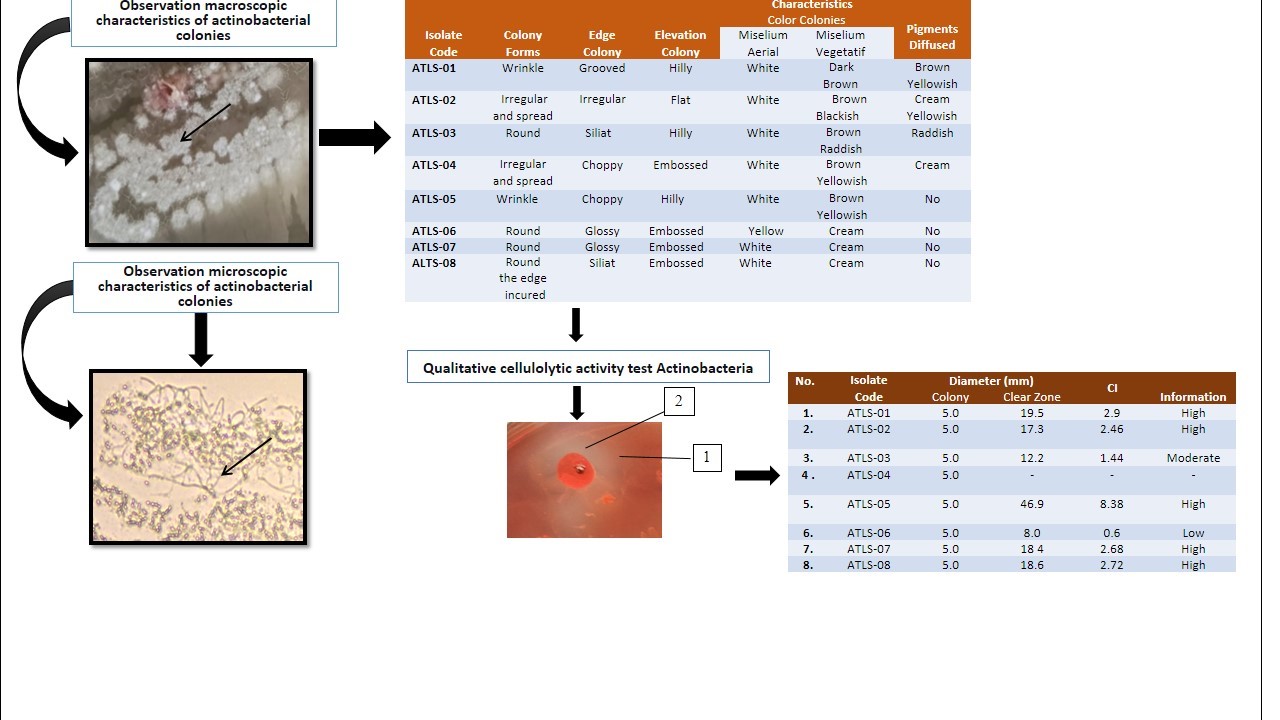
Downloads
References
MK Swandi and PD Nurmiati. Isolation of palm oil industrial wastewater degrading bacteria. Jurnal Biologi Universitas Andalas 2015; 4, 71-6.
P Naibaho. Palm oil processing technology. Oil Palm Research Center, Medan, Indonesia, 2003.
A Nurkanto. Diversity of actinomycetes Waigeo island, Raja Ampat, Papua and its potential as cellulose degrading. Berita Biologi 2008; 1, 10-1.
SM Lee and YM Koo. Pilot-scale production of cellulose using Trichoderma reesei Rut C-30 in fed batch mode. J. Microbiol. Biotech. 2001; 2, 229-33.
W Schwarz. The cellulosome and cellulose degradation by anaerobic bacteria. Appl. Microbiol Biotechnol. 2001; 56, 634-49.
N Chaudhary and P Shraddha. Thermophilic actinomycetes from hot water spring capable of producing enzymes of industrial importance. Int. J. Res. Stud. Biosci. 2016; 4, 29-35.
Seprianto. Isolasi dan penapisan bakteri selulolitik dari berbagai jenis tanah sebagai penghasil enzim selulase. Indones. J. Biotechnol. Biodiver. 2017; 1, 64-70.
H Murtiyaningsih and M Hazmi. Isolasi dxan uji aktivitas enzim selulase pada bakteri selulolitik asal tanah sampah. Agritop 2015; 15, 294-7.
SYT Adeline, A Hermanto and LP Ka. Indigenous actinomycetes from empty fruit bunch compost of oil palm: Evaluation on enzymatic and antagonic properties. Biocatal. Agr. Biotechnol. 2014; 3, 310-5.
E Armaida and S Khotimah. Karakterisasi Actinomycetes yang berasosiasi dengan Porifera (Axinella spp.) dari perairan Pulau Lekumutan Kalimantan Barat. Protobiont 2016; 5, 68-73.
H Nurkaya. Isolasi Mikroba selulolitik di beberapa lokasi industri minyak sawit. Jurnal Teknologi Pertanian 2017; 9, 26-33.
A Basilio, I Gonzalez, MF Vicente, J Gorrochategui, A Cabello, A Gonzalez and O Genilloud. Patterns of antimicrobial activities from soil actinomycetes isolated under different conditions of pH and salinity. J. Apll. Microbiol. 2003; 95, 815-6.
D Dhanasekaran and Y Jiang. Actinobacteria - Basics and biotechnological applications. IntechOpen, London, 2016.
Sulistiyani TR, Widhyastuti N. isolasi, seleksi dan identifikasi molekuler Aktinomisetes penghasil antibiotikWidyariset, 2011; 14, 533-44.
M Hayakawa, Y Yoshida and Y Iimura Y. Selective isolation of bioactive soil Actinomycetes belonging to the Streptomyces violaceusniger phenotypic cluster. J. Appl. Microbiol. 2004; 96, 973-81.
W Retnowati. The molecular mechanism of insertion of aminoglycoside resistant genes with the gene Streptomyces sp. Indonesian local isolate. Airlangga University, Surabaya, Indonesia, 2008.
S Suhartono and R Nursanty. 2012. Bioprospecting soil actinomycetes isolation and antibacterial assay. Jurnal Biologi Edukasi 2012; 4, 1-6.
C Mendez, AF Brana, MB Manzanal and C Hardison. Role of susbtrate mycelium in colony development in Streptomyces. J. Microbiol. 1985; 31, 446-7.
P Amit, A Imran, SB Kailash, G Tanushri and S Vidyottma. Isolation and characterization of actinomycetes from soil and evaluation of antibacterial activitas of actinomycetes against pathogens. Int. J. Appl. Biol. Pharmaceut. Tech. 2011; 2, 384-92.
E Astuty. Isolasi dan karakterisasi morfologi Aktinomiset indigenus asal Tanah Gambut. J. Environ. Sci. 2017; 8, 7-15.
M Husnah, Suhartono and C Yulvizar. Isolation of soil actinomycetes from forest park of Pocut Merah Intan as potential producers of antimicrobial compounds. In: Proceedings of the 2nd Annual International Conference & the 8th IMT-GT Uninet Biosciences Conference, Banda Aceh, Indonesia. 2012. p. 307-12.
H Nonomura. Key for classiffication and identification of 458 spesies of the Streptomyces included in ISP. J. Ferment. Tech. 1974; 2, 78-92.
R Anandan, D Dharumadurai and GP Manogaran. An introduction to actinobacteria. basics and biotechnology application. In: D Dhanasekaran and Y Jiang (Eds.). Actinobacteria. IntechOpen, London, 2016.
Y Jiang, Q Li, X Chen and C Jiang. Isolation and cultivation methods of actinobacteria. Basics Biotechnol. Appl. 2016; 2, 42-6.
M Oskay. Antifungal and antibacterial compounds from Streptomyces strains. Afr. J. Biotechnol. 2009; 8, 3007-17.
E Utarti, A Suwanto, TM Suhartono and A Meryandini. Identifikasi aktinomiset selulolitik dan xilanolitik indigenous. Berkala Sainstek 2020; 8, 1-5.
S Wulandari and S Nanik. Pengaruh media terhadap pertumbuhan isolat Actinomycetes kode Al35 serta optimasi produksi metabolit antibakteri berdasarkan waktu fermentasi dan pH. Media Farmasi 2016; 13, 186-98.
D Lidiani, D Dhanumadurai and GP Manogaran. Identifikasi isolat aktinomisetes yang di isolasi dari Tanah Gambut Pontianak Utara. Jurnal Kimia Khatulistiwa 2019; 2, 41-5.
A Asnani, D Ryandini and Suwandri. Karakterisasi dan identifikasi spesies aktinomisetes K-3E. In: Proceedings of the Seminar Nasional Pengembangan Sumber Daya Perdesaan dan Kearifan Lokal Berkelanjutan V, Purwokerto, Indonesia. 2015.
Elsie, N Herlina and TR Putri. Isolation of endophytic Actinomycetes from vetiver plants (Vetiveria zizanioides) and test the activity of antibacterial compounds against Staphylococcus aureus and Escherichia coli. Photon 2018; 8,13-5.
NS Rao. Soil microorganisms and plant growth. 2nd eds. UI press, Jakata, Indonesia, 1994.
MR Khan and ST William. Studies on the ecology of actinomycetes in soil strains for the production of antifungal metabolites. Afr. J. Microbiol. Res. 1975; 1, 27-32.
UN Mahida. Water pollution and utilization of industrial waste. Raja Grafindo Persada, Jakata, Indonesia, 1993.
K Mahfut. Analysis of the quality of wastewater in anaerobic ponds iv at the wastewater treatment plant (WTP) of PT. Perkebunan Nusantara VII (Persero) bekri business unit. Biogenesis 2013; 2, 85-7.
A Pesrita, MT Lindaand S Devi. Selection and activity of riau local actinomycetes cellulase enzymes on sugarcane lignocellulose media. Jurnal Riau Biologia 2017; 2, 8-13.
AS Ponnambalam, RS Deepthi and AR Ghosh. Qualitative display and measurement of enzyme activity of isolated cellulolytic bacteria. Biotechnol. Bioinf. Bioeng, 2011; 1, 33-35.
MI Puspawati, WD Atmaja and WS Sutari. Exploration of cellulolytic bacteria from organic waste Denpasar City. J. Trop. Agroecotechnol. 2018; 7, 363-73.
A Saini, A Aggarwal and A Yadav. Cellulolytic potential of actinomycetes isolated from different habitats. Bioeng. Biosci. 2016; 4, 88-94.
P Das, R Solanki and M Khanna. Isolation and screening of cellulolytic actinomycetes from diverse habitats. Int. J. Adv. Biotech Res. 2014; 15, 438-51.
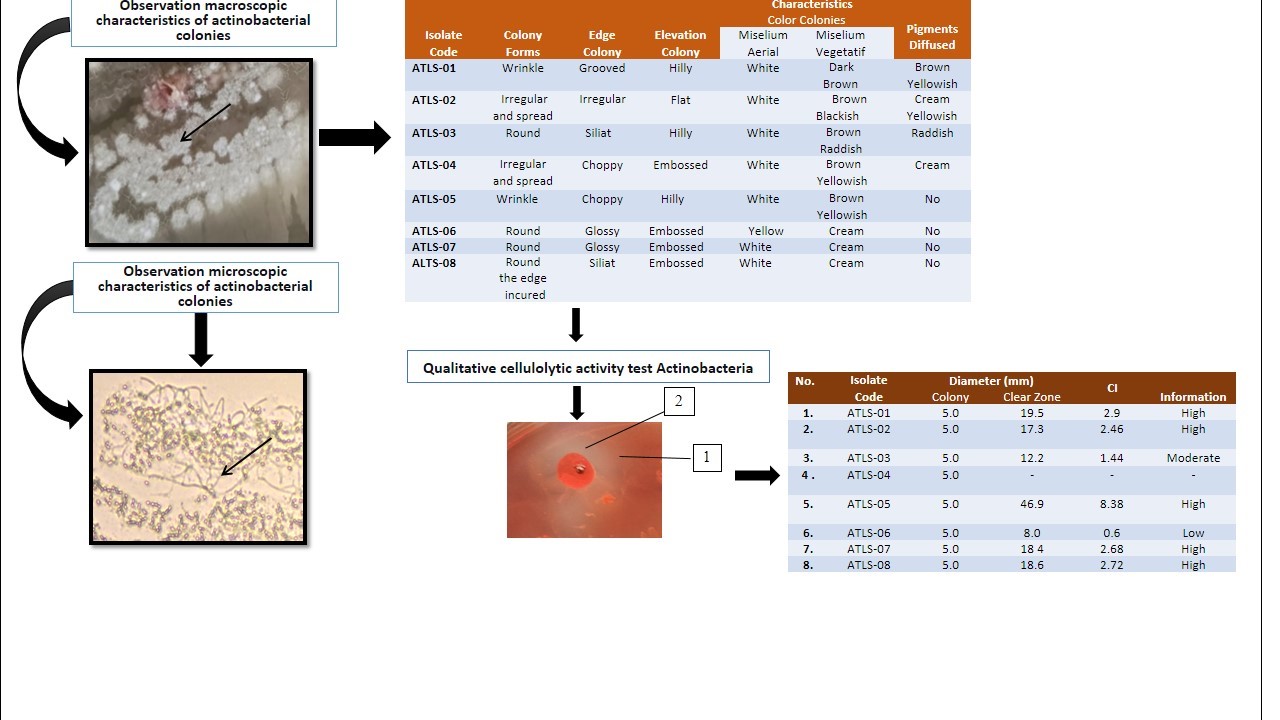
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






