Unveiling the Significant Role on Synthesis of Magnetic CuFe2O4 Photocatalyst for Dye Degradation
DOI:
https://doi.org/10.48048/tis.2026.12296Keywords:
Magnetic, Metal ferrite, Photocatalyst, Spinel ferriteAbstract
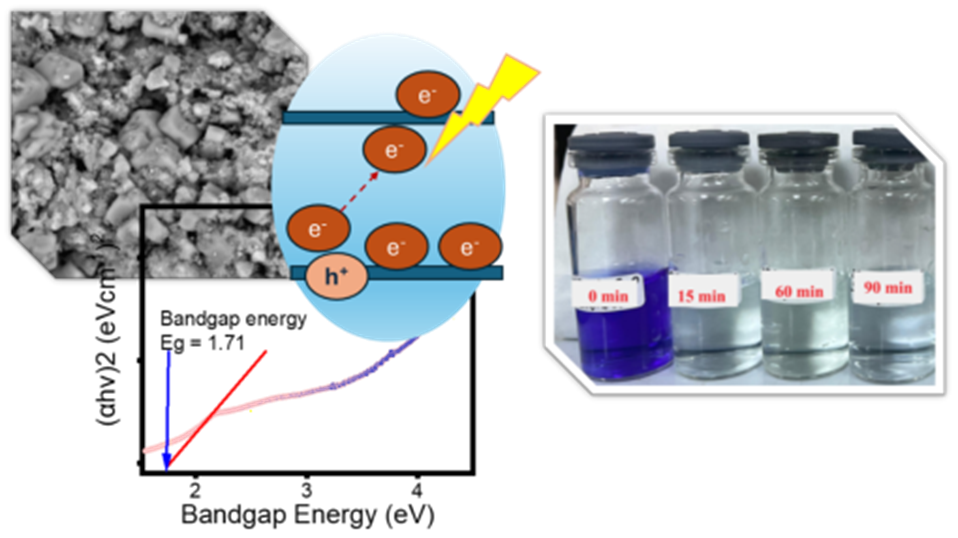
Metal ferrites have been known and utilized in various applications, including as photocatalyst for organic contaminated water treatment. In this work, exploration on synthesis of copper ferrite (CuFe2O4) by evaluating the role of synthesis method and the use of surfactant on physicochemical characteristics has been performed. Effect of hydrothermal (HT) and ultrasound-irradiation methods with and without the addition of cetyltrimethylammonium bromide (CTAB) as templating agent on physical and optical features was analysed using X-ray diffraction (XRD), scanning electron microscopy, Fourier-Transform Infra-red, particle’s size analysis, diffuse reflectance UV-Visible spectrophotometry (UV-DRS), and vibrating sample magnetometer (VSM). Photocatalytic activity of the prepared samples was examined in methyl violet photocatalytic degradation under UV and visible lights exposure. The HT treatment produced higher crystallite and particle’s size, and the increased size is correlated to decreased band gap energy of nanomaterials. In addition, the particle size which also influenced to magnetism. The band gap energy of materials are ranging at 1.71 - 2.01 eV, while magnetism ranging at 14.45 - 18.41 emu/g with the highest value gained by HT-prepared nanomaterial, while the lowest value is observed by ultrasound-irradiated method. Photocatalytic performance of the prepared materials is excellent as shown by an efficient methyl violet degradation under UV and visible light exposure. The maximum degradation of 99.9% was achieved by CuFe2O4 with the lowest band gap and largest crystallite size prepared under hydrothermal method without CTAB addition.
HIGHLIGHTS
- Crystallite size of CuFe2O4 is determined by synthesis procedure and influenced by the presence of cetyltrimethylammonium bromide as templating agent physicochemical character and photocatalytic activity
- Band gap energy and magnetism of CuFe2O4 are correlated to the crystallite size, which depends on synthesis procedure.
- CuFe2O4 showed a high potentiality to be photocatalyst in methyl violet removal under UV and visible lights.
GRAPHICAL ABSTRACT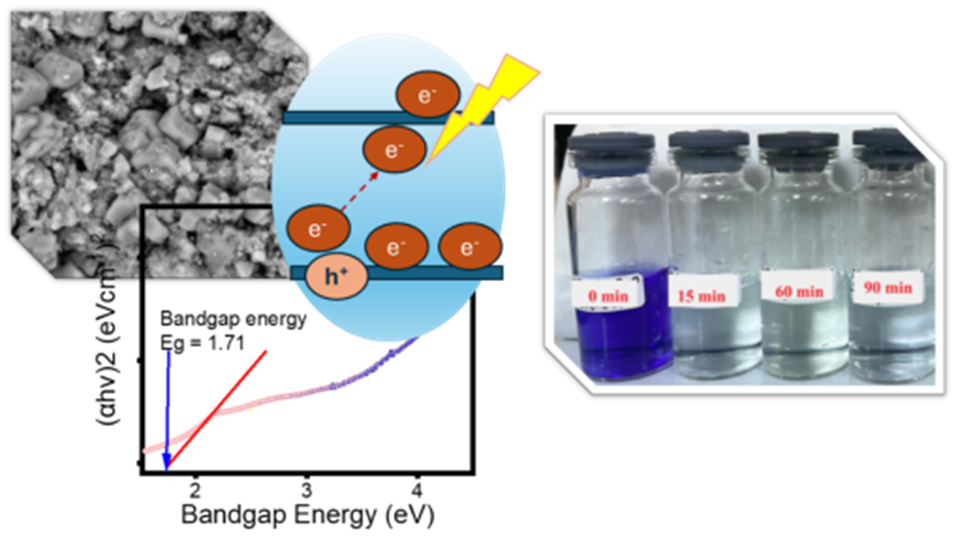
Downloads
References
JR la Calvo-De and M Segarra. Optimization of the synthesis of copper ferrite nanoparticles by a polymer-assisted sol-gel method. ACS Omega 2019; 4(19), 18289-18298.
FH Mulud, NA Dahham and IF Waheed. Synthesis and characterization of copper ferrite nanoparticles. IOP Conference Series: Materials Science and Engineering 2020; 928, 072125.
MQ Fahem, MH Jawad, RO Abdulsada and ZT Turki. The structure and electrical properties of NiFe2O4 and NiMgFe2O4 prepared via sol-gel method. Ionics 2024; 31, 6475-6481.
SP Keerthana, R Yuvakkumar, G Ravi, S Pavithra, M Thambidurai, C Dang and D Velauthapillai. Pure and Ce-doped spinel CuFe2O4 photocatalysts for efficient rhodamine B degradation. Environmental Research 2021; 200, 111528.
MH Jawad, AA Assi and AM Hameed. Hydrothermal synthesis of zinc oxide nanostructures using varied reactor designs: A comparative study. Plasmonic 2025; 20, 7883-7894.
G Nabiyouni, D Ghanbari, A Yousofnejad, M Seraj and Z Mirdamadian. Microwave-Assisted synthesis of CuFe2O4 nanoparticles and Starch-Based magnetic nanocomposites. Journal of Nanostructures 2013; 3(2), 155-160.
H Hou, G Xu, S Tan and S Xiang. A facile hydrothermal synthesis of nanoscale CuFe2O4 spinels with enhanced infrared radiation performance. Journal of Alloys and Compounds 2018; 735, 2205-2211.
U Naresh, RJ Kumar and T Ram. Optical properties of copper ferrite nano-particle synthesized via hydrothermal technique. Bulletin of Pure & Applied Sciences- Physics 2018; 37(2), 172.
T Ramesh, V Madhavi, PV Rao, K Vagdevi, N Basavegowda and KH Baek. Impact of ultrasonic-assisted co-precipitation synthesis and ultrasonication duration on cobalt ferrite nanostructures: A comparative study of structural, morphological, optical and magnetic properties. Inorganic Chemistry Communications 2025; 174(1), 114035.
P Paramasivan and P Venkatesh. Controllable synthesis of CuFe2O4 nanostructures through simple hydrothermal method in the presence of thioglycolic acid. Physica E: Low-Dimensional Systems and Nanostructures 2016; 84, 258-262.
AR Abbasian, SS Hosseini, M Shayesteh, MS Afarani and MR Esmaeilzaei. Ultrasonic-assisted solvothermal synthesis of self-assembled Copper Ferrite nanoparticles. International Journal of Nano Dimension 2020; 11(2), 130-144.
SF Rus, P Vlazan, S Novaconi, P Sfirloaga and I Grozescu. Synthesis and characterization CuFe2O4 nanoparticles prepared by the hydrothermal ultrasonic assisted method. Journal of Optoelectronics and Advanced Materials 2012; 14(3), 293-297.
A Memar, CM Phan and MO Tade. Influence of surfactants on Fe2O3 nanostructure photoanode. International Journal of Hydrogen Energy 2012; 37(22), 16835-16843.
AS Patil, RT Patil and VJ Fulari. Surfactant assisted synthesis of CuO nanostructures for nonenzymatic glucose sensor. Materials Today: Proceedings 2021; 46(6), 2340-2346.
KW Jeon, JW Park, RR Lee, JH Gong, WJ Jang, JO Shim and YW Ju. Facile synthesis of CuFe2O4 catalyst by the electrospinning method to produce hydrogen via the water gas shift of waste-derived syngas. Journal of Environmental Chemical Engineering 2023; 11(3), 110105.
I Singh and RK Bedi. Surfactant-assisted synthesis, characterizations, and room temperature ammonia sensing mechanism of nanocrystalline CuO. Solid State Sciences 2011; 13(11), 2011-2018.
S Karuppiah, S Thangaraj, SA Palaniappan and SO Lakshmanan. Influence of surfactants on structural, morphological, optical and antibacterial properties of SnO2 nanoparticles. IET Nanobiotechnology 2019; 13(9), 952-956.
H Liu, S Ji, H Yang, H Zhang and M Tang. Ultrasonic-assisted ultra-rapid synthesis of monodisperse meso-SiO2@Fe3O4 microspheres with enhanced mesoporous structure. Ultrasonics Sonochemistry 2014; 21(2), 505-512.
CC Vidyasagar and YA Naik. Surfactant (PEG 400) effects on crystallinity of ZnO nanoparticles. Arabian Journal of Chemistry 2016; 9(4), 507-510.
RS López, VF Puntes, NG Bastús, C Hervés and C Jaime. Nucleation and growth of gold nanoparticles in the presence of different surfactants. A dissipative particle dynamics study. Scientific Reports 2022; 12, 13926.
RS Babu, E Madai, DS Nair, P Gonugunta, SM Armaki, R Hendrikx, T Panneerselvam, R Murugan, VVRK Kumar, P Taheri, A Mol, N Satyanarayana and PR Anusuyadevi. Effect of synthesis conditions on morphology, surface chemistry and electrochemical performance of nickel ferrite nanoparticles for lithium ‑ ion battery applications. Journal of Materials Science: Materials in Electronics 2025; 36, 865.
M Sharma, G Sharma, N Tyagi, AM Siddiqui and M Khanuja. Advanced photocatalytic degradation of textile dyes and removal of heavy metal ions from MFe2O4 using photo-Fenton mechanism. Journal of Materials Science: Materials in Electronics 2024; 35, 497.
A Meidanchi and H Ansari. Copper spinel ferrite superparamagnetic nanoparticles as a novel radiotherapy enhancer effect in cancer treatment. Journal of Cluster Science 2020; 32, 657-663.
B Ghalib and M Hessien. Magnetic and antibacterial properties of substituted cobalt spinel ferrite nanocomposites synthesized via henna green microwave hydrothermal method. Scientific Reports 2025; 15, 16653.
I Fatimah, G Purwiandono, MH Jauhari, AAAP Maharani, S Sagadevan, WC Oh and RA Doong. Synthesis and control of the morphology of SnO2 nanoparticles via various concentrations of Tinospora cordifolia stem extract and reduction methods. Arabian Journal of Chemistry 2022; 15(4), 103738.
M Janulevicius, V Klimkevicius, A Vanetsev, V Plausinaitiene, S Sakirzanovas and A Katelnikovas. Controlled hydrothermal synthesis, morphological design and colloidal stability of GdPO4·nH2O particles. Materials Today Communications 2020; 23, 100934.
RS Yadav, I Kuřitka, J Vilcakova, J Havlica, J Masilko, L Kalina, J Tkacz, M Hajdúchová and V Enev. Structural, dielectric, electrical and magnetic properties of CuFe2O4 nanoparticles synthesized by honey mediated sol-gel combustion method and annealing effect. Journal of Materials Science: Materials in Electronics 2017; 28, 6245-6261.
MK Satheeshkumar, ER Kumar, C Srinivas, G Prasad, SS Meena, I Pradeep, N Suriyanarayanan and DL Sastry. Structural and magnetic properties of CuFe2O4 ferrite nanoparticles synthesized by cow urine assisted combustion method. Journal of Magnetism and Magnetic Materials 2019; 484, 120-125.
X Wang, M Zhu, LK Koopal, W Li, W Xu, F Liu, J Zhang, Q Liu, X Feng and DL Sparks. Effects of crystallite size on the structure and magnetism of ferrihydrite. Environmental Science Nano 2016; 3(1), 190-202.
TR Neves, LFL Schildt, MLLSE Silva, VVV Vasconcelos, CD Conzo, F Mura, M Rossi, G Varvaro, M Abdolrahimi, S Quaranta, SAD Ferreira and EC Paris. Magnetic CuFe2O4 nanoparticles immobilized on modified rice husk-derived zeolite for chlorogenic acid adsorption. Magnetochemistry 2024; 10(11), 87.
S Anandan, T Selvamani, G Guru Prasad, AM Asiri and JJ Wu. Magnetic and catalytic properties of inverse spinel CuFe2O4 nanoparticles. Journal of Magnetism and Magnetic Materials 2017; 432, 437-443.
M Awad and I Zhitomirsky. Magnetic CuFe2O4 spinel-polypyrole pseudocapacitive composites for energy storage. Materials 2024; 17(21), 5249.
S Singhal, T Namgyal, N Laxhmi and S Bansal. Doping assisted structural transition in copper substituted MgFe2O4 ferrite: Thermal, electrical and magnetic properties. Scientia Iranica 2013; 20(6), 2323-2331.
M Singh and A Singhal. Modeling of shape and size effects for the band gap of semiconductor nanoparticles. In: Proceedings of the 2018 2nd International Conference on Micro-Electronics and Telecommunication Engineering (ICMETE), Ghaziabad, India. 2018, p. 339-342.
HV Ganvir, B Jonathan, KS Kumar, VP Raju, K Prabhakaran and NP Hegde. Validation of size of semiconductor nano material: Effect on size and shape. Communication on Applied Nonlinear Analysis 2025; 32(2), 113-122.
GT Sherka and HD Berry. Insight into impact of size and shape on optoelectronic properties of InX (X = As, Sb, and P) semiconductor nanoparticles: A theoretical study. Frontiers in Physics 2024; 12, 1447997.
M Singh, M Goyal and K Devlal. Size and shape effects on the band gap of semiconductor compound nanomaterials. Journal of Taibah University for Science 2018; 12(4), 470-475.
A Gautam, K As, S Banerjee, D Vv and K Pk. UVC-Shielding by Nano-TiO 2/PMMA composite: A chemical approach. Journal of Materials Science & Nanotechnology 2016; 4(1), 105.
A Modwi, MA Ghanem, AM Al-Mayouf and A Houas. Lowering energy band gap and enhancing photocatalytic properties of Cu/ZnO composite decorated by transition metals. Journal of Molecular Structure 2018; 1173, 1-6.
M Khatun, P Mitra and S Mukherjee. Effect of band gap and particle size on photocatalytic degradation of NiSnO3 nanopowder for some conventional organic dyes. Hybrid Advances 2023; 4, 100079.
S Khosravi, N Chaibakhsh, S Jafari and M Nilkar. Enhanced photocatalytic activity of ZnS/TiO2 nanocomposite by nitrogen and tetrafluoromethane plasma treatments. Scientific Reports 2024; 14(1), 28385.
S Hussain, S Hussain, A Waleed, MM Tavakoli, Z Wang, S Yang, Z Fan and MA Nadeem. Fabrication of CuFe2O4/α-Fe2O3 composite thin films on FTO Coated Glass and 3-D Nanospike structures for efficient photoelectrochemical water splitting. ACS Applied Materials & Interfaces Journal 2016; 8(51), 35315-35322.
A Alnehia, M Hadi, H Alnahari and A Al‑Sharabi. Optical, structural and antibacterial properties of phase heterostructured nanocomposite. Scientific Reports 2024; 14(1), 14392.
A Aslam, MZ Abid, K Rafiq, A Rauf and E Hussain. Tunable sulphur doping on CuFe2O4 nanostructures for the selective elimination of organic dyes from water. Scientific Reports 2023; 13(1), 6306.
H Madhu and S Rajendra. Green synthesis and reflux method of CuFe2O4 and Clay/CuFe2O4 nanocomposite for photocatalysis and antioxidant studies. Chemistry of Inorganic Materials 2025; 6, 100099.
F Touahra, S Zemmache, S Djema, R Chebout, D Lerari and K Bachari. A new approach to the synthesis of CuFe2O4@CeO2 direct Z-scheme with a core-shell structure for enhanced photo-degradation of methyl violet under ultraviolet and visible-light irradiation. Environmental Progress and Sustainable Energy 2022; 41(5), 13865.
FMA Alzahran, J Arshad, MS Al-Buriahi, ZA Alrowaili and S Munir. Synthesis of graphene-based Ag-doped CuFe2O4 composite for improved photocatalytic activity against industrial effluents. Journal of Taibah University for Science 2023; 17(1), 2209676.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






