Hyperglycemia-Induced Apoptosis and Phagocytosis Suppression in Drosophila
DOI:
https://doi.org/10.48048/tis.2026.11170Keywords:
Hyperglycemia, Apoptosis, Phagocytosis Receptors, Inflammation, Drosophila melanogaster, Hyperglycemia, Apoptosis, Phagocytosis receptors, Inflammation, Drosophila melanogasterAbstract
Hyperglycemia, characterized by elevated blood glucose levels, is a major risk factor for the development of type 2 diabetes mellitus and its associated complications, including organ dysfunction and immune system impairment. Oxidative stress induced by hyperglycemia is known to disrupt cellular immune mechanisms, including phagocytosis, which is crucial for eliminating pathogens and damaged cells. This study aimed to assess the effects of a high-sugar diet (HSD) on phenotypic traits, phagocytosis, and expression of immune-related genes in Drosophila melanogaster wild-type w1118 and mutant itgbn2, which serves as a model organism for hyperglycemia. A phenotypic assay was conducted by observing glucose levels, development, body weight, crawling, and activity, as well as molecular analysis using the RT-qPCR method. The results showed that HSD treatment significantly increased hemolymph glucose levels and reduced larvae body weight, crawling ability, and developmental progression to the pupae and adult stages. Molecular analysis revealed a significant upregulation of pro-apoptotic gene expression in w1118 larvae, rpr expression increased by approximately 9% (p < 0.05), while in the itgbn² mutant, the increase was more substantial, reaching about 21% (p < 0.01). The grim expression was also significantly elevated in itgbn² larvae by around 25% (p < 0.001). In contrast, the phagocytic receptor gene drpr was markedly downregulated in w1118 larvae, showing a 50% reduction under HSD (p < 0.0001). In contrast, no significant changes were observed in the itgbn² mutant, a phagocytosis receptor involved in the elimination of apoptotic cells, which may reflect limited activation of integrin associated signaling pathways necessary for transcriptional responsiveness under hyperglycemic stress. Overall, these findings suggest that hyperglycemia induced by an HSD is associated with increased apoptosis and reduced phagocytic activity, possibly through disrupted integrin-mediated signaling in the itgbn² mutant, which may contribute to cellular immune dysfunction under hyperglycemic conditions. This study offers new insights into the molecular mechanisms underlying immune dysregulation in hyperglycemic conditions, highlighting the therapeutic potential of targeting phagocytic pathways to prevent diabetes-related complications in the future.
HIGHLIGHTS
- A high-sugar diet triggers hyperglycemia-like symptoms in Drosophila
- Immune disruption was evident through reduced phagocytosis and increased apoptotic activity.
- The phagocytic receptor integrin βν plays an important role in glucose homeostasis.
- Integrin βν dysfunction leads to impaired cellular immunity under hyperglycemic conditions.
- These findings offer novel insights into the link between apoptosis, phagocytosis, and metabolic stress.
GRAPHICAL ABSTRACT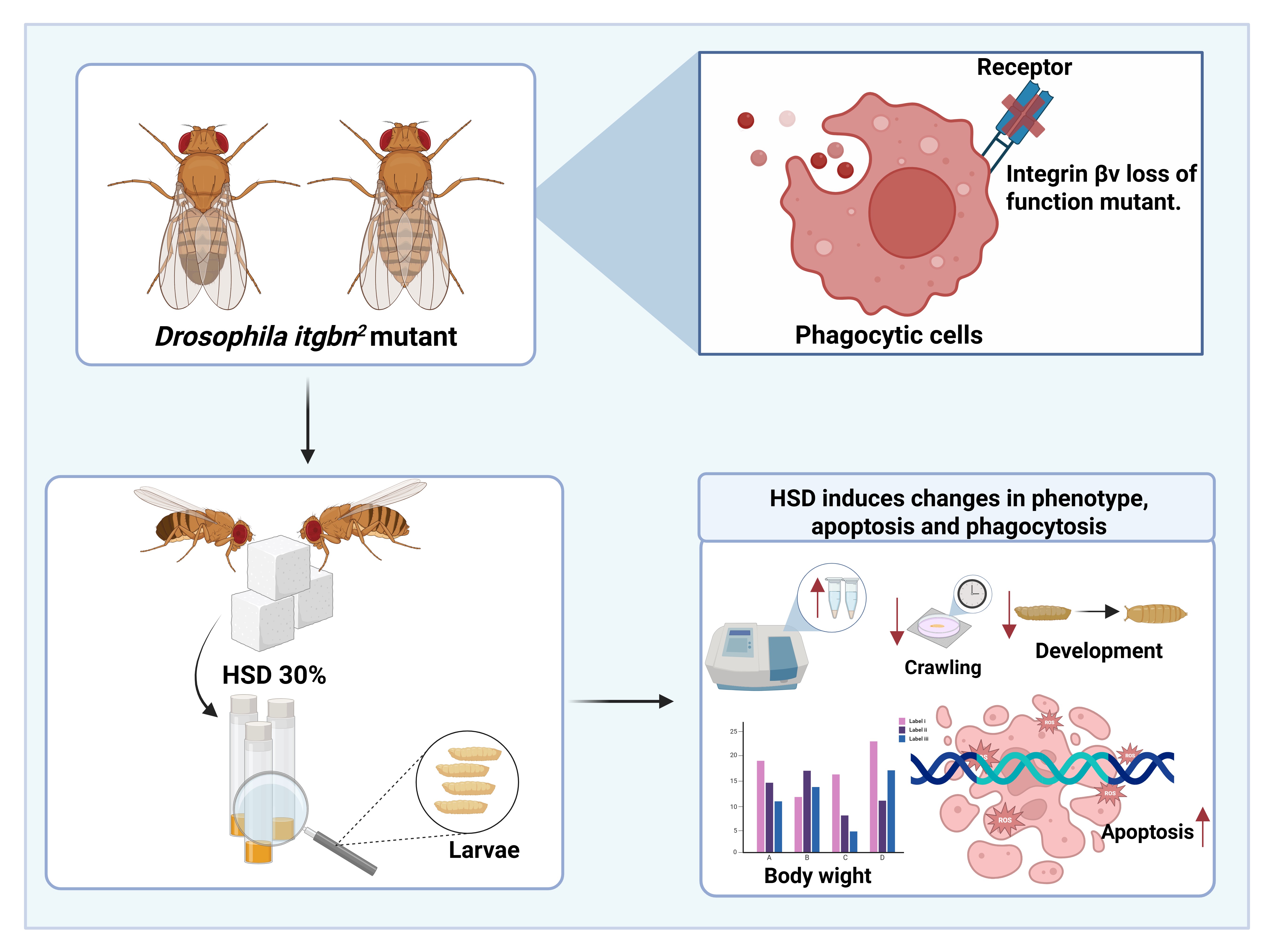
Downloads
References
M Islam, MA Hoshen, FI Ayshasiddeka and T Yeasmin. Antimicrobial, membrane stabilizing and thrombolytic activities of ethanolic extract of Curcuma zedoaria Rosc. Rhizome. Journal of Pharmacognosy and Phytochemistry 2017; 6(5), 38-41.
KL Ong, LK Stafford, SA McLaughlin, EJ Boyko, SE Vollset, AE Smith, BE Dalton, J Duprey, JA Cruz, H Hagins, PA Lindstedt, A Aali, YH Abate, MD Abate, M Abbasian, Z Abbasi-Kangevari, M Abbasi-Kangevari, SA ElHafeez, R Abd-Rabu, …, T Vos. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. The Lancet 2023; 402(10397), 203-234.
ID Federation. Diabetes around the world 2021. International Diabetes Federation, Brussels, Belgium, 2025.
MZ Banday, AS Sameer and S Nissar. Pathophysiology of diabetes: An overview. Avicenna Journal of Medicine 2020; 10(04), 174-188.
D Reyes-Umpierrez, G Davis, S Cardona, FJ Pasquel, L Peng, S Jacobs, P Vellanki, M Fayfman, S Haw, M Halkos, RA Guyton, VH Thourani and GE Umpierrez. Inflammation and oxidative stress in cardiac surgery patients treated to intensive versus conservative glucose targets. The Journal of Clinical Endocrinology & Metabolism 2017; 102(1), 309-315.
Y Li, Y Liu, S Liu, M Gao, W Wang, K Chen, L Huang and Y Liu. Diabetic vascular diseases: Molecular mechanisms and therapeutic strategies. Signal Transduction and Targeted Therapy 2023; 8(1), 152.
S Tangvarasittichai. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World Journal of Diabetes 2015; 6(3), 456.
B Zhang, Y Chen, Q Shen, G Liu, J Ye, G Sun and X Sun. Myricitrin attenuates high glucose-induced apoptosis through activating Akt-Nrf2 signaling in H9c2 cardiomyocytes. Molecules 2016; 21(7), 880.
B Giri, S Dey, T Das, M Sarkar, J Banerjee and SK Dash. Chronic hyperglycemia mediated physiological alteration and metabolic distortion leads to organ dysfunction, infection, cancer progression and other pathophysiological consequences: An update on glucose toxicity. Biomedicine & Pharmacotherapy 2018; 107, 306-328.
S Riwaldt, TJ Corydon, D Pantalone, J Sahana, P Wise, P Wise, M Wehland, M Krüger, D Melnik, Daniela, S Kopp, M Infanger and D Grimm. Role of apoptosis in wound healing and apoptosis alterations in microgravity. Frontiers in Bioengineering and Biotechnology 2021; 9, 679650.
SA Antar, NA Ashour, M Sharaky, M Khattab, NA Ashour, RT Zaid, EJ Roh, A Elkamhawy and AA Al-Karmalawy. Diabetes mellitus: Classification, mediators, and complications; A gate to identify potential targets for the development of new effective treatments. Biomedicine & Pharmacotherapy 2023; 168, 115734.
S Arandjelovic and KS Ravichandran. Phagocytosis of apoptotic cells in homeostasis. Nature Immunology 2015; 16(9), 907-917.
IKH Poon, CD Lucas, AG Rossi and KS Ravichandran. Apoptotic cell clearance: basic biology and therapeutic potential. Nature Reviews Immunology 2014; 14(3), 166-180.
S Nagata. Autoimmune diseases caused by defects in clearing dead cells and nuclei expelled from erythroid precursors. Immunological Reviews 2007; 220(1), 237-250.
S Pavlou, J Lindsay, R Ingram, H Xu and M Chen. Sustained high glucose exposure sensitizes macrophage responses to cytokine stimuli but reduces their phagocytic activity. BMC Immunology 2018; 19(1), 24.
TS Salminen and PF Vale. Drosophila as a model system to investigate the effects of mitochondrial variation on innate immunity. Frontiers in Immunology 2020; 11, 521.
F Nainu, Y Tanaka, A Shiratsuchi and Y Nakanishi. Protection of insects against viral infection by apoptosis-dependent phagocytosis. The Journal of Immunology 2015; 195(12), 5696-5706.
N Buchon, N Silverman and S Cherry. Immunity in Drosophila melanogaster--from microbial recognition to whole-organism physiology. Nature Reviews Immunology 2014; 14(12), 796-810.
S Yu, F Luo, Y Xu, Y Zhang and LH Jin. Drosophila innate immunity involves multiple signaling pathways and coordinated communication between different tissues. Frontiers in Immunology 2022; 13, 905370.
KA Hope and LT Reiter. Understanding human genetic disease with the fly. In: K Walz and JI Young (Eds.). Cellular and animal models in human genomics research. Academic Press, New York, 2019, p. 69-87.
J Manaka, T Kuraishi, A Shiratsuchi, Y Nakai, H Higashida, P Henson and Y Nakanishi. Draper-mediated and phosphatidylserine-independent phagocytosis of apoptotic cells by Drosophila hemocytes/macrophages. Journal of Biological Chemistry 2004; 279(46), 48466-48476.
K Nagaosa, R Okada, S Nonaka, K Takeuchi, Y Fujita, T Miyasaka, J Manaka, I Ando and Y Nakanishi. Integrin βν-mediated phagocytosis of apoptotic cells in Drosophila embryos. Journal of Biological Chemistry 2011; 286(29), 25770-25777.
F Nainu, A Shiratsuchi and Y Nakanishi. Induction of apoptosis and subsequent phagocytosis of virus-infected cells as an antiviral mechanism. Frontiers in Immunology 2017; 8, 1220.
N Baenas and AE Wagner. Drosophila melanogaster as a model organism for obesity and type-2 diabetes mellitus by applying high-sugar and high-fat diets. Biomolecules 2022; 12(2), 307.
F Liguori, E Mascolo and F Vernì. The genetics of diabetes: What we can learn from Drosophila. International Journal of Molecular Sciences 2021; 22(20), 11295.
F Lourido, D Quenti, D Salgado-Canales and N Tobar. Domeless receptor loss in fat body tissue reverts insulin resistance induced by a high-sugar diet in Drosophila melanogaster. Scientific Reports 2021; 11(1), 3263.
CD Nichols, J Becnel and UB Pandey. Methods to assay Drosophila behavior. Journal of Visualized Experiments 2012; 61, e3795.
YH Alam, R Kim and C Jang. Metabolism and health impacts of dietary sugars. Journal of Lipid and Atherosclerosis 2022; 11(1), 20.
C Guney and F Akar. The possible mechanisms of high-fructose diet-induced pancreatic disturbances. Journal of Research in Pharmacy 2023; 27(2), 753-761.
K Jones and I Eleftherianos. A simple protocol for isolating hemolymph from single Drosophila melanogaster adult flies. Methods and Protocols 2023; 6(5), 100.
K Kannan and YW Fridell. Functional implications of Drosophila insulin-like peptides in metabolism, aging, and dietary restriction. Frontiers in Physiology 2013; 4, 288.
LP Musselman, JL Fink, K Narzinski, PV Ramachandran, SS Hathiramani, RL Cagan and TJ Baranski. A high-sugar diet produces obesity and insulin resistance in wild-type Drosophila. Disease Models & Mechanisms 2011; 4(6), 842-849.
D Chen, J Yang, Z Xiao, S Zhou and L Wang. A diet-induced type 2 diabetes model in Drosophila. Science China Life Sciences 2021; 64, 326-329.
DR Nässel and JV Broeck. Insulin/IGF signaling in Drosophila and other insects: Factors that regulate production, release and post-release action of the insulin-like peptides. Cellular and Molecular Life Sciences 2016; 73, 271-290.
C Melcarne, B Lemaitre and E Kurant. Phagocytosis in Drosophila: From molecules and cellular machinery to physiology. Insect Biochemistry and Molecular Biology 2019; 109, 1-12.
SNS Morris, C Coogan, K Chamseddin, SO Fernandez-Kim, S Kolli, JN Keller and JH Bauer. Development of diet-induced insulin resistance in adult Drosophila melanogaster. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2012; 1822(8), 1230-1237.
BM Rovenko, OI Kubrak, DV Gospodaryov, NV Perkhulyn, IS Yurkevych, A Sanz, OV Lushchak and VI Lushchak. High sucrose consumption promotes obesity whereas its low consumption induces oxidative stress in Drosophila melanogaster. Journal of Insect Physiology 2015; 79, 42-54.
J Kim and T Neufeld. Dietary sugar promotes systemic TOR activation in Drosophila through AKH-dependent selective secretion of Dilp3. Nature Communications 2015; 6(1), 6846.
MR Azmin, H Habibie, F Filmaharani, A Roosevelt, A Nurhidayah, MR Pratama, W Hardiyanti, NP Latada, M Mudjahid, D Yuliana and F Nainu. Aspirin-mediated reduction of glucose level and inflammation in Drosophila melanogaster. ACS Omega 2025; 10(18), 18622-18628.
V Eickelberg, K Lüersen, S Staats and G Rimbach. Phenotyping of Drosophila melanogaster - a nutritional perspective. Biomolecules 2022; 12(2), 221.
L Cao, X Liu, H Cao, Q Lv and N Tong. Modified high‐sucrose diet‐induced abdominally obese and normal‐weight rats developed high plasma free fatty acid and insulin resistance. Oxidative Medicine and Cellular Longevity 2012; 2012(1), 374346.
SV Bădescu, CP Tătaru, L Kobylinska, EL Georgescu, DM Zahiu, AM Zăgrean and L Zăgrean. Effects of caffeine on locomotor activity in streptozotocin-induced diabetic rats. Journal of Medicine and Life 2016; 9(3), 275.
R Umargamwala, J Manning, L Dorstyn, D Denton and S Kumar. Understanding developmental cell death using Drosophila as a model system. Cells 2024; 13(4), 347.
C Peterson, GE Carney, BJ Taylor and K White. Reaper is required for neuroblast apoptosis during Drosophila development. Development 2002; 129(6), 1467-1476.
X Ye, XJ Zhou and H Zhang. Exploring the role of autophagy-related gene 5 (ATG5) yields important insights into autophagy in autoimmune/autoinflammatory diseases. Frontiers in Immunology 2018; 9, 2334.
J Kim, D Bilder and TP Neufeld. Mechanical stress regulates insulin sensitivity through integrin-dependent control of insulin receptor localization. Genes & Development 2018; 32(2), 156-164.
S Dey, N Murmu, T Mondal, I Saha, S Chatterjee, R Manna, S Haldar, SK Dash, TR Sarkar and B Giri. Multifaceted entrancing role of glucose and its analogue, 2-deoxy-D-glucose in cancer cell proliferation, inflammation, and virus infection. Biomedicine & Pharmacotherapy 2022; 156, 113801.
TJ LaRocca, SA Sosunov, NL Shakerley, VS Ten and AJ Ratner. Hyperglycemic conditions prime cells for RIP1-dependent necroptosis. Journal of Biological Chemistry 2016; 291(26), 13753-13761.
TM Belete. A recent achievement in the discovery and development of novel targets for the treatment of type-2 diabetes mellitus. Journal of Experimental Pharmacology 2020; 12, 1-15.
S Dhankhar, S Chauhan, DK Mehta, Nitika, K Saini, M Saini, R Das, S Gupta and V Gautam. Novel targets for potential therapeutic use in Diabetes mellitus. Diabetology & Metabolic Syndrome 2023; 15(1), 17.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






