Biofloc Meal-based Diets Enhance Immunostimulatory Activity and Survival of Penaeus vannamei Against Vibrio harveyi
DOI:
https://doi.org/10.48048/tis.2026.10950Keywords:
Biofloc technology, Penaeus vannamei, Vibrio harveyi, Shrimp feed, Immunostimulatory activity, Total hemocyte count, Phagocytic activity, Biofloc technology, Penaeus vannamei, Vibrio harveyi, Shrimp feed, Immunostimulatory activity, Total hemocyte count, Phagocytic activityAbstract
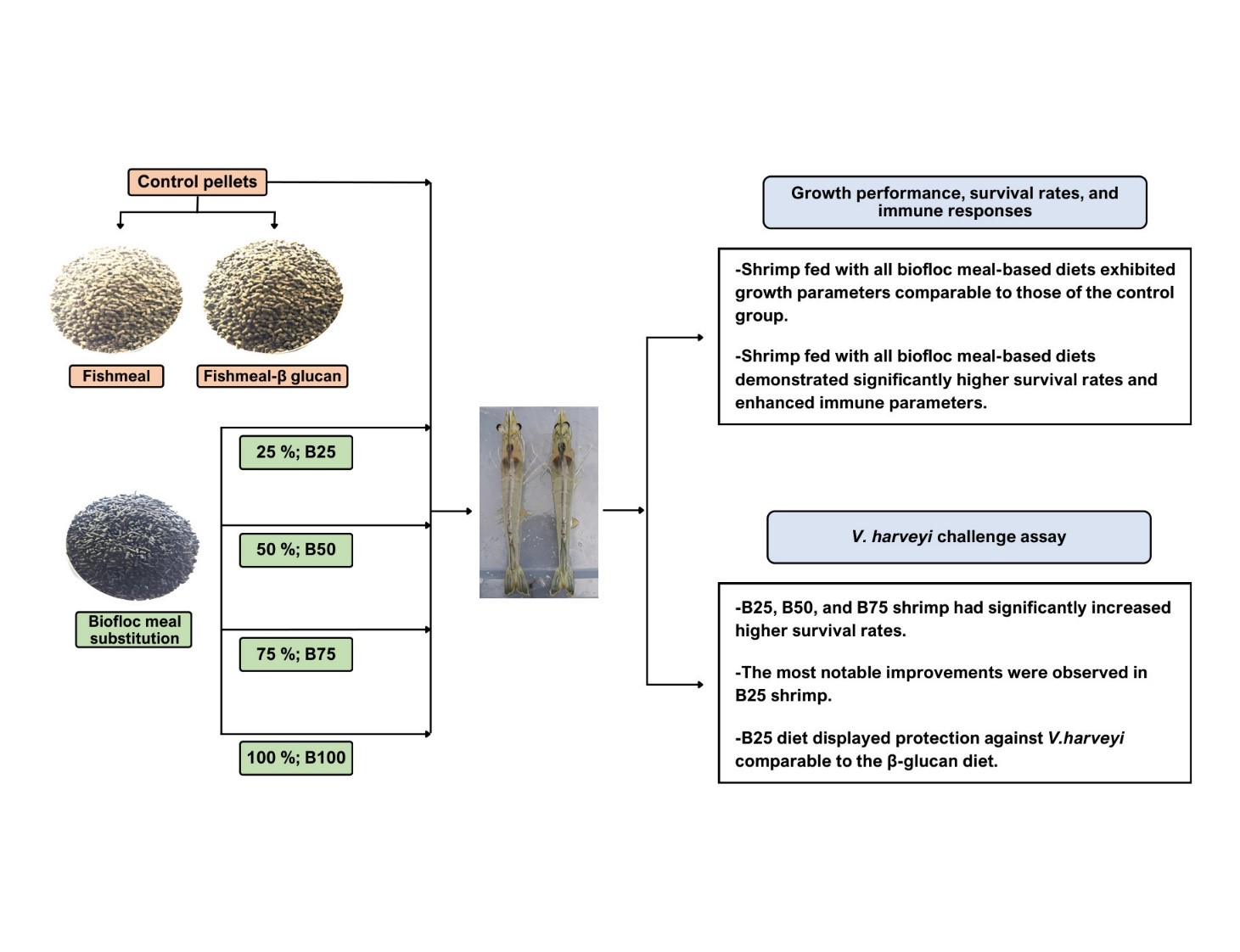
Vibrio harveyi (V. harveyi) is a major bacterial pathogen that significantly impacts shrimp cultivation. This study aimed to evaluate the potential of bioflocs, harvested from shrimp pond systems, as a fishmeal substitute to combat V. harveyi infection in shrimp Penaeus vannamei (P. vannamei). Shrimp were fed with 6 diet formulations: Four experimental diets in which fishmeal was replaced with biofloc meal at 25, 50, 75 and 100 % (designated B25, B50, B75, and B100, respectively), a fishmeal-based control diet without biofloc (B0), and a positive control diet supplemented with β-glucan (BG). Feeding trials were conducted for 30 days, followed by a challenge with V. harveyi. Growth performance, survival rates, total hemocyte count (THC), phagocytic activity, and histopathological changes were evaluated. The results demonstrated that all biofloc-fed groups had growth performance comparable to the B0 and BG groups. Shrimp fed with the B25, B50, and B75 diets exhibited significantly enhanced immune responses, with elevated THC and phagocytic activity. In the V. harveyi challenge, the B25 group showed the highest survival rate among biofloc diets, comparable to that of the BG group. Histological analysis revealed that B25 and B50 diets reduced hepatopancreatic and muscle damage associated with infection. In conclusion, this study provides evidence that substituting fishmeal with biofloc (25 - 50 %) effectively enhances immune function and confers protection against V. harveyi infection in P. vannamei, supporting its potential as a sustainable functional feed ingredient in shrimp aquaculture.
HIGHLIGHTS
- Biofloc meal partially replaced fishmeal in shrimp feed at 25-50 % substitution levels.
- Growth performance and feed conversion ratios were comparable to commercial feeds.
- Total hemocyte count and phagocytic activity were significantly improved.
- Biofloc diets enhanced the survival of Penaeus vannamei challenged with Vibrio harveyi.
- The 25 -50 % biofloc diet offered protection comparable to β-glucan-supplemented feed
GRAPHICAL ABSTRACT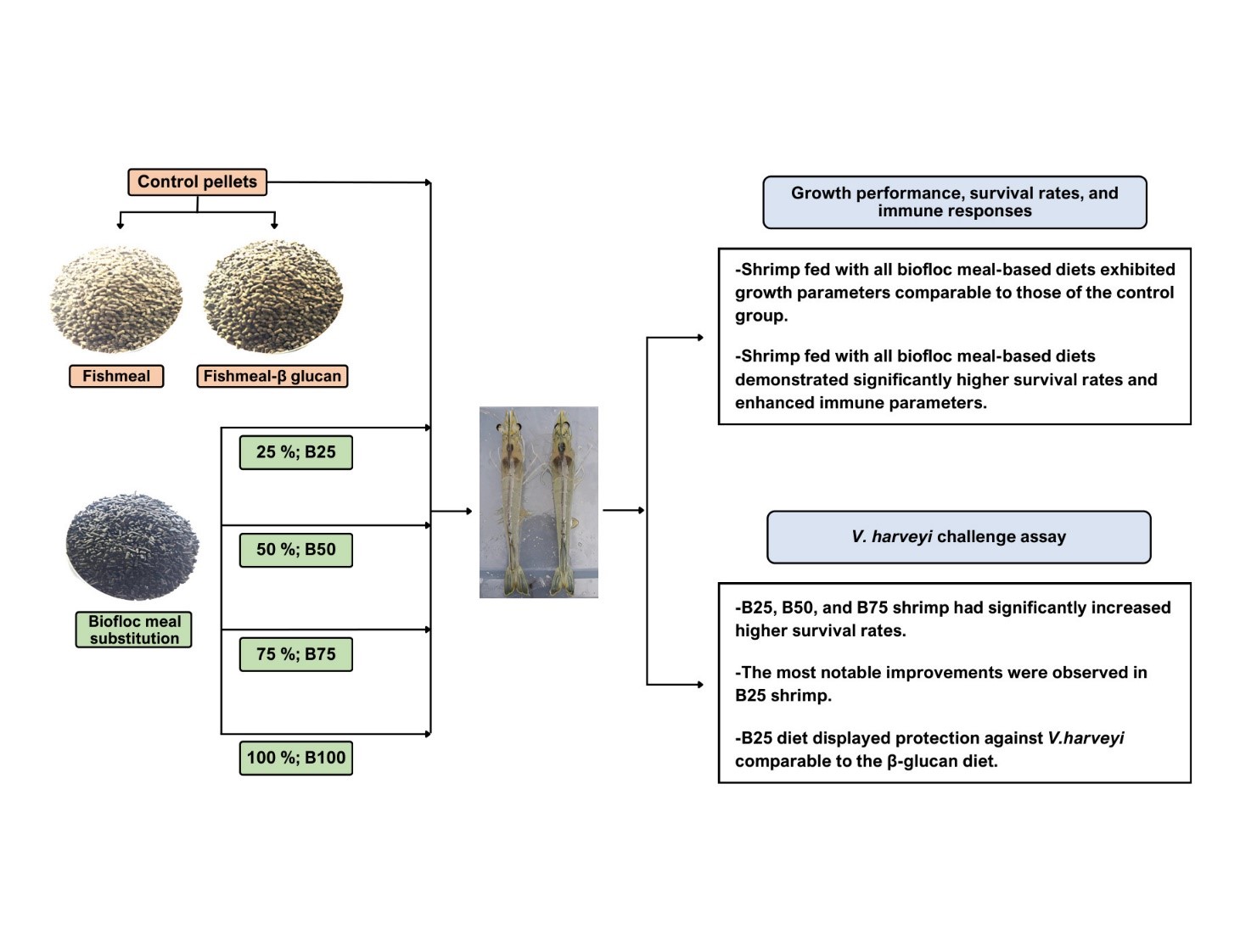
Downloads
References
CDS Valente and AHL Wan. Vibrio and major commercially important vibriosis diseases in decapod crustaceans. Journal of Invertebrate Pathology 2021; 181, 107527.
WO Haifa-Haryani, MA Amatul-Samahah, M Azzam-Sayuti, YK Chin, M Zamri-Saad, I Natrah, MNA Amal, WH Satyantini and MY Ina-Salwany. Prevalence antibiotics resistance and plasmid profiling of Vibrio spp. isolated from cultured shrimp in Peninsular Malaysia. Microorganisms 2022; 10(9), 1851.
SV Alavandi, V Manoranjita, KK Vijayan, N Kalaimani and TC Santiago. Phenotypic and molecular typing of Vibrio harveyi isolates and their pathogenicity to tiger shrimp larvae. Letters in Applied Microbiology 2006; 43(5), 566-570.
I Karunasagar, R Pai, GR Malathi and I Karunasagar. Mass mortality of Penaeus monodon larvae due to antibiotic-resistant Vibrio harveyi infection. Aquaculture 1994; 128(3-4), 203-209.
T Defoirdt, N Boon, P Sorgeloos, W Verstraete and P Bossier. Short-chain fatty acids and poly-β-hydroxyalkanoates: (New) biocontrol agents for a sustainable animal production. Biotechnology Advances 2009; 27(6), 680-685.
BM Marshall and SB Levy. Food animals and antimicrobials: Impacts on human health. Clinical Microbiology Reviews 2011; 24(4), 718-733.
RH Reboucas, OVD Sousa, AS Lima, FR Vasconcelos, PBD Carvalho and RHSDF Vieira. Antimicrobial resistance profile of Vibrio species isolated from marine shrimp farming environments (Litopenaeus vannamei) at Ceará, Brazil. Environmental Research 2011; 111(1), 21-24.
WA Bray, RR Williams, DV Lightner and A Lawrence. Growth, survival and histological responses of the marine shrimp, Litopenaeus vannamei, to three dosage levels of oxytetracycline. Aquaculture 2006; 258(1-4), 97-108.
JXH Goh, LTH Tan, JWF Law, KY Khaw, G Zengin, KG Chan, V Letchumanan, LH Lee and BH Goh. Probiotics: Comprehensive exploration of the growth promotion mechanisms in shrimps. Progress in Microbes & Molecular Biology 2023; 6(1), 0000324.
G Agurto-Rodriguez, C Dominguez-Borbor, C Tomala-Beltran, R Malave and J Rodriguez. Immunomodulatory and anti-vibrio properties of plant extract to manage the health of the shrimp Penaeus vannamei. AquaTechnica, Revista Iberoamericana de Acuicultura 2022; 4(2), 109-123.
G Wei, H Tan, S Ma, G Sun, Y Zhang, Y Wu, S Cai, Y Huang and J Jian. Protective effects of β-glucan as adjuvant combined inactivated Vibrio harveyi vaccine in pearl gentian grouper. Fish & Shellfish Immunology 2020; 106, 1025-1030.
Y Kilawati and RA Islamy. Immunostimulant activity of Gracilaria sp. and Padina sp. on immune system of vannamei shrimp (Litopenaeus vannamei) against Vibrio harveyi. Journal of Aquaculture and Fish Health 2021; 10(2), 252-257.
MH Khanjani, M Sharifinia and MGC Emerenciano. Biofloc Technology (BFT) in aquaculture: What goes right, what goes wrong? a scientific-based snapshot. Aquaculture Nutrition 2024; 2024(1), 7496572.
BCS Valle, JEM Dantas, JFX Silva, RS Bezerra, ES Correia, SRM Peixoto and RB Soares. Replacement of fishmeal by fish protein hydrolysate and biofloc in the diets of Litopenaeus vannamei postlarvae. Aquaculture Nutrition 2014; 21(1), 105-112.
J Figueroa-Espinoza, ME Rivas-Vega, MA Mariscal-Lopez, MGC Emerenciano, M Martinez-Porchas and A Miranda-Baeza. Reusing water in a biofloc culture system favors the productive performance of the Nile tilapia (Oreochromis niloticus) without affecting the health status. Aquaculture 2022; 558, 738363.
JA Hargreaves. Biofloc production systems for aquaculture. Southern Regional Aquaculture Center 2013; 1, 4503.
Y Avnimelech. Biofloc technology a practical guide book. The World Aquaculture Society, Louisiana, 2009.
MH Khanjani, M Sharifinia and S Hajirezaee. Biofloc: A sustainable alternative for improving the production of farmed cyprinid species. Aquaculture Reports 2023; 33, 101748.
P Promthale, P Pongtippatee, B Withyachumnarnkul and K Wongprasert. Bioflocs substituted fishmeal feed stimulates immune response and protects shrimp from Vibrio parahaemolyticus infection. Fish & Shellfish Immunology 2019; 93, 1067-1075.
M Nethaji, B Ahilan, A Kathirvelpandiyan, N Felix, A Uma, TS Mosses and RSS Lingam. Biofloc meal incorporated diet improves the growth and physiological responses of Penaeus vannamei. Aquaculture International 2022; 30(5), 2705-2724.
A Panigrahi, RR Das, MR Sivakumar, A Saravanan, C Saranya, NS Sudheer and G Gopikrishna. Bio-augmentation of heterotrophic bacteria in biofloc system improves growth, survival, and immunity of Indian white shrimp Penaeus indicus. Fish & Shellfish Immunology 2020; 98, 477-487.
B Raza, Z Zheng and W Yang. A review on biofloc system technology, history, types, and future economical perceptions in aquaculture. Animals 2024; 14(10), 1489.
RD Miles and FA Chapman. The benefits of fish meal in aquaculture diets: FA122/FA122, 5/2006. EDIS 2006. https://doi.org/10.32473/edis-fa122-2006
GB Misir, S Kutlu and S Çibuk. Determination of total lipid and fatty acid composition of pearl mullet (Chalcalburnus tarichi, Pallas 1811). Turkish Journal of Fisheries and Aquatic Sciences 2013; 13(5), 777-783.
S Rahimnejad, X Yuan, L Wang, K Lu, K Song and C Zhang. Chitooligosaccharide supplementation in low-fish meal diets for Pacific white shrimp (Litopenaeus vannamei): Effects on growth, innate immunity, gut histology, and immune-related genes expression. Fish & Shellfish Immunology 2018; 80, 405-415.
A Tacon, J Cody, L Conquest, S Divakaran, I Forster and O Decamp. Effect of culture system on the nutrition and growth performance of Pacific white shrimp Litopenaeus vannamei (Boone) fed different diets. Aquaculture Nutrition 2002; 8(2), 121-137.
GL Moullac and Philippe Haffner. Environmental factors affecting immune responses in Crustacea. Aquaculture 2000; 191(1-3), 121-131.
T Itami, Y Takahashi, E Tsuchihira, H Igusa and M Kondo. Enhancement of disease resistance of kuruma prawn Penaeus japonicus and increase in phagocytic activity of prawn hemocytes after oral administration of β-1,3-glucan. In Asian Fish Society 1994; 1, 375-378.
J Joshi, J Srisala, VH Truong, I-T Chen, B Nuangsaeng, O Suthienkul, CF Lo, TW Flegel, K Sritunyalucksana and S Thitamadee. Variation in Vibrio parahaemolyticus isolates from a single Thai shrimp farm experiencing an outbreak of acute hepatopancreatic necrosis disease (AHPND). Aquaculture 2014; 428-429, 297-302.
P Majluf, K Matthews, D Pauly, DJ Skerritt and MLD Palomares. A review of the global use of fishmeal and fish oil and the Fish In: Fish Out metric. Science Advances 2024; 10(42), 5650.
U Nisar, D Peng, Y Mu and Y Sun. A solution for sustainable utilization of aquaculture waste: A comprehensive review of biofloc technology and aquamimicry. Frontiers in Nutrition 2022; 8, 791738.
FAO. The state of world fisheries and aquaculture (SOFIA). Food and Agricultural Organization Fisheries, Rome, Italy, 2012.
SA Hassan, ZZ Sharawy, AFE Nahas, SA Hemeda, E El‐Haroun and EM Abbas. Modulatory effects of various carbon sources on growth indices, digestive enzymes activity and expression of growth-related genes in Whiteleg shrimp, Litopenaeus vannamei reared under an outdoor zero-exchange system. Aquaculture Research 2022; 53(16), 5594-5605.
M Menaga, P Rajasulochana, S Felix, S Sudarshan, A Kapoor, K Gandla, MM Saleh, AE Ibrahim and SE Deeb. Evaluation of biofloc-based probiotic isolates on growth performance and physiological responses in Litopenaeus vannamei. Water 2023; 15(16), 3010.
P Promthale. 2020, Production of high-quality bioflocs as a protein substitute for fishmeal in shrimp pellets. Ph. D. Dissertation, Ghent University, Gent, Belgium.
MA Islam, SS Islam, J Bir, P Debnath, MR Ullah and KA Huq. Effect on water quality, growth performance and economics of giant freshwater prawn, Macrobrachium rosenbergii with partial feed in biofloc system. Aquaculture, Fish and Fisheries 2023; 3(5), 435-446.
DN Andrianti and A Baihani. The effectiveness of white turmeric extract (Curcuma zedoaria) against the immune system of Vannamei Shrimp (Litopenaeus vannamei). Journal of Fish Health 2022; 2(1), 14-23.
F Azhar, A Mukhlis, DP Lestari and M Marzuki. Application of kappa-carrageenan as immunostimulant agent in non-specific defense system of vannamei shrimp. AACL Bioflux 2023; 16(1), 616-624.
KV Rajendran, K Sreedharan, A Deepika and A Kulkarni. Shrimp immune system and immune responses. Fish Immune System and Vaccines 2022; 1, 17-43.
M Gustilatov, W Widanarni, J Ekasari, PGS Julyantoro and DE Waturangi. Biofloc system supplemented by Pseudoalteromonas piscicida 1Ub protects the Pacific white shrimp Penaeus vannamei from Vibrio parahaemolyticus infection. Aquaculture and Fisheries 2024; 9(6), 967-974.
B Hostins, W Wasielesky, OC Decamp, P Bossier and PD Schryver. Managing input C/N ratio to reduce the risk of Acute Hepatopancreatic Necrosis Disease (AHPND) outbreaks in biofloc systems - a laboratory study. Aquaculture 2019; 508, 60-65.
L Gan, J Zheng, W Xu, J Lin, J Liu, Y Zhang, Z Wu, Z Lv, Y Jia, Q Guo, S Chen, C Liu, T Defoirdt, Q Qin and Y Liu. Deciphering the virulent Vibrio harveyi causing spoilage in muscle of aquatic crustacean Litopenaeus vannamei. Scientific Reports 2022; 12(1), 16296.
SA Soto-Rodriguez, B Gomez-Gil, R Lozano, RD Rio-Rodriguez, AL Dieguez and JL Romalde. Virulence of Vibrio harveyi responsible for the “Bright-red” Syndrome in the Pacific white shrimp Litopenaeus vannamei. Journal of Invertebrate Pathology 2012; 109(3), 307-317.
J Zhou, W Fang, X Yang, S Zhou, L Hu, X Li, X Qi, H Su and L Xie. A nonluminescent and highly virulent Vibrio harveyi strain is associated with “Bacterial white Tail Disease” of Litopenaeus vannamei shrimp. Plos One 2012; 7(2), 29961.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






