Increased Lipogenesis in the Liver of Seawater-Acclimated Nile Tilapia, Oreochromis niloticus
DOI:
https://doi.org/10.48048/tis.2025.9257Keywords:
Tilapia, High salinity, Lipogenesis, Lipid storageAbstract
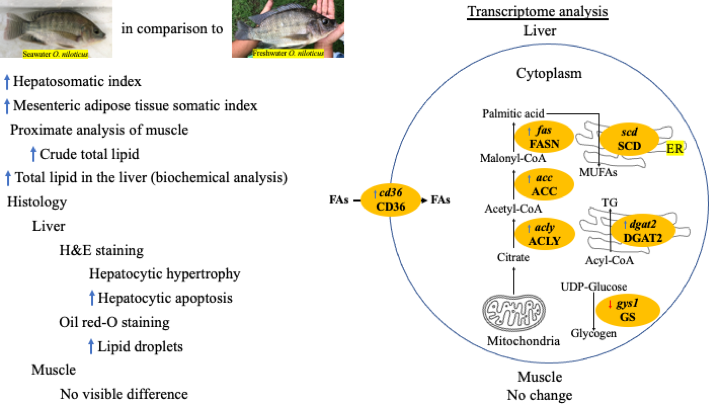
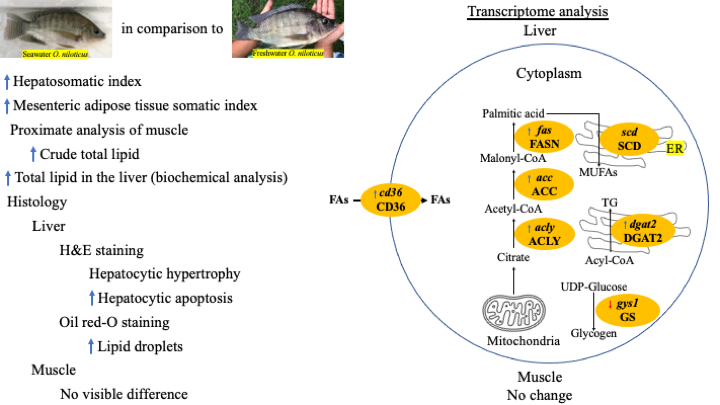
Seawater (SW)-acclimatized Oreochromis niloticus (in 27 - 30 ppt) has been generated for more than 10 generations in a private farm in Thailand, and it was noted that the muscle of the fish was softer than that of the fish reared under freshwater (FW) (in 0 - 3 ppt). We hypothesized that high lipid content in the muscle might be the cause. Proximate analysis of the muscle from FW and O. niloticus reared in SW revealed significantly higher (P < 0.05) lipid content in the SW fish. The SW fish also had significantly higher hepatosomatic index (HIS), mesenteric adipose tissue somatic index, and lipid content in the liver (all, P < 0.05), compared to the FW fish. Liver histology of the 2 groups revealed hypertrophy of the hepatocytes and a significant increase in the percentage of apoptosis of the hepatocytes, as well as an increasing size of lipid droplets observed by Oil Red-O staining. In the liver, the genes accl, acca, and fas, which are related to lipogenesis, were expressed at significantly higher levels in the SW fish, compared to the FW fish. The genes dgat2, targeting the enzyme catalyzing the last step of triglyceride synthesis, and cd36, targeting a glycoprotein for the transportation of long-chain fatty acids across cell membrane, were also expressed at significantly higher levels in the SW fish. The only gene tested that did not show a difference between the FW and SW fish was scd, which targets the enzyme catalyzing saturated fatty acid to monounsaturated fatty acid. On the contrary, the gene gys1, targeting glycogen synthase, was down-regulated. All the data indicate that O. niloticus reared in SW adapted, or re-differentiated, themselves to cope with a high-salinity environment by increasing liver biosynthesis and storage of lipid.
HIGHLIGHTS
- niloticus reared in seawater had higher lipid content in muscle than those in freshwater, resulting in softer muscle.
- Compared with niloticus reared in freshwater, the fish reared in seawater exhibited larger hepatocytes, increased apoptosis, bigger lipid droplets, and higher expressions of lipogenesis genes (accl, accα, and fas), a gene that mobilizes fatty acid into diglyceride (dgat2) to form triglyceride, and a gene that facilitates the entry of long-chain fatty acid into hepatocytes (cd36). In addition, O. niloticus reared in seawater also had lower expression of the gene that form glycogen (gys1), suggesting that glucose is shunted into the lipid biosynthesis channel.
- It is likely that niloticus reared in seawater generate more lipid in the liver to suite its increasing demand for energy source (lipid) while living under high salinity. Lipid produced in the hepatocytes is then mobilized to the muscle, where energy is expensed.
GRAPHICAL ABSTRACT
Downloads
References
The Food and Agriculture Organization of the United Nations. The state of world fisheries and aquaculture 2018 - Meeting the sustainable development goals. The Food and Agriculture Organization of the United Nations, Rome, Italy, 2018.
AFM El‐Sayed and K Fitzsimmons. From Africa to the world - the journey of Nile tilapia. Reviews in Aquaculture 2023; 15(S1), 6-21.
PJ Schofield, MS Peterson, MR Lowe, NJ Brown-Peterson and WT Slack. Survival, growth and reproduction of non-indigenous Nile tilapia, Oreochromis niloticus (Linnaeus 1758). I. Physiological capabilities in various temperatures and salinities. Marine and Freshwater Research 2011; 62(5), 439-449.
B Withyachumnarnkul, I Palang, J Reungsri, S Sirithammajak, S Jitrakorn, W Kiatpathomchai, V Saksmerprome, P Pongtippatee and B Withyachumnarnkul. Nile tilapia reared under full-strength seawater: Hemato-immunological changes and susceptibility to pathogens. Aquaculture 2017; 480, 42-50.
B Withyachumnarnkul, B Foroutan, C Kerdmusik, R Vanijviriyakij and B Withyachumnarnkul. Oogenesis, spermatogenesis and hatching rate of seawater-acclimated Nile Tilapia, Oreochromis niloticus. Songklanakarin Journal of Science and Technology 2022; 44(1), 21-25.
M Grosell, CN Laliberte, S Wood, FB Jensen and CM Wood. Intestinal HCO3- secretion in marine teleost fish: Evidence for an apical rather than a basolateral Cl-/HCO3- exchanger. Fish Physiology and Biochemistry 2001; 24, 81-95.
CLE Yen, SJ Stone, S Koliwad, C Harris and RVJ Farese. Thematic review series: Glycerolipids. DGAT enzymes and triacylglycerol biosynthesis. Journal of Lipid Research 2008; 49(11), 2283-2301.
D Black and ER Skinner. Features of the lipid transport system of fish as demonstrated by studies on starvation in the rainbow trout. Journal of Comparative Physiology B 1986; 156, 497-502.
H Akiyoshi and A Inoue. Comparative histological study of teleost livers in relation to phylogeny. Zoological Science 2004; 21(8), 841-850.
C Alasalvar, KDA Taylor, E Zubcov, F Shahidi and M Alexis. Differentiation of cultured and wild sea bass (Dicentrarchus labrax): Total lipid content, fatty acid and trace mineral composition. Food Chemistry 2002; 79(2), 145-150.
Hİ Haliloǧlu, A Bayır, AN Sirkecioglu, AN Sirkecioǧlu, NM Aras and M Atamanalp. Comparison of fatty acid composition in some tissues of rainbow trout (Oncorhynchus mykiss) living in seawater and freshwater. Food Chemistry 2004; 86(1), 55-59.
AC Burke and MW Huff. ATP-citrate lyase: Genetics, molecular biology and therapeutic target for dyslipidemia. Current Opinion in Lipidology 2017; 28(2), 193-200.
H Park, VK Kaushik, S Constant, M Prentki, E Przybytkowski, NB Ruderman and AK Saha. Coordinate regulation of malonyl-CoA decarboxylase, sn-glycerol-3-phosphate acyltransferase, and acetyl-CoA carboxylase by AMP-activated protein kinase in rat tissues in response to exercise. Journal of Biological Chemistry 2002; 277(36), 32571-32577.
D Saggerson. Malonyl-CoA, a key signaling molecule in mammalian cells. Annual Review of Nutrition 2008; 28, 253-272.
KD Chavin, RN Fiorini, S Shafizadeh, G Cheng, C Wan, Z Evans, D Rodwell, C Polito, JK Haines, GM Baillie and MG Schmidt. Fatty acid synthase blockade protects steatotic livers from warm ischemia reperfusion injury and transplantation. American Journal of Transplantation 2004; 4(9), 1440-1447.
A Jones, HM Davies and TA Voelker. Palmitoyl-acyl carrier protein (ACP) thioesterase and the evolutionary origin of plant acyl-ACP thioesterases. The Plant Cell 1995; 7(3), 359-371.
P Kunjara and P Chatikavanij. Research animals for scientific purposes in Thailand: Ethics & policies. In: Proceedings of the 6th World Congress on Alternatives & Animal Use in the Life Sciences, Tokyo, Japan. 2007, p. 189-191.
The Association of Official Analytical Chemists. Official methods of analysis. The Association of Official Analytical Chemists, Washington DC, 1990.
EG Bligh and WJ Dyer. A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology 1959; 37(8), 911-917.
C Breil, MA Vian, T Zemb, W Kunz and F Chemat. “Bligh and Dyer” and Folch methods for solid-liquid-liquid extraction of lipids from microorganisms. Comprehension of solvatation mechanisms and towards substitution with alternative solvents. International Journal of Molecular Sciences 2017; 18(4), 708.
HX Li, J Qiang, CY Song and P Xu. Acanthopanax senticosus promotes survival of tilapia infected with Streptococcus iniae by regulating the PI3K/AKT and fatty acid metabolism signaling pathway. Frontiers in Physiology 2021; 12, 699247.
N Krahmer, RVJ Farese and TC Walther. Balancing the fat: Lipid droplets and human disease. EMBO Molecular Medicine 2013; 5(7), 973-983.
P Chomczynski and N Sacchi. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Analytical Biochemistry 1987; 162(1), 156-159.
A Jayakumar, SS Chirala, AC Chinault, A Baldini, L Abu-Elheiga and SJ Wakil. Isolation and chromosomal mapping of genomic clones encoding the human fatty acid synthase gene. Genomics 1994; 23(2), 420-424.
JM Ntambi and M Miyazaki. Recent insights into stearoyl-CoA desaturase-1. Current Opinion in Lipidology 2003; 14(3), 255-261.
L Tong. Acetyl-coenzyme A carboxylase: Crucial metabolic enzyme and attractive target for drug discovery. Cellular and Molecular Life Sciences 2005; 62, 1784-1803.
G Monteuuis, F Suomi, JM Kerätär, AJ Masud and AJ Kastaniotis. A conserved mammalian mitochondrial isoform of acetyl-CoA carboxylase ACC1 provides the malonyl-CoA essential for mitochondrial biogenesis in tandem with ACSF3. The Biochemical Journal 2017; 474(22), 3783-3797.
B Bhatt-Wessel, TW Jordan, JH Miller and L Peng. Role of DGAT enzymes in triacylglycerol metabolism. Archives of Biochemistry and Biophysics 2018; 655, 1-11.
CT Coburn, FFJ Knapp, M Febbraio, AL Beets, RL Silverstein and NA Abumrad. Defective uptake and utilization of long chain fatty acids in muscle and adipose tissues of CD36 knockout mice. Journal of Biological Chemistry 2000; 275(42), 32523-32529.
A Buschiazzo, JE Ugalde, ME Guerin, W Shepard, RA Ugalde and PM Alzari. Crystal structure of glycogen synthase: Homologous enzymes catalyze glycogen synthesis and degradation. The EMBO Journal 2004; 23(16), 3196-3205.
A Lund, SM Knudsen, H Vissing, B Clark and N Tommerup. Assignment of human elongation factor 1alpha genes: EEF1A maps to chromosome 6q14 and EEF1A2 to 20q13.3. Genomics 1996; 36(2), 359-361.
KJ Livak and TD Schmittgen. Analysis of relative gene ex-pression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001; 25(4), 402-408.
S Li, Y Zhang, N Liu, J Chen, L Guo, Z Dai, C Wang, Z Wu and G Wu. Dietary L-arginine supplementation reduces lipid accretion by regulating fatty acid metabolism in Nile tilapia (Oreochromis niloticus). Journal of Animal Science and Biotechnology 2020; 11, 82.
X Zhang, Z Han, H Zhong, Q Yin, J Xiao, F Wang, Y Zhou and Y Luo. Regulation of triglyceride synthesis by estradiol in the livers of hybrid tilapia (Oreochromis niloticus♀ × O. aureus ♂). Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology 2019; 238, 110335.
Y Zhou, X Zhang, Q Xu, J Yan, F Yu, J Xiao, Z Guo, Y Luo and H Zhong. Nonadditive expression of lipid metabolism pathway-related genes in intestine of hybrids of Nile tilapia females (Oreochromis niloticus) and blue tilapia males (Oreochromis aureus). Moleuclar Biology Reports 2019; 46(1), 425-432.
C Agius and RJ Roberts. Melano‐macrophage centres and their role in fish pathology. Journal of Fish Diseases 2003; 26(9), 499-509.
G Li, AJ Sinclair and D Li. Comparison of lipid content and fatty acid composition in the edible meat of wild and cultured freshwater and marine fish and shrimps from China. Journal of Agricultural and Food Chemistry 2011; 59(5), 1871-1881.
F Taddesse, MD Huh, SC Bai and J Vijverberg. Histological changes of liver in overfed young Nile Tilapia. Journal of Fisheries and Aquatic Science 2014; 9(2), 63-74.
ZH Kareem, YM Abdelhadi, A Christianus, M Karim and N Romano. Effects of some dietary crude plant extracts on the growth and gonadal maturity of Nile tilapia (Oreochromis niloticus) and their resistance to Streptococcus agalactiae infection. Fish Physiology and Biochemistry 2016; 42(2), 757-769.
SS Omar, AY Anwar, MS Hassaan, E El-Haroun and SJ Davies. Preliminary assessment of a novel fermented wheat protein concentrate from a bio-distillation source as a dietary ingredient contribution for tilapia (Oreochromis niloticus). Aquaculture Reports 2022; 24, 101101.
LG Wang, MQ Liu, XD Xie, YB Sun, ML Zhang, Y Zhao, Q Chen, YQ Ding, ML Yu, ZM Liang, TJ Hu, WW Liang and YY Wei. Effects of different water quality regulators on growth performance, immunologic function, and domestic water quality of GIFT tilapia. PLoS One 2023; 18(8), e0290854.
O Carman, AT Mukti, M Zairin and Alimuddin. Reproductive performances of triploid male and female Nile tilapia Oreochromis niloticus (Linnaeus, 1758) at different ages. Biodiversitas 2023; 24(8), 4235-4242.
AY He, LJ Ning, LQ Chen, YL Chen, Q Xing, JM Li, F Qiao, DL Li, ML Zhang and ZY Du. Systemic adaptation of lipid metabolism in response to low‐and high‐fat diet in Nile tilapia (Oreochromis niloticus). Physiological Reports 2015; 3(8), e12485.
LJ Ning, AY He, DL Lu, JM Li, F Qiao, DL Li, ML Zhang, LQ Chen and ZY Du. Nutritional background changes the hypolipidemic effects of fenofibrate in Nile tilapia (Oreochromis niloticus). Scientific Reports 2017; 7, 41706.
Y Liu, JJ Wen, LJ Ning, JG Jiao, F Qiao, LQ Chen, ML Zhang and ZY Du. Comparison of effects of dietary‐specific fatty acids on growth and lipid metabolism in Nile tilapia. Aquaculture Nutrition 2019; 25(4), 862-872.
B Foroutan, P Pongtippatee, C Kerdmusic, W Sirimanapong, R Vanichviriyakit and B Withyachumnarnkul. Myo-inositol supplement helps the performance of seawater-acclimated Nile tilapia, Oreochromis niloticus. Aquaculture and Fisheries 2024; 9(4), 597-602.
CA Vicentini, IB Franceschini-Vicentini, MTS Bombonato, B Bertolucci, SG Lima and AS Santos. Morphological study of the liver in the teleost Oreochromis niloticus. International Journal of Morphology 2005; 23(3), 211-216.
S Nejedli and IT Gajger. Hepatopancreas in some sea fish from different species and the structure of the liver in teleost fish, common pandora, Pagellus erythinus (Linnaeus, 1758) and whiting, Merlangius merlangus euxinus (Nordmann, 1840). Veterinary Archives 2013; 83(4), 441-452.
CK Faccioli, RA Chedid, MTS Bombonato, CA Vicentini and IBF Vicentini. Morphology and histochemistry of the liver of carnivorous fish Hemisorubim platyrhynchos. International Journal of Morphology 2014; 32(2), 715-720.
DM Mokhtar. Histological, histochemical and ultrastructural characterization of the pancreas of the grass carp (Ctenopharyngodon idella). European Journal of Anatomy 2015; 19(2), 145-153.
CF Sales, RF Silva, MGC Amaral, FFT Domingos, RIMA Ribeiro, RG Thomé and HB Santos. Comparative histology in the liver and spleen of three species of freshwater teleost. Neotropical Ichthyology 2017; 15(1), 160041.
RM Palmer, A Sandbach and BA Buckley. Tissue-specific effects of temperature and salinity on the cell cycle and apoptosis in the Nile Tilapia (Oreochromis niloticus). Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 2024; 297, 111706.
R Jia, LP Cao, JL Du, Q He, ZY Gu, G Jeney, P Xu and GJ Yin. Effects of high-fat diet on antioxidative status, apoptosis and inflammation in liver of tilapia (Oreochromis niloticus) via Nrf2, TLRs and JNK pathways. Fish and Shellfish Immunology 2020; 104, 391-401.
J Zhu, JY Feng, JX Chen, RY Du, L Luo, SM Lin and Y Chen. An evaluation of the growth, blood biochemistry, hepatic glucose metabolism and hepatocyte apoptosis in the genetically improved farmed tilapia Oreochromis niloticus fed diets with distinct protein to corn starch ratios. Aquaculture Research 2021; 52(12), 6514-6524.
L Cao, G Yin, J Du, R Jia, J Gao, N Shao, Q Li, H Zhu, Y Zheng, Z Nie, W Ding and G Xu. Salvianolic acid B regulates oxidative stress, autophagy and apoptosis against cyclophosphamide-induced hepatic injury in Nile Tilapia (Oreochromis niloticus). Animals 2023; 13(3), 341.
BD Kammerer and D Kültz. Prolonged apoptosis in mitochondria-rich cells of tilapia (Oreochromis mossambicus) exposed to elevated salinity. Journal of Comparative Physiology B 2009; 179(4), 535-542.
H AnvariFar, AK Amirkolaie, HK Miandare, H Ouraji, MA Jalali and Sİ Üçüncü. Apoptosis in fish: Environmental factors and programmed cell death. Cell and Tissue Research 2017; 368(3), 425-439.
RE Wolke. Piscine macrophage aggregates: A review. Annual Review of Fish Diseases 1992; 2, 91-108.
HJ Ribeiro, MS Procópio, JMM Gomes, FO Vieira, RC Russo, K Balzuweit, H Chiarini-Garcia, ACS Castro, E Rizzo and JDJ Corrêa. Functional dissimilarity of melanomacrophage centres in the liver and spleen from females of the teleost fish Prochilodus argenteus. Cell and Tissue Research 2011; 346, 417-425.
SV Pronina, MDD Batueva and NM Pronin. Characteristics of melanomacrophage centers in the liver and spleen of the roach Rutilus rutilus (Cypriniformes: Cyprinidae) in Lake Kotokel during the Haff disease outbreak. Journal of Ichthyology 2014; 54, 104-110.
L Diaz-Satizabal and BG Magor. Isolation and cytochemical characterization of melanomacrophages and melanomacrophage clusters from goldfish (Carassius auratus, L.). Developmental and Comparative Immunology 2015; 48(1), 221-228.
JMM Gomes, HJ Ribeiro, MS Procópio, BM Alvarenga, ACS Castro, WO Dutra, JBBD Silva and JDC Junior. What the erythrocytic nuclear alteration frequencies could tell us about genotoxicity and macrophage iron storage? PLoS One 2015; 10(11), e0143929.
MP Herráez and AG Zapata. Structure and function of the melano-macrophage centres of the goldfish Carassius auratus. Veterinary Immunology and Immunopathology 1986; 12(1-4), 117-126.
IS Rabitto, JRMA Costa, HCSD Assis, EE Pelletier, FM Akaishi, A Anjos, MAF Randi and CAO Ribeiro. Effects of dietary Pb(II) and tributyltin on neotropical fish, Hoplias malabaricus: Histopathological and biochemical findings. Ecotoxicology and Environmental Safety 2005; 60(2), 147-156.
WG Manrique, GDS Claudiano, TR Petrillo, MPD Castro, MAP Figueiredo, MADA Belo, JRED Moraes and FRD Moraes. Response of splenic melanomacrophage centers of Oreochromis niloticus (Linnaeus, 1758) to inflammatory stimuli by BCG and foreign bodies. Journal of Applied Ichthyology 2014; 30(5), 1001-1006.
B Chen, W Xiao, Z Zou, J Zhu, D Li, J Yu and H Yang. Comparing transcriptomes reveals key metabolic mechanisms in superior growth performance Nile tilapia (Oreochromis niloticus). Frontiers in Genetics 2022; 13, 879570.
XY Ma, J Qiang, J He, NN Gabriel and P Xu. Changes in the physiological parameters, fatty acid metabolism, and SCD activity and expression in juvenile GIFT tilapia (Oreochromis niloticus) reared at three different temperatures. Fish Physiology and Biochemistry 2015; 41, 937-950.
J Qiang, YF Tao, J He, YL Sun and P Xu. miR-29a modulates SCD expression and is regulated in response to a saturated fatty acid diet in juvenile genetically improved farmed tilapia (Oreochromis niloticus). The Journal of Experimental Biology 2017; 220, 1481-1489.
CL Kien, JY Bunn, CL Tompkins, JA Dumas, KI Crain, DB Ebenstein, TR Koves and DM Muoio. Substituting dietary monounsaturated fat for saturated fat is associated with increased daily physical activity and resting energy expenditure and with changes in mood. The American Journal of Clinical Nutrition 2013; 97(4), 689-697.
B Yan, ZH Wang and JL Zhao. Mechanism of osmoregulatory adaptation in tilapia. Molecular Biology Reports 2013; 40(2), 925-931.
SB Weiss, EP Kennedy and JY Kiyasu. The enzymatic synthesis of triglycerides. The Journal of Biological Chemistry 1960; 235, 40-44.
H Zhong, Y Zhou, H Zhang and W Xiao. DNA methylation pattern is associated with elevated expression of DGAT2 in hybrid tilapia. Aquaculture Nutrition 2021; 27(5), 1750-1760.
J Zhu, J Zhu, X Wang, Y Huang, C Qin, F Qiao, J Qin and L Chen. Alleviation of the adverse effect of dietary carbohydrate by supplementation of myo-inositol to the diet of Nile tilapia (Oreochromis niloticus). Animals 2020; 10(11), 2190.
F Chen, Y He, X Li, H Zhu, Y Li and D Xie. Improvement in muscle fatty acid bioavailability and volatile flavor in tilapia by dietary α-linolenic acid nutrition strategy. Foods 2024; 13(7), 1005.
VA Drover, DV Nguyen, CC Bastie, YF Darlington, NA Abumrad, JE Pessin, E London, D Sahoo and MC Phillips. CD36 mediates both cellular uptake of very long chain fatty acids and their intestinal absorption in mice. Journal of Biological Chemistry 2008; 283(19), 13108-13115.
IA Zani, SL Stephen, NA Mughal, D Russell, S Homer-Vanniasinkam, SB Wheatcroft and S Ponnambalam. Scavenger receptor structure and function in health and disease. Cells 2015; 4(2), 178-201.
ML Albert, SF Pearce, LM Francisco, B Sauter, P Roy, RL Silverstein and N Bhardwaj. Immature dendritic cells phagocytose apoptotic cells via αvβ5 and CD36, and cross-present antigens to cytotoxic T lymphocytes. The Journal of Experimental Medicine 1998; 188(7), 1359-1368.
H Hu, H Xia, L Xu, Z Wang, Y Lu and J Jian. The scavenger receptor CD36 protects Nile tilapia (Oreochromis niloticus) against Streptococcus agalactiae infection and promotes the expression of inflammatory factors. Aquaculture 2023; 572, 739501.
X Zhang, H Zhong, Z Han, Z Tang, J Xiao, Z Guo, F Wang, Y Luo and Y Zhou. Effects of waterborne exposure to 17β-estradiol on hepatic lipid metabolism genes in tilapia (Oreochromis niloticus). Aquaculture Reports 2020; 17, 100382.
YJ Chen, TY Zhang, HY Chen, SM Lin, L Luo and DS Wang. An evaluation of hepatic glucose metabolism at the transcription level for the omnivorous GIFT tilapia, Oreochromis niloticus, during postprandial nutritional status transition from anabolism to catabolism. Aquaculture 2017; 473, 375-382.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






