Oogenesis and Spermatogenesis of the Triploid Black Tiger Shrimp, Penaeus monodon
DOI:
https://doi.org/10.48048/tis.2024.8217Keywords:
Penaeus monodon, Triploid, Oogenesis, Spermatogenesis, Synaptonemal complexAbstract
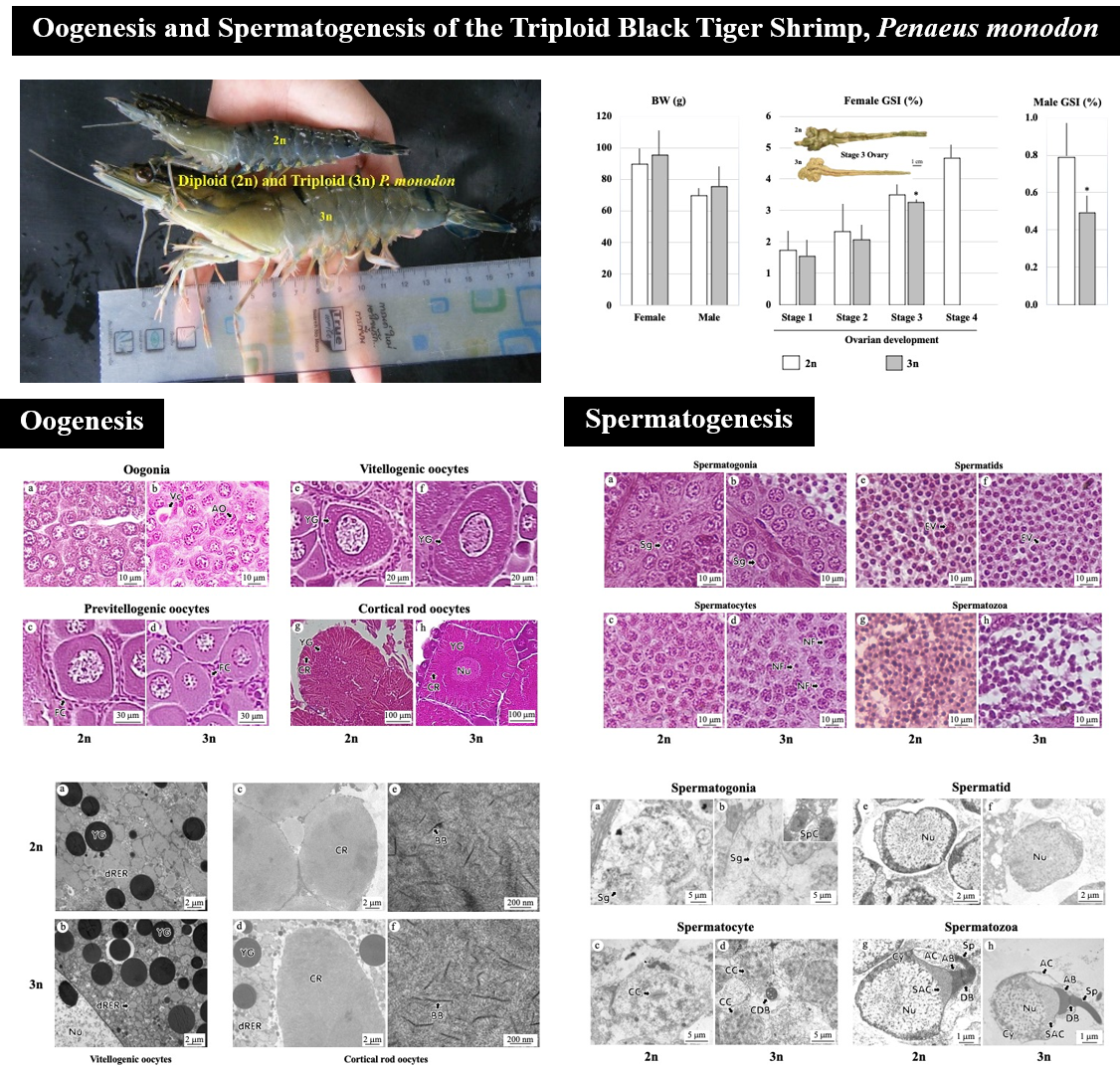
Triploidy (3n) induction of Penaeus monodon was performed separately by Australian and Thai groups of researchers 14 years ago, incidentally at the same period. The Australian group employed the chemical induction method, while the Thai group the cold shock one, and both groups obtained 3n P. monodon in their attempts. The success has led to several studies on the physiology of the 3n P. monodon, including growth, survival and reproductive functions. Both groups reported sterility of the 3n shrimp. The Australian group reported defected oogenesis and spermatogenesis of the 3n shrimp examined at subadult stage. Since the evaluation of the reproductive functions should be performed at the adult stage of the animals, we, therefore, performed in-depth studies on oogenesis and spermatogenesis of adult 3n P. monodon induced by cold shock. The studies include gross observation of the ovary, light (LM) and transmission electron (TEM) microscopy and image analysis of sex cells. The ovary of the 3n P. monodon females could not develop to full-maturation and their gonadosomatic index (GSI) was significantly lower than that of the 2n shrimp. Oogenesis of the 3n females proceeded from oogonia to cortical rod oocytes, but with significantly lower percentage than that of the 2n shrimp. The GSI of the 3n males was significantly lower than that of the 2n shrimp. Their spermatogenesis, however, proceeded normally from spermatogonia to spermatozoa, but with low density of spermatozoa in the seminiferous tubule and vas deferens, and none in the terminal ampoule. The average nuclear area of the 3n spermatozoa was significantly lower than that of the 2n shrimp as well. Altogether, these results suggest that 3n P. monodon induced by cold shock had defect in both oogenesis and spermatogenesis, probably more in quantity than in quality.
HIGHLIGHTS
- Oogenesis and spermatogenesis of the triploid black tiger shrimp, monodon, induced by cold shock were studied.
- The study found abnormalities in the spermatogenesis, but not oogenesis, of the triploid P. monodon induced by cold shock.
- It may be concluded that triploidy condition of P. monodon induced by cold shock affects meiotic, but not mitotic activity of the sex cells.
GRAPHICAL ABSTRACT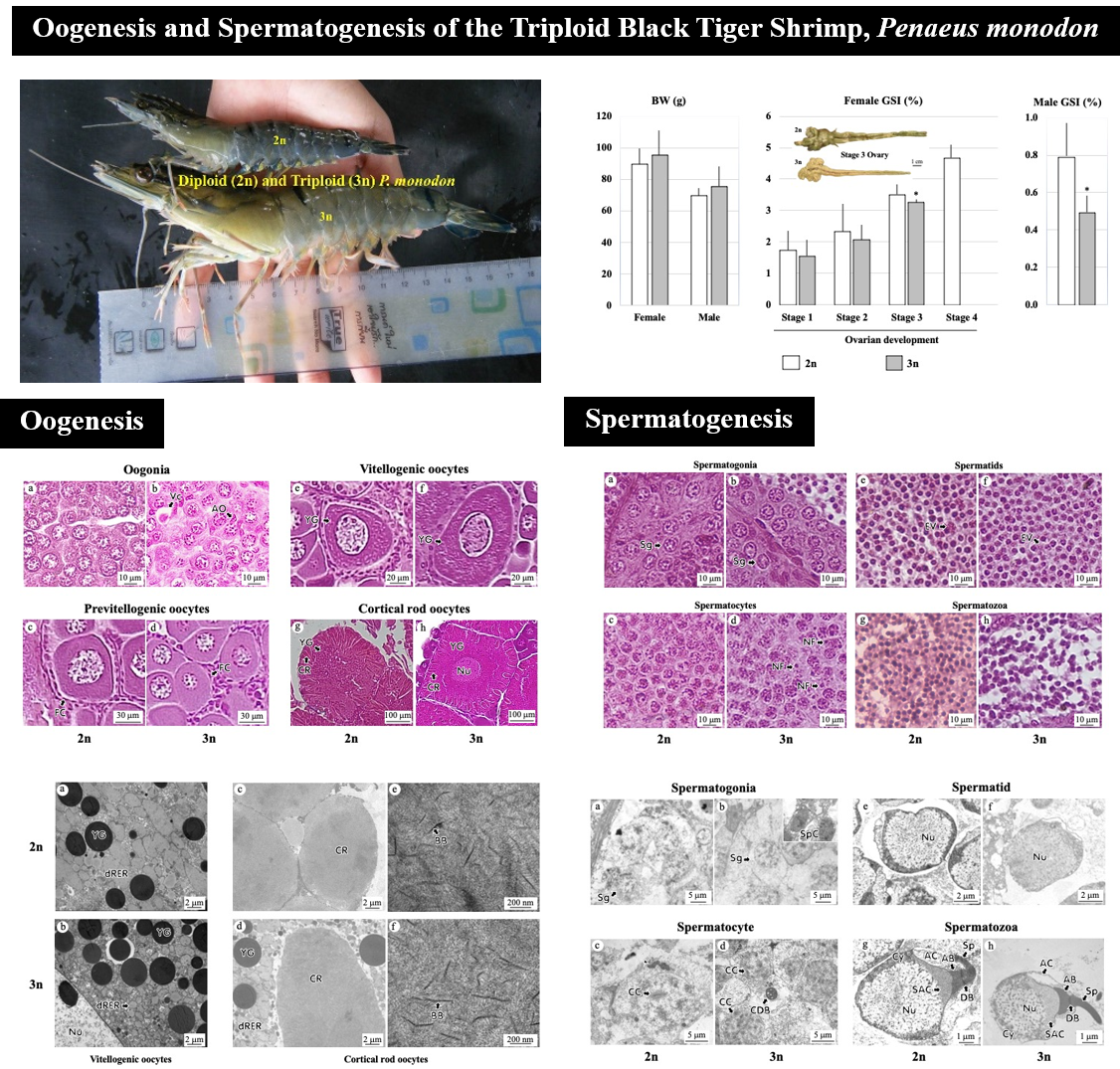
Downloads
References
P Warinyupa. 2020, Shrimp farming in Thailand: A pathway to sustainability. Thesis. Pomona College, Claremont, California.
B Withyachumnarnkul. Results from black tiger shrimp Penaeus monodon culture ponds stocked with postlarvae PCR-positive or -negative for white-spot syndrome virus (WSSV). Dis. Aquat. Org. 1999; 39, 21-7.
L Tran, L Nunan, RM Redman, LL Mohney, CR Pantoja, K Fitzsimmons and DV Lightner. Determination of the infectious nature of the agent of acute hepatopancreatic necrosis syndrome affecting penaeid shrimp. Dis. Aquat. Org. 2013; 105, 45-55.
T Chaijarasphong, N Munkongwongsiri, GD Stentiford, DJ Aldama-Cano, K Thansa, TW Flegel, K Sritunyalucksana and O Itsathitphaisarn. The shrimp microsporidian Enterocytozoon hepatopenaei (EHP): Biology, pathology, diagnostics and control. J. Invertebr. Pathol. 2021; 186, 107458.
S Tourtip, S Wongtripop, GD Stentiford, KS Bateman, S Sriurairatana, J Chavadej, K Sritunyalucksana and B Withyachumnarnkul. Enterocytozoon hepatopenaei sp. nov. (Microsporida: Enterocytozoonidae), a parasite of the black tiger shrimp Penaeus monodon (Decapoda: Penaeidae): Fine structure and phylogenetic relationships. J. Invertebr. Pathol. 2009; 102, 21-9.
MJ Sellars, A Wood, B Murphy, RM McCulloch and NP Preston. Triploid black tiger shrimp (Penaeus monodon) performance from egg to harvest egg. Aquaculture 2012; 324-5, 242-9.
P Pongtippatee, K Laburee, P Taweetumsevi, R Hiranphan, S Asuwapongpattana, W Weerachatyanukul, T Srisawat and B Withyachumnarnkul. Triploid Penaeus monodon: Sex ratio and growth rate. Aquaculture 2012; 356-7, 7-12.
P Pongtippatee, B Withyachumnarnkul and C Kerdmusik. Field evaluation of culture performance of triploid Penaeus monodon, compared with its diploid counterpart. Agr. Sci. Res. J. 2020; 10, 100-4.
JD Tan-Fermin and RA Pudadera. Ovarian maturation stages of the wild giant tiger prawn, Penaeus monodon Fabricius. Aquaculture 1989; 77, 229-42.
W Dall, BJ Hill, PC Rothlisberg and DJ Staples. Reproduction. In: JHS Blaxter and AJ Southward (Eds.). Advances in marine biology. Academic Press, London, 1990, p. 251-81.
H Motoh. Biology and ecology of Penaeus monodon. In: Proceedings of the 1st International Conference on the Culture of Penaeid Prawns/Shrimps, Iloilo City, Philippines. 1984, p. 27-36.
T Maoka. Carotenoids in marine animals. Mar. Drugs 2011; 9, 278-93.
A Babin, S Motreuil, M Teixeira, A Bauer, T Rigaud, J Moreau and Y Moreau. Origin of the natural variation in the storage of dietary carotenoids in freshwater amphipod crustaceans. PLoS One 2020; 15, e0231247.
LR D’Abramo, NA Baum, CE Bordner and DE Conklin. Carotenoids as a source of pigmentation in juvenile lobsters fed a purified diet. Can. J. Fish. Aquat. Sci. 1983; 40, 699-704.
W Wyban, G Martinez and J Sweeney. Adding paprika to Penaeus vannamei maturation diet improves nauplii quality. World Aquacult. 1997; 28, 59-62.
T Feng, B Paterson and SD Johnston. New insights into the spermatogenesis of the black tiger prawn, Penaeus monodon. J. Morphol. 2017; 278, 689-703.
R Hiransuchalert, N Thamniemdee, B Khamnamtong, K Yamano and S Klinbunga. Expression profiles and localization of vitellogenin mRNA and protein during ovarian development of the giant tiger shrimp Penaeus monodon. Aquaculture 2013; 412-3, 193-201.
SHK Tiu, JHL Hui, ASC Mak, JG He and SM Chan. Equal contribution of hepatopancreas and ovary to the production of vitellogenin (PmVg1) transcripts in the tiger shrimp, Penaeus monodon. Aquaculture 2006; 254, 666-74.
JJ Meusy and GG Payen. Female reproduction in malacostracan Crustacea. Zool. Sci. 1988; 5, 217-65.
LS Quackenbush. Yolk synthesis in the marine shrimp, Penaeus vannamei. Am. Zool. 2001; 41, 458-64.
P Pongtippatee-Taweepreda, J Chavadej, P Plodpai, B Pratoomchart, P Sobhon, W Weerachatyanukul and B Withyachumnarnkul. Egg activation in the black tiger shrimp Penaeus monodon. Aquaculture 2004; 234, 183-98.
P Pongtippatee, R Luppanakane, P Thaweethamsewee, P Kirirat, W Weerachatyanukul and B Withyachumnarnkul. Delay of the egg activation process in the black tiger shrimp Penaeus monodon by manipulation of magnesium levels in spawning water. Aquacult. Res. 2010; 41, 227-32.
H Kruevaisayawan, R Vanichviriyakit, W Weerachatyanukul, B Withyachumnarnkul, J Chavadej and P Sobhon. Oogenesis and formation of cortical rods in the black tiger shrimp, Penaeus monodon. Aquaculture 2010; 301, 91-8.
JW Lynn and HCJ Wallis. Physiological and biochemical investigations of the egg jelly release in Penaeus aztecus. Biol. Bull. 1987; 173, 451- 60.
FH Li, LH Zhou, JH Xiang, XD Liu and JZ Zhu. Triploid induction with heat shocks to Penaeus chinensis and their effects on gonad development. Chin. J. Oceanol. Limnol. 1999; 17, 57-61.
F Li, J Xiang, X Zhang, L Zhou, C Zhang and C Wu. Gonad development characteristics and sex ratio in triploid Chinese shrimp (Fenneropenaeus chinensis). Mar. Biotechnol. 2003; 5, 528-35.
JH Xiang, F Li, C Zhang, X Zhang, K Yu, L Zhou and C Wu. Evaluation of induced triploid shrimp Peneus (Fenneropenaeus) chinensis cultured under laboratory conditions. Aquaculture 2006; 259, 108-15.
X Guo and SK Allen. Viable tetraploids in the Pacific oyster (Crassostrea gigas Thuneberg) produced by inhibiting polar body 1 in eggs from triploids. Mol. Mar. Biol. Biotechnol. 1994; 3, 42-50.
N Cuñado, J Terrones, L Sánchez, P Martínez and JL Santos. Sex-dependent synaptic behaviour in triploid turbot, Scophthalmus maximus (Pisces, Scophthalmidae). Heredity 2002; 89, 460-4.
Y Xie, F Li, B Wang, S Li, D Wang, H Jiang, C Zhang, K Yu and J Xiang. Screening of genes related to ovary development in Chinese shrimp Fenneropenaeus chinensis by suppression subtractive hybridization. Comp. Biochem. Physiol. 2010; 5, 98-104.
LA Borin, IC Martins-Santos and C Oliveira. A natural triploid in Trichomycterus davisi (Siluriformes, Trichomycteridae): Mitotic and meiotic characterization by chromosome banding and synaptonemal complex analyses. Genetica 2002; 115, 253-8.
Y Xie, F Li, C Zhang, K Yu and J Xiang. Synaptonemal complex analysis in spermatocytes of diploid and triploid Chinese shrimp Fenneropenaeus chinensis. Tissue Cell. 2008; 40, 343-50.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






