Intercalation of Rhodamine B Dye into Zinc-Aluminum Layered Double Hydroxides: Structural, Textural, and Morphological Insights
DOI:
https://doi.org/10.48048/tis.2025.10225Keywords:
Layered double hydroxide, Rhodamine B, Intercalation, Zinc-aluminum layered double hydroxide, Co-precipitation methodAbstract
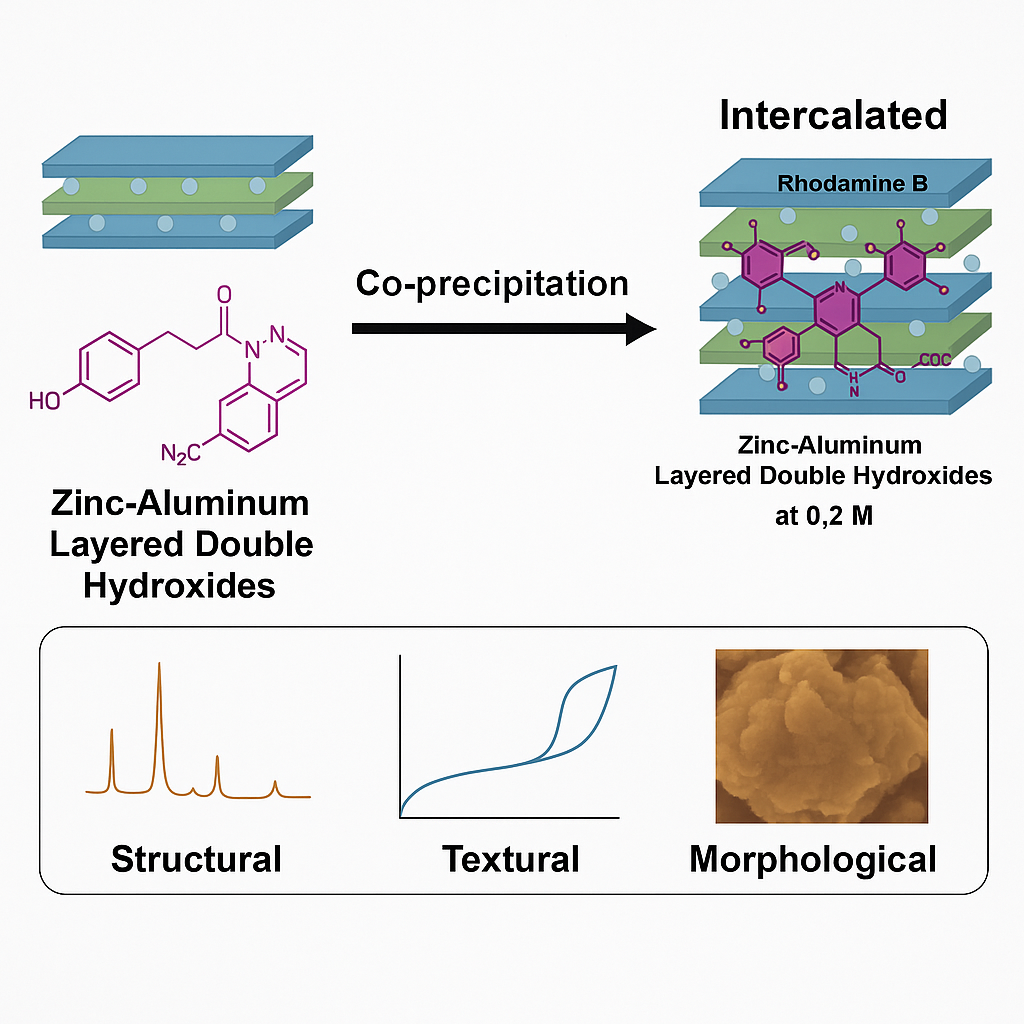
This research reports of Zinc-Aluminium layered double hydroxide material (ZAL) intercalated with the organic dye rhodamine b (ZARB) through a co-precipitation technique. The intercalation of ZAL with rhodamine b (RB) was conducted at various concentrations of rhodamine b ranging from 0.025 to 0.6 M, at a pH of 7.0 ± 0.5, and with an aging time of 18 h. ZAL and ZARB were characterized using PXRD, FTIR-ATR, BET, and SEM-EDX to confirm the intercalation of guest ions into the interlayer of the lamellar structure. The overall PXRD pattern of ZAL and the optimal concentration of ZARB at 0.2 M demonstrated an expansion of basal spacing from 8.9 to 11.0 Å, indicating successful intercalation of the ZAL compound with RB. The FTIR-ATR spectrum of ZAL and ZARB at 0.2 M exhibited peaks at 3,375 cm−1 (O-H), 1,617 cm−1 (C=C), 1,097 cm−1 (C-O), 758 and 608 cm−1 (Zn-OH and Al-OH), while small peaks at 1,334 cm−1 (N-O) indicated that RB was intercalated between the layered structure. The spatial orientation of ZARB at 0.2 M exhibited an interlayer region value of 6.19 Å for the ZAL nanocomposites. BET analysis revealed that ZAL and ZARB exhibited Type IV nitrogen adsorption-desorption isotherms with H1 hysteresis loops. ZAL surface area was 5.19 m2g−1, BJH desorption pore volume is 0.0238 cm3g−1 and BJH average pore diameter is 18.05 nm. Upon modification with RB, ZARB surface area increased to 14.30 m2g−1, BJH desorption pore volume to 0.0538 cm3g−1 and BJH average pore diameter is 20.27 nm. SEM-EDX morphology of ZAL revealed aggregated hexagonal plate-like with non-uniform sizes and shapes. Upon intercalation with RB into the layered double hydroxide, the structure formed flaky plate-like particles with a higher surface area. These results demonstrate the effective intercalation of RB into the ZAL framework, resulting in a nanocomposite with modified structural and morphological properties.
HIGHLIGHTS
- Successful intercalation of Rhodamine B (RB) into zinc-aluminum layered double hydroxide (ZAL) was achieved at an optimal concentration of 0.2 M, with basal spacing expansion from 8.9 to 11.0 Å confirmed by PXRD and FTIR-ATR.
- BET analysis showed a significant increase in surface area (from 5.19 to 14.30 m²/g) and porosity upon intercalation, indicating improved textural properties of the resulting ZARB nanocomposite.
- Morphological transformation was observed via SEM-EDX, where ZAL’s hexagonal plate-like structure changed to flaky plate-like particles with higher surface area after intercalation.
- Spatial modeling suggested a vertical orientation of RB anions within the interlayer region (6.19 Å), consistent with the intercalation-induced structural changes.
- ZARB nanocomposite demonstrated potential for environmental remediation and sensing applications by effectively encapsulating RB, reducing its leaching and enhancing material stability.
GRAPHICAL ABSTRACT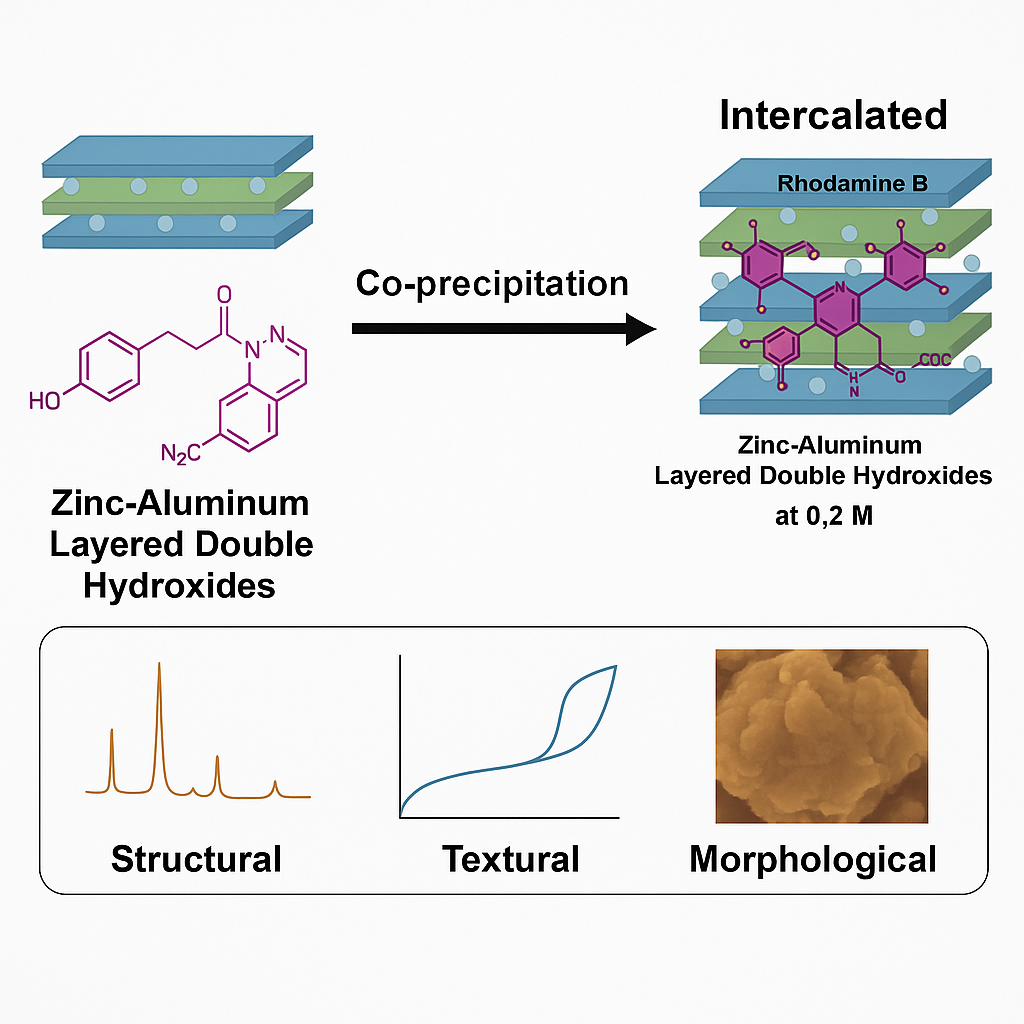
Downloads
References
MA Al-Hamyd, AS Al-Asadi and MF Al-Mudhaffer. Preparation and characterization of zinc-aluminum layered doubled hydroxide/ graphene nanosheets composite for supercapacitor el ectrode. Physica E: Low-Dimensional Systems and Nanostructures 2022; 136, 115005.
FL Bohari, SAISM Ghazali, NN Dzulkifl, SNA Baharin, I Fatimah and S Poddar. Studies on the intercalation of calcium-aluminium layered double hydroxide-MCPA and its controlled release mechanism as a potential green herbicide. Open Chemistry 2023; 20(1), 20220291.
M Ebadi, K Buskaran, S Bullo, MZ Hussein, S Fakurazi and G Pastorin. Drug delivery system based on magnetic iron oxide nanoparticles coated with (polyvinyl alcohol-zinc/aluminium-layered double hydroxide-sorafenib). Alexandria Engineering Journal 2021; 60(1), 733-743.
SAISM Ghazali, SH Sarijo and MZ Hussein. New synthesis of binate herbicide-interleaved anionic clay material: Synthesis, characterization and simultaneous controlled-release properties. Journal of Porous Materials 2021; 28, 495-505.
OMA Halim, NH Mustapha, SNM Fudzi, R Azhar, NIN Zanal, NF Nazua, AH Nordin, MSM Azami, MAM Ishak, W Ismail and Z Ahmad. A review on modified ZnO for the effective degradation of Methylene Blue and Rhodamine B. Results in Surfaces and Interfaces 2025; 18, 100408.
S Iftekhar, ME Kucuk, V Srivastava, E Repo and M Sillanpaa. Application of zinc-aluminium layered double hydroxides for adsorptive removal of phosphate and sulfate: Equilibrium, kinetic and thermodynamic. Chemosphere 2018; 209, 470-479.
SH Jasim and AS Al-Asadi. Graphene oxide/zinc-aluminum double layered hydroxide nanocomposite for hybrid supercapacitor. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2025; 709, 136113.
J Karthikeyan, H Fjellvag, S Bundli and AO Sjastad. Efficient exfoliation of layered double hydroxides; effect of cationic ratio, hydration state, anions and their orientations. Materials 2021; 14(2), 346.
NASM Sandollah., SAISM Ghazali, WNW Ibrahim and R Rusmin. Adsorption-desorption profile of methylene blue dye on raw and acid activated kaolinite. Indonesian Journal of Chemistry 2020; 20(4), 755-765.
LBND Farias, GG Carbajal-Arizaga, LGG Sante, L Effting, JACDS Fernandesa and A Bail. Nanoflakes of chloride zinc-iron-aluminum-based layered double hydroxides obtained from industrial waste: A green approach to mass-scale production. RSC Advances 2021; 11(29), 17760-17768.
A Sharma, S Kumari, S Sharma, T Singh, S Kumar, A Thakur, SK Bhatiam and AK Sharma. Layered double hydroxides: An insight into the role of hydrotalcite-type anionic clays in energy and environmental applications with current progress and recent prospects. Materials Today Sustainability 2023; 22, 100399.
A Yu, Y Song, N Wang, Y Tian and H Chen. Study on the corrosion and self-healing behavior of different anion-intercalated layered double hydroxides coatings on Mg alloy surfaces. Surfaces and Interfaces 2025; 56, 105680.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






