Pathavi Apo Vayo Formulary Extract Treatment Under High Glucose Conditions on Foam Cell Formation in RAW264.7 Macrophages
DOI:
https://doi.org/10.48048/tis.2025.10210Keywords:
Pathavi Apo Vayo formulary, High glucose, oxLDL, Foam cell, LOX-1, ProteomicsAbstract
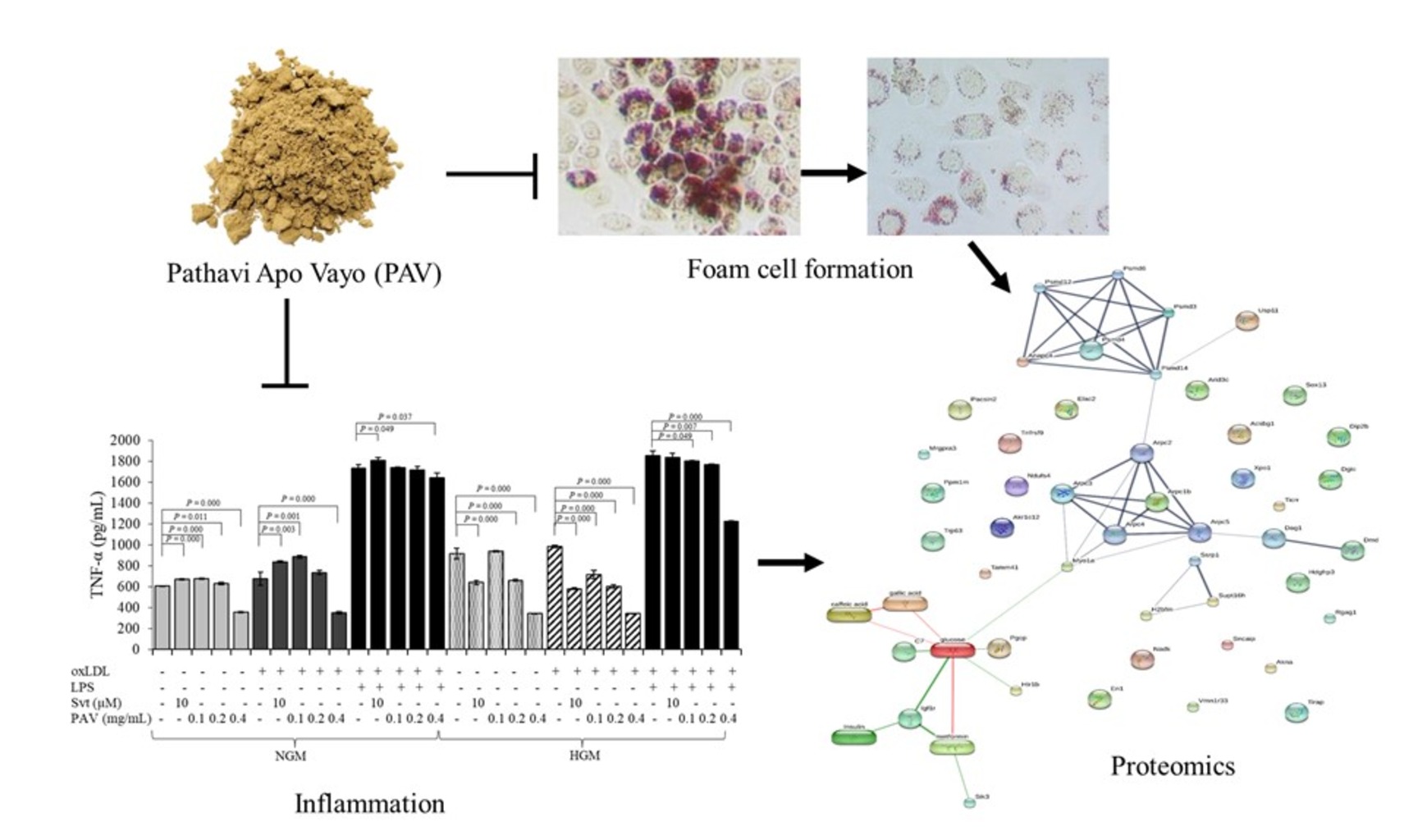
The objective of the present study was to investigate the effect of Pathavi Apo Vayo formulary extract (PAV) extracted by 50% ethanol on foam cell formation of macrophages (RAW264.7), in high glucose medium (HGM). Foam cell formation and lipid accumulation, gene expression, cytokine production, proteomic analysis, protein difference and protein-chemical correlation were studied using Oil Red O staining, Real-Time PCR, ELISA, Liquid Chromatography-Tandem Mass Spectrometry (LC/MS-MS), and bioinformatics (Venn diagram and STITCH), respectively. Oxidized low-density lipoprotein (oxLDL) was used to induce the formation of foam cells. In oxLDL induction, PAV co-treatment suppressed lipid accumulation in foam cells slightly, while a higher concentration of PAV (200 - 400 µg/mL) increased the lipid accumulation in the treated cells slightly. Similarly, PAV up-regulated the gene expressions of oxLDL receptors including LOX-1. However, PAV down-regulated the expression of pro-inflammatory cytokine genes including TNF-α. In addition, PAV also decreased cytokine production after co-treatment with oxLDL and LPS in both normal glucose medium (NGM) and HGM. In total 7,192 proteins were identified from 6 glucose medium conditions using shotgun proteome analysis, 42 of which were expressed only in HGM and HGM +400 µg/mL PAV conditions. A proteome mechanism during the process of PAV treatment under high glucose conditions is proposed. In conclusion, PAV could slightly decrease foam cell formation, but might increase lipid accumulation in foam cells when using higher concentrations. Therefore, this traditional medicine should be used carefully in hyperlipidemia and diabetes patients.
HIGHLIGHTS
- Pathavi Apo Vayo formulary (PAV) is one Thai traditional medicine for treating diabetes.
- Forty-two proteins were detected uniquely in high glucose and high glucose + 400 µg/mL PAV conditions.
- PAV treatment under high glucose conditions correlated with the reduction of pro-inflammatory cytokines and was associated with lipid accumulation of foam cell formation in RAW264.7 macrophages.
GRAPHICAL ABSTRACT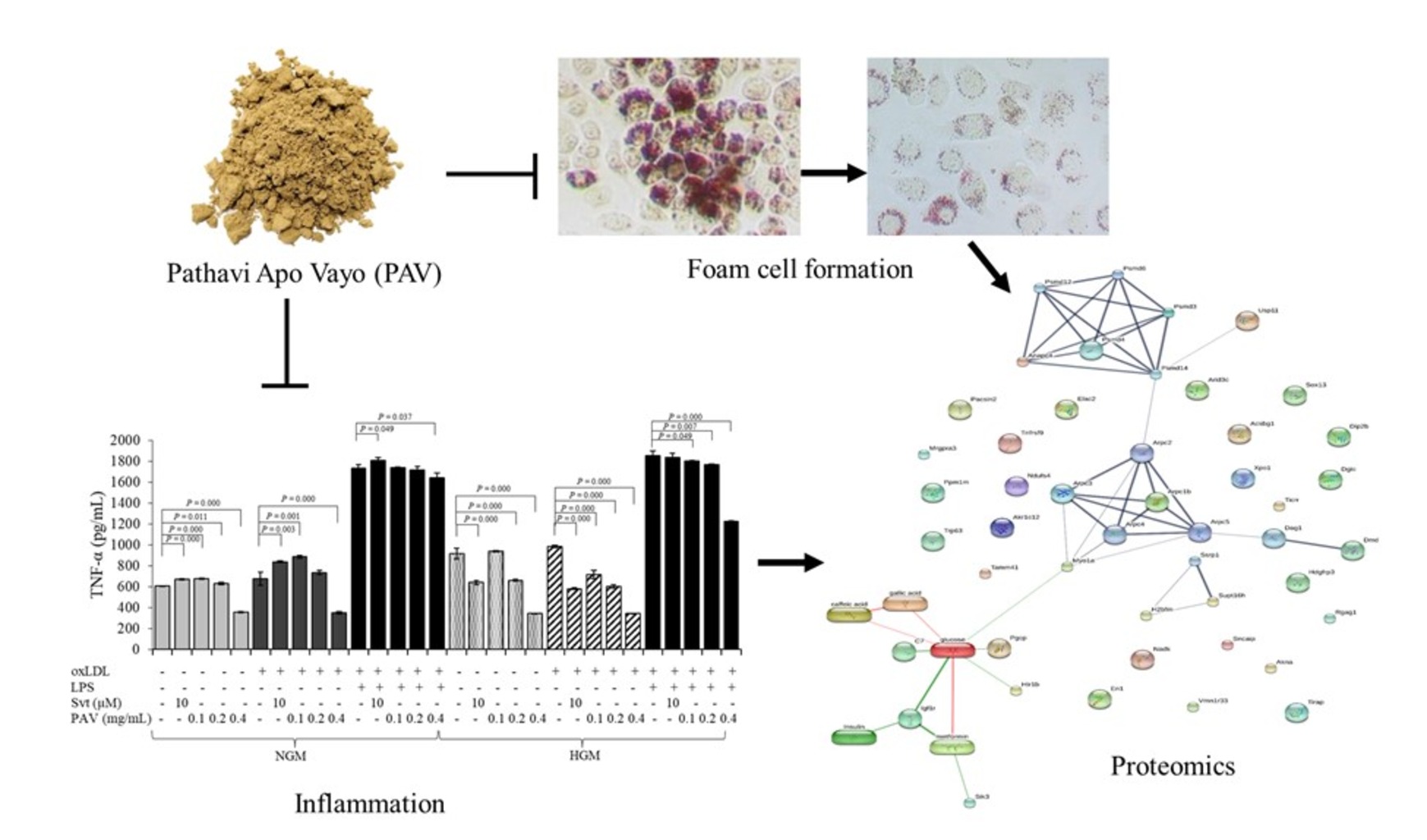
Downloads
References
A Poznyak, AV Grechko, P Poggio, VA Myasoedova, V Alfieri and AN Orekhov. The diabetes mellitus-atherosclerosis connection: the role of lipid and glucose metabolism and chronic inflammation. International Journal of Molecular Sciences 2020; 21(5), 1835.
ZC Wang, JO Machuki, MZ Li, KX Li and HJ Sun. A narrative review of plant and herbal medicines for delaying diabetic atherosclerosis: an update and future perspectives. Reviews in Cardiovascular Medicine 2021; 22(4), 1361-1381.
C Alan and KE Bornfeldt. Diabetes and atherosclerosis: is there a role for hyperglycemia? Journal of Lipid Research 2009; 50(S), S335-S339.
A Pirillo, GD Norata and AL Catapano. LOX-1, OxLDL, and atherosclerosis. Mediators of Inflammation 2013; 2013, 152786.
DF Schaeffer, M Riazy, KS Parhar, JH Chen, V Duronio, T Sawamura and UP Steinbrecher. LOX-1 augments oxLDL uptake by lysoPC-stimulated murine macrophages but is not required for oxLDL clearance from plasma. Journal of Lipid Research 2009; 50(8), 1676-1684.
L Li, T Sawamura and G Renier. Glucose enhances human macrophage LOX-1 expression: role for LOX-1 in glucose-induced macrophage foam cell formation. Circulation Research 2004; 94(7), 892-901.
M Nagase, J Abe, K Takahashi, J Ando, S Hirose and T Fujita. Genomic organization and regulation of expression of the lectin-like oxidized low-density lipoprotein receptor (LOX-1) gene. The Journal of Biological Chemistry 1998; 273(50), 33702-33707.
N Kume, T Murase, H Moriwaki, T Aoyama, T Sawamura, T Masaki and T Kita. Inducible expression of lectin-like oxidized LDL receptor-1 in vascular endothelial cells. Circulation Research 1998; 83(3), 322-327.
M Hashizume and M Mihara. Blockade of IL-6 and TNF- inhibited oxLDL-induced production of MCP-1 via scavenger receptor induction. European Journal of Pharmacology 2012; 689(1-3), 249-254.
J Persson, J Nilsson and MW Lindholm. Interleukin-1beta and tumor necrosis factor-alpha impede neutral lipid turnover in macrophage-derived foam cells. BMC Immunology 2008; 9(1), 1-11.
M Hashizume and M Mihara. Atherogenic effects of TNF- and IL-6 via up-regulation of scavenger receptors. Cytokine 2012; 58(3), 424-430.
CH Kim, KA Han, HJ Oh, KE Tan, R Sothiratnam and A Tjokroprawiro. Safety, tolerability, and efficacy of metformin extended-release oral antidiabetic therapy in patients with type 2 diabetes: an observational trial in Asia. Journal of Diabetes 2012; 4(4), 395-406.
C Andrade, NGM Gomes, S Duangsrisai, PB Andrade, DM Pereira and P Valentão. Medicinal plants utilized in Thai traditional medicine for diabetes treatment: Ethnobotanical surveys, scientific evidence and phytochemicals. Journal of Ethnopharmacology 2020; 263, 113177.
P Peungvicha, O Vallisuta, S Mangmool, T Sirithamwanich and R Sirithamwanich. Anti-hyperglycemic effect and subchronic toxicity of the combined extract from Sattagavata-Mathurameha-Tubpikarn anti-diabetic herbal formulae. Thai Journal of Pharmaceutical Sciences 2018; 42(1), 6-13.
B Prajanban, O Jaisamut and N Fangkrathok. Quality control, cytotoxicity and inhibitory effect on nitric oxide production of Pathavi Apo Vayo formulary extract. Huachiew Chalermprakiet Science and Technology Journal 2022; 8(2), 92-106.
R Li, T Chaicherdsakul, V Kunathigan, S Roytrakul, A Paemanee and S Kittisenachai. Shotgun proteomic analysis of germinated rice (Oryza sativa L.) under salt stress. Applied Science and Engineering Progress 2020; 13(1), 76-85.
H Kim, J Chen, B Prescott, ME Walker, ME Grams, B Yu, RS Vasan, JS Floyd, N Sotoodehnia, NL Smith, DE Arking, J Coresh and CM Rebholz. Plasma proteins associated with plant-based diets: results from the atherosclerosis risk in communities (ARIC) study and framingham heart study (FHS). Clinical Nutrition 2024; 43(8), 1929-1940.
MR Rooney, J Chen, JB Echouffo-Tcheugui, KA Walker, P Schlosser, A Surapaneni, O Tang, J Chen, CM Ballantyne, E Boerwinkle, CE Ndumele, RT Demmer, JS Pankow, PL Lutsey, LE Wagenknecht, Y Liang, X Sim, R Dam, ES Tai, ME Grams, …, J Coresh. Proteomic predictors of incident diabetes: Results from the atherosclerosis risk in communities (ARIC) study. Diabetes Care 2023; 46(4), 733-741.
B Huang, M Jin, H Yan, Y Cheng, D Huang, S Ying and L Zhang. Simvastatin enhances oxidized‑low density lipoprotein‑induced macrophage autophagy and attenuates lipid aggregation. Molecular Medicine Reports 2015; 11(2), 1093-1098.
X Yang, M Yin, L Yu, M Lu, H Wang, F Tang and Y Zhang. Simvastatin inhibited oxLDL-induced proatherogenic effects through calpain-1-PPARγ-CD36 pathway. Canadian Journal of Physiology and Pharmacology 2016; 94(12), 1336-1343.
ZH Wu, YQ Chen and SP Zhao. Simvastatin inhibits ox-LDL-induced inflammatory adipokines secretion via amelioration of ER stress in 3T3-L1 adipocyte. Biochemical and Biophysical Research Communications 2013; 432(2), 365-369.
T Hu, B Chen, S Zhou and J Mao. Simvastatin inhibits inflammatory response in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages through the microRNA-22/Cyr61 axis. International Journal of Clinical and Experimental Pathology 2018; 11(8), 3925-3933.
M Rahmati-Yamchi, S Ghareghomi, G Haddadchi, M Milani, M Aghazadeh and H Daroushnejad. Fenugreek extract diosgenin and pure diosgenin inhibit the hTERT gene expression in A549 lung cancer cell line. Molecular Biology Reports 2014; 41(9), 6247-6252.
G Tian, D Wilcockson, VH Perry, PM Rudd, RA Dwek, FM Platt and N Platt. Inhibition of alpha-glucosidases I and II increases the cell surface expression of functional class A macrophage scavenger receptor (SR-A) by extending its half-life. The Journal of Biological Chemistry 2004; 279(38), 39303-39309.
N Fangkrathok, J Junlatat and B Sripanidkulchai. In vivo and in vitro anti-inflammatory activity of Lentinus polychrous extract. Journal of Ethnopharmacology 2013; 147(3), 631-637.
S Tyanova, T Temu, P Sinitcyn, A Carlson, MY Hein, T Geiger, M Mann and J Cox. The Perseus computational platform for comprehensive analysis of proteomics data. Nature Methods 2016; 13(9), 731-740.
EA Howe, R Sinha, D Schlauch and J Quackenbush. RNA-Seq analysis in MeV. Bioinformatics 2011; 27(22), 3209-3210.
H Mi, A Muruganujan, D Ebert, X Huang and PD Thomas. PANTHER version 14: More genomes, a new PANTHER GO-slim and improvements in enrichment analysis tools. Nucleic Acids Research 2019; 47(D1), D419-D426.
P Bardou, J Mariette, F Escudié, C Djemiel and C Klopp. jvenn: An interactive Venn diagram viewer. BMC Bioinformatics 2014; 15(1), 293.
D Szklarczyk, A Santos, C von Mering, LJ Jensen, P Bork and M Kuhn. STITCH 5: augmenting protein-chemical interaction networks with tissue and affinity data. Nucleic Acids Research 2016; 44(D1), D380-D384.
A Roy, U Saqib, K Wary and MS Baig. Macrophage neuronal nitric oxide synthase (NOS1) controls the inflammatory response and foam cell formation in atherosclerosis. International Immunopharmacology 2020; 83(1), 106382.
B Huang, M Jin, H Yan, Y Cheng, D Huang and S Ying. Simvastatin enhances oxidized low density lipoprotein induced macrophage autophagy and attenuates lipid aggregation. Molecular Medicine Reports 2015; 11(2),1093-1098.
M Saxena, D Ali, DR Modi, MHA Almarzoug, SA Hussain and S Manohrdas. Association of TNF-α gene expression and release in response to anti-diabetic drugs from human adipocytes in vitro. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 2020; 13, 2633-2640.
A Ihantola-Vormisto, J Summanen, H Kankaanranta, H Vuorela, ZM Asmawi and E Moilanen. Anti-inflammatory activity of extracts from leaves of Phyllanthus emblica. Planta Medica 1997; 63(6), 518-524.
M Tanaka, Y Kishimoto, E Saita, N Suzuki-Sugihara, T Kamiya, C Taguchi, K Iida and K Kondo. Terminalia bellirica extract inhibits low-density lipoprotein oxidation and macrophage inflammatory response in vitro. Antioxidants 2016; 5(2), 20.
A Gonen, D Harats, A Rabinkov, T Miron, D Mirelman, M Wilchek, L Weiner, E Ulman, H Levkovitz, D Ben-Shushan and A Shaish. The antiatherogenic effect of allicin: possible mode of action Pathobiology 2005; 72(6), 325-334.
D Patel, S Desai, T Gajaria, R Devkar and AV Ramachandran. Coriandrum sativum L. seed extract mitigates lipotoxicity in RAW 264.7 cells and prevents atherogenic changes in rats. EXCLI Journal 2013; 12, 313-334.
GA Agbor, JA Vinson, J Sortino and R Johnson. Antioxidant and anti-atherogenic activities of three Piper species on atherogenic diet fed hamsters. Experimental and Toxicologic Pathology 2012; 64(4), 387-391.
N Oršolić, D Sirovina, D Odeh, G Gajski, V Balta, L Šver and MJ Jembrek, Efficacy of caffeic acid on diabetes and its complications in the mouse. Molecules 2021; 26(11), 3262.
M Tanaka, A Sato, Y Kishimoto, H Mabashi-Asazuma, K Kondo and K Iida. Gallic acid inhibits lipid accumulation via AMPK pathway and suppresses apoptosis and macrophage-mediated inflammation in hepatocytes. Nutrients 2020; 12(5), 1479.
J Boucher, A Kleinridders and CR Kahn. Insulin receptor signaling in normal and insulin-resistant states. Cold Spring Harbor Perspectives in Biology 2014; 6(1), a009191.
DB Jacobs, GR Hayes, JA Truglia and DH Lockwood. Effects of metformin on insulin receptor tyrosine kinase activity in rat adipocytes. Diabetologia 1986; 29(11), 798-801.
R Al-Zoairy, MT Pedrini, MI Khan, J Engl, A Tschoner, C Ebenbichler, G Gstraunthaler, K Salzmann, R Bakry and A Niederwanger. Serotonin improves glucose metabolism by serotonylation of the small GTPase Rab4 in L6 skeletal muscle cells. Diabetology and Metabolic Syndrome 2017; 9(1), 1.
Z Sun, Q Jiang, J Li and J Guo. The potent roles of salt-inducible kinases (SIKs) in metabolic ho¬meostasis and tumorigenesis. Signal Transduction and Targeted Therapy 2020; 5(1), 150.
M Okamoto, H Takemori and Y Katoh. Salt-inducible kinase in steroidogenesis and adipogenesis. Trends in Endocrinology and Metabolism 2004; 15(1), 21-26.
T Uebi, Y Itoh, O Hatano, A Kumagai, M Sanosaka, T Sasaki, S Sasagawa, J Doi, K Tatsumi, K Mitamura, E Morii, K Aozasa, T Kawamura, M Okumura, J Nakae, H Takikawa, T Fukusato, M Koura, M Nish, A Hamsten, …, H Takemori. Involvement of SIK3 in glucose and lipid homeostasis in mice. PLoS One 2012; 7(5), e37803.
S Grès, S Canteiro, J Mercader and C Carpéné. Oxidation of high doses of serotonin favors lipid accumulation in mouse and human fat cells. Molecular Nutrition and Food Research 2013; 57(6), 1089-1099.
R Aarsetøy, T Ueland, P Aukrust, AE Michelsen, RL Fuente, H Grundt, H Staines, O Nygaard and D WT Nilsen. Complement component 7 is asso¬ciated with total- and cardiac death in chest-pain patients with suspected acute coronary syndrome. BMC Cardiovascular Disorders 2021; 21(1), 496.
SI Vlaicu, A Tatomir, D Boodhoo, S Vesa, PA Mircea and H Rus. The role of complement system in adipose tissue-related inflammation. Immunologic Research 2016; 64(3), 653-664.
SI Vlaicu, A Tatomir, V Rus, AP Mekala, PA Mircea, F Niculescu and H Rus. The role of complement activation in atherogenesis: The first 40 years. Immunologic Research 2016; 64(1), 1-13.
NJ Darling, R Toth, JS Arthur and K Clark. Inhibition of SIK2 and SIK3 during differentiation enhances the anti-inflammatory phenotype of macrophages. The Biochemical journal 2017; 474(4), 521-537.
K Clark, KF MacKenzie, K Petkevicius, Y Kristariyanto, J Zhang, HG Choi, M. Peggie, L Plater, PGA Pedrioli, E McIver, NS Gray, JSC Arthur and P Cohen. Phosphorylation of CRTC3 by the salt-inducible kinases controls the interconversion of classically activated and regulatory macrophages. Proceedings of the National Academy of Sciences of the United States of America 2012; 109(42), 16986-16991.
SY Kim, S Jeong, KH Chah, E Jung, KH Baek, ST Kim, JH Shim, E Chun and KY Lee. Salt-inducible kinases 1 and 3 negatively regulate Toll-like receptor 4-mediated signal. Molecular Endocrinology 2013; 27(11), 1958-1968.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






