Biwa (Eriobotrya japonica) Leaf Nanoherbal for Diabetes Therapy: In Silico and In Vivo Study on Blood Glucose and Pancreatic Histology
DOI:
https://doi.org/10.48048/tis.2025.9930Keywords:
Eriobotrya japonica, Nanoherbal, Antidiabetic therapy, In Silico, In Vivo, Molecular dockingAbstract
Diabetes mellitus is a global metabolic disorder with an increasing prevalence. This study investigates the antidiabetic potential of Biwa (Eriobotrya japonica) leaf nanoherbal through in silico and in vivo approaches. A total of 12 bioactive compounds from E. japonica leaf extract were screened using in silico approaches to assess their pharmacokinetics, toxicity, and molecular interactions with PTP1B and DPP4. Molecular docking analysis revealed that 11 out of 12 compounds exhibited strong interactions with these key enzymes involved in glucose metabolism, particularly Quinic Acid (–7.7 kcal/mol) and Luteolin (–7.6 kcal/mol) for PTP1B, as well as Oleanolic Acid (–9.2 kcal/mol), Luteolin (–9.1 kcal/mol), and quercetin (–9.0 kcal/mol) for DPP4. For the in vivo study, 30 Wistar rats were divided into 5 groups, where the negative control [C(–)] received physiological saline, while the diabetic control [C(+)] and treatment groups were induced with alloxan (120 mg/kg BW). After diabetes induction, [T1] received E. japonica nanoherbal at 250 mg/kg BW, [T2] received E. japonica nanoherbal at 500 mg/kg BW, and [T3] received glibenclamide at 5 mg/kg BW. Blood glucose levels were monitored, and pancreatic histology was analyzed. The in vivo results showed that nanoherbal treatment significantly reduced blood glucose levels, with the 500 mg/kg BW dose decreasing glucose levels from 277.8 to 157.4 mg/dL on day 14. Histological analysis further demonstrated improved pancreatic islet structure and reduced cellular degeneration in treated groups. These findings suggest that E. japonica nanoherbal has strong antidiabetic potential by targeting glucose-regulating enzymes and enhancing pancreatic histology.
HIGHLIGHTS
- Strong interactions were observed in the molecular docking analysis between the key bioactive compounds of japonica, where Quinic Acid and luteolin exhibited strong binding affinities with PTP1B, while Oleanolic Acid, luteolin, and quercetin showed high affinities for DPP4.
- A novel nanoherbal formulation enhanced the bioavailability and efficacy of E. japonica bioactive compounds for diabetes therapy.
- Extensive ADMET profiling ensured the safety and suitability of the bioactive compounds for long-term therapeutic applications.
- Significant glucose level reduction was achieved in diabetic rats, dropping from 277.80 to 157.40 mg/dL in 14 days with a 500 mg/kg BW dosage.
- At higher doses, enhanced cellular regeneration in pancreatic tissues was observed, indicating potential protective effects against pancreatic damage caused by diabetes.
GRAPHICAL ABSTRACT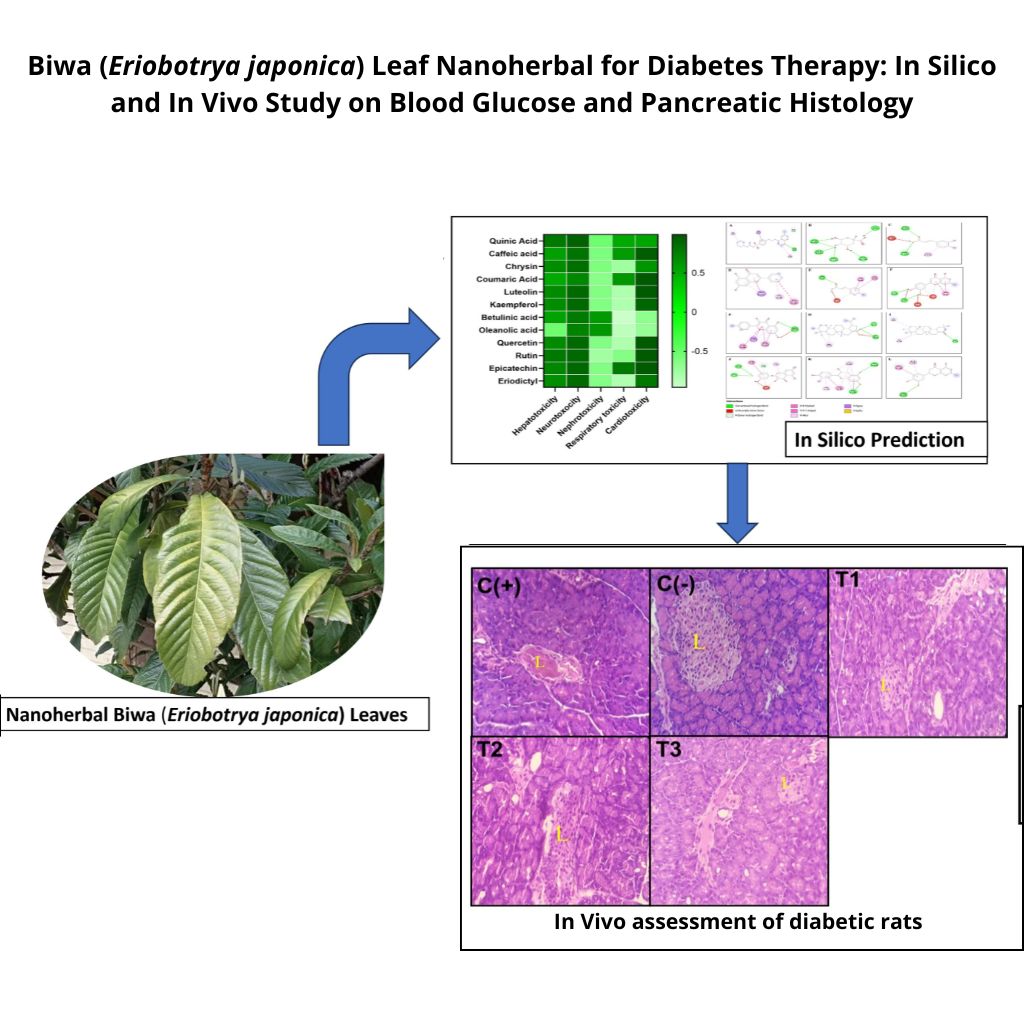
Downloads
References
Vaishali, N Sharma and A Chauhan. A subjective study of diabetes mellitus, its complications and homeopathic management. International Journal of Homoeopathic Sciences 2024; 8(1), 399-402.
R Jiang, Z Cong, L Zheng, L Zhang, Q Guan, S Wang, J Fang, J Chen and M Liu. Global research trends in regulating gut microbiome to improve type 2 diabetes mellitus: Bibliometrics and visual analysis. Frontiers in Endocrinology 2024; 15, 1401070.
R Rodriguez-Gutierrez, LG Mancillas-Adame, G Rodríguez-Tamez, AD Gonzalez-Colmenero, RC Solis-Pacheco, AS Elizondo-Plazas, KM Santos-Santillana, L Gonzalez-Sariñana, V Gonzalez-Nava and JG Gonzalez-Gonzalez. Hypertriglyceridemia and its association with HbA1c test: A prospective in vivo controlled study. International Journal of Endocrinology 2019; 2019, 4784313.
A Kulkarni, AR Thool and S Daigavane. Understanding the clinical relationship between diabetic retinopathy, nephropathy, and neuropathy: A comprehensive review. Cureus 2024; 16(3), e56674.
MF Brereton, M Rohm, K Shimomura, C Holland, S Tornovsky-Babeay, D Dadon, M Iberl, MV Chibalina, S Lee, B Glaser, Y Dor, P Rorsman, A Clark and FM Ashcroft. Hyperglycaemia induces metabolic dysfunction and glycogen accumulation in pancreatic β-cells. Nature Communications 2016; 7, 13496.
RW Sibony, O Segev, S Dor and I Raz. Drug therapies for diabetes. International Journal of Molecular Sciences 2023; 24(24), 17147.
X Xie, C Wu, Y Hao, T Wang, Y Yang, P Cai, Y Zhang, J Huang, K Deng, D Yan and H Lin. Benefits and risks of drug combination therapy for diabetes mellitus and its complications: A comprehensive review. Frontiers in Endocrinology 2023; 14, 1301093.
FE Hasibuan, W Syahfitri, S Ilyas and S Hutahaean. Phytochemical screening, antioxidant activity and thin-layer chromatography test of methanol extract and simplicia leaves of loquat (Eriobotrya japonica Lindl). In: Proceedings of the 3rd Nommensen International Conference on Technology and Engineering 2019, Medan, Indonesia. 2020, p. 12069.
BN Bisso, PN Kayoka-Kabongo, RT Tchuenguem and JP Dzoyem. Phytochemical analysis and antifungal potentiating activity of extracts from loquat (Eriobotrya japonica) against Cryptococcus neoformans clinical isolates. Advances in Pharmacological and Pharmaceutical Sciences 2022; 2022, 6626834.
Y Liu, W Zhang, C Xu and X Li. Biological activities of extracts from Loquat (Eriobotrya japonica Lindl.): A review. International Journal of Molecular Sciences 2016; 17(12), 1983.
W Syahfitri, F Elizabth, S Ilyas and S Hutahaen. Effect of loquat leaves methanol extract on morphology of benzo(A)pyrene induced rats. International Journal of Scientific and Technology Research 2020; 9(3), 1174-1177.
S Kumari, A Goyal, ES Gürer, EA Yapar, M Garg, M Sood and RK Sindhu. Bioactive loaded novel nano-formulations for targeted drug delivery and their therapeutic potential. Pharmaceutics 2022; 14(5), 1091.
H Zolkepli, RT Widodo, S Mahmood, N Salim, K Awang, N Ahmad and R Othman. A review on the delivery of plant-based antidiabetic agents using nanocarriers: Current status and their role in combatting hyperglycaemia. Polymers 2022; 14(15), 2991.
AFT Gomes, WFD Medeiros, I Medeiros, G Piuvezam, JKD Silva-Maia, IWL Bezerra and AHDA Morais. In silico screening of therapeutic targets as a tool to optimize the development of drugs and nutraceuticals in the treatment of diabetes mellitus: A systematic review. International Journal of Molecular Sciences 2024; 25(17), 9213.
Masfria, H Lucida, Y Atifah, H Syahputra and HM Sihombing. Inhibition activity of liquid smoke Cocos nucifera L. on Dpp-Iv and age-rage in silico and in vitro: Antidiabetic and anti-inflammatory activity. International Journal of Applied Pharmaceutics 2024; 16(5), 275-282.
S Shaikh, EJ Lee, K Ahmad, SS Ahmad, JH Lim and I Choi. A comprehensive review and perspective on natural sources as dipeptidyl peptidase-4 inhibitors for management of diabetes. Pharmaceuticals 2021; 14(6), 591.
PSR Dwivedi, P Khanal, VP Gaonkar, VP Rasal and BM Patil. Identification of PTP1B regulators from Cymbopogon citratus and its enrichment analysis for diabetes mellitus. In Silico Pharmacology 2021; 9(1), 30.
M Eddouks, D Chattopadhyay and NA Zeggwagh. Animal models as tools to investigate antidiabetic and anti-inflammatory plants. Evidence-based Complementary and Alternative Medicine 2012; 2012, 142087.
P Santoso, S Ilyas, YH Midoen and A Yuniarti. Protective effect of Vitis gracilis Wall (Vitaceae) leaf decoction on sexual vitality and testis of alloxan-induced diabetic mice. Traditional and Integrative Medicine 2023; 8(3), 256-268.
R Roskoski. Properties of FDA-approved small molecule protein kinase inhibitors: A 2024 update. Pharmacological Research 2024; 200, 107059.
DP Wati, S Ilyas and D Khairani. In silico study of compounds from nanoherbal jopan (Clibadium surinamense L.) leaves as inhibitors AKT1 interaction. Trends in Sciences 2024; 22(1), 8729.
T Shamsuddin, MA Hosen, MS Alam, TB Emran and SMA Kawsar. Uridine derivatives: Antifungal, PASS outcomes, ADME/T, drug-likeliness, molecular docking and binding energy calculations. Medicine Science International Medical Journal 2021; 10(4), 1373-1386.
P Rath, A Ranjan, A Ghosh, A Chauhan, M Gurnani, HS Tuli, H Habeeballah, MF Alkhanani, S Haque, K Dhama, NK Verma and T Jindal. Potential therapeutic target proteina tyrosine phosphatase-1B for modulation of insulin resistance with polyphenols and its quantitative structure - activity relationship. Molecules 2022; 27(7), 2212.
G Xiong, Z Wu, J Yi, L Fu, Z Yang, C Hsieh, M Yin, X Zeng, C Wu, A Lu, X Chen, T Hou and D Cao. ADMETlab 2.0: An integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic Acids Research 2021; 49(W1), W5-W14.
A Jalili, R Bagherifar, A Nokhodchi, B Conway and Y Javadzadeh. Current advances in nanotechnology-mediated delivery of herbal and plant-derived medicines. Advanced Pharmaceutical Bulletin 2023; 13(4), 712-722.
CGP Rumahorbo, S Ilyas, S Hutahaean and CF Zuhra. Advancing sustainable herbal medicine: Nanoparticles from medicinal plants synthesizing. In: Proceedings of the 5th Talenta Conference on Engineering, Science and Technology, Medan-North Sumatera, Indonesia. 2024, p. 3038.
L Zhang, FR Saber, G Rocchetti, G Zengin, MM Hashem and L Lucini. UHPLC-QTOF-MS based metabolomics and biological activities of different parts of Eriobotrya japonica. Food Research International 2021; 143, 110242.
M Bitew, T Desalegn, TB Demissie, A Belayneh, M Endale and R Eswaramoorthy. Pharmacokinetics and drug-likeness of antidiabetic flavonoids: Molecular docking and DFT study. PLoS One 2021; 16(12), e0260853.
FSG Silva, PJ Oliveira and MF Duarte. Oleanolic, ursolic, and betulinic acids as food supplements or pharmaceutical agents for type 2 diabetes: Promise or illusion? Journal of Agricultural and Food Chemistry 2016; 64(15), 2991-3008.
X Li, JJ Geng-Ji, YY Quan, LM Qi, Q Sun, Q Huang, HM Jiang, ZJ Sun, HM Liu and X Xie. Role of potential bioactive metabolites from traditional Chinese medicine for type 2 diabetes mellitus: An overview. Frontiers in Pharmacology 2022; 13, 1023713.
H Yi, H Peng, X Wu, X Xu, T Kuang, J Zhang, L Du and G Fan. The therapeutic effects and mechanisms of quercetin on metabolic diseases: Pharmacological data and clinical evidence. Oxidative Medicine and Cellular Longevity 2021; 2021, 6678662.
L Miao, H Zhang, MS Cheong, R Zhong, P Garcia-Oliveira, MA Prieto, KW Cheng, M Wang, H Cao, S Nie, J Simal-Gandara, WS Cheang and J Xiao. Anti-diabetic potential of apigenin, luteolin, and baicalein via partially activating PI3K/Akt/Glut-4 signaling pathways in insulin-resistant HepG2 cells. Food Science and Human Wellness 2023; 12(6), 1991-2000.
H Bouta. A comprehensive review of Moroccan medicinal plants for diabetes management. Diseases 2024; 12(10), 246.
Y Yang and X Liu. Imbalance of drug transporter-CYP450s interplay by diabetes and its clinical significance. Pharmaceutics 2020; 12(4), 348.
P Santoso, S Ilyas, YH Midoen, R Maliza and DF Belahusna. Predictive bioactivity of compounds from Vitis gracilis leaf extract to counteract doxorubicin-induced cardiotoxicity via sirtuin 1 and adenosine monophosphate-activated protein kinase: An in-silico study. 2024; 14(4), 99-114.
TM Karpiński and M Ożarowski. Plant organic acids as natural inhibitors of foodborne pathogens. Applied Sciences 2024; 14(14), 6340.
A Ali, JJ Cottrell and FR Dunshea. Antioxidant, alpha-glucosidase inhibition activities, in silico molecular docking and pharmacokinetics study of phenolic compounds from native Australian fruits and spices. Antioxidants 2023; 12(2), 254.
A Gasmi, PK Mujawdiya, S Noor, R Lysiuk, R Darmohray, S Piscopo, L Lenchyk, H Antonyak, K Dehtiarova, M Shanaida, A Polishchuk, V Shanaida, M Peana and G Bjørklund. Polyphenols in metabolic diseases. Molecules 2022; 27(19), 6280.
N Rizwana, V Agarwal and M Nune. Antioxidant for neurological diseases and neurotrauma and bioengineering approaches. Antioxidants 2022; 11(1), 72.
AL Lomize, JM Hage, K Schnitzer, K Golobokov, MB Lafaive, AC Forsyth and ID Pogozheva. PerMM: A web tool and database for analysis of passive membrane permeability and translocation pathways of bioactive molecules. Journal of Chemical Information and Modeling 2019; 59(7), 3094-3099.
D Vijh and P Gupta. GC-MS analysis, molecular docking, and pharmacokinetic studies on Dalbergia sissoo barks extracts for compounds with anti-diabetic potential. Scientific Reports 2024; 14, 24936.
T Róg, M Girych and A Bunker. Mechanistic understanding from molecular dynamics in pharmaceutical research 2: Lipid membrane in drug design. Pharmaceuticals 2021; 14(10), 1062.
C Proença, M Freitas, D Ribeiro, JLC Sousa, F Carvalho, AMS Silva, PA Fernandes and E Fernandes. Inhibition of protein tyrosine phosphatase 1B by fl avonoids: A structure - activity relationship study. Food and Chemical Toxicology 2018; 111, 474-481.
JF Zhao, LH Li, XJ Guo, HX Zhang, LL Tang, CH Ding and WS Liu. Identification of natural product inhibitors of PTP1B based on high-throughput virtual screening strategy: In silico, in vitro and in vivo studies. International Journal of Biological Macromolecules 2023; 243, 125292.
S Shanak, N Bassalat, A Barghash, S Kadan, M Ardah and H Zaid. Drug discovery of plausible lead natural compounds that target the insulin signaling pathway: Bioinformatics approaches. Evidence-based Complementary and Alternative Medicine 2022; 2022, 2832889.
HJ Jung, SH Seong, MY Ali, BS Min, HA Jung and JS Choi. α-Methyl artoflavanocoumarin from Juniperus chinensis exerts anti-diabetic effects by inhibiting PTP1B and activating the PI3K/Akt signaling pathway in insulin-resistant HepG2 cells. Archives of Pharmacal Research 2017; 40(12), 1403-1413.
SR Lin, CH Chang, MJ Tsai, H Cheng, JC Chen, MK Leong and CF Weng. The perceptions of natural compounds against dipeptidyl peptidase 4 in diabetes: From in silico to in vivo. Therapeutic Advances in Chronic Disease 2019; 10, 1-16.
M Nauck. Incretin therapies: Highlighting common features and differences in the modes of action of glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Diabetes, Obesity and Metabolism 2016; 18(3), 203-216.
Q Quranayati, M Iqhrammullah, N Saidi, N Nurliana, R Idroes and R Nasution. Extracts from Phyllanthus emblica L stem barks ameliorate blood glucose level and pancreatic and hepatic injuries in streptozotocin-induced diabetic rats. Arabian Journal of Chemistry 2023; 16(9), 105082.
MK Feleke, T Bekele, G Dessie, T Ayelgn, A Nigatu, T Jemere and AN Alene. Effect of Justicia schimperiana (Acanthaceae) roots extract on blood glucose level and lipid profiles in streptozotocin-induced diabetic mice. Metabolism Open 2024; 21, 100270.
D Satria, J Silalahi, G Haro, S Ilyas and PAZ Hsb. Antioxidant and antiproliferative activities of an ethylacetate fraction of picria Fel-terrae Lour. herbs. Asian Pacific Journal of Cancer Prevention 2017; 18(2), 399-403.
T Khouya, M Ramchoun, H Elbouny, A Hmidani, EDT Bouhlali and C Alem. Loquat (Eriobotrya japonica (Thunb) Lindl.): Evaluation of nutritional value, polyphenol composition, antidiabetic effect, and toxicity of leaf aqueous extract. Journal of Ethnopharmacology 2022; 296, 115473.
JS Bhatti, A Sehrawat, J Mishra, IS Sidhu, U Navik, N Khullar, S Kumar, GK Bhatti and PH Reddy. Oxidative stress in the pathophysiology of type 2 diabetes and related complications: Current therapeutics strategies and future perspectives. Free Radical Biology and Medicine 2022; 184, 114-134.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






