Synthesis and Bioinformatics Study of 2-Nitrocinnamaldehyde Derivatives as an Anti MCF-7 Breast Cancer Cells
DOI:
https://doi.org/10.48048/tis.2025.9586Keywords:
Anticancer, Bioinformatics, Cinnamaldehyde, Claisen-Schmidt, Docking, In-vitro assay, MCF-7Abstract
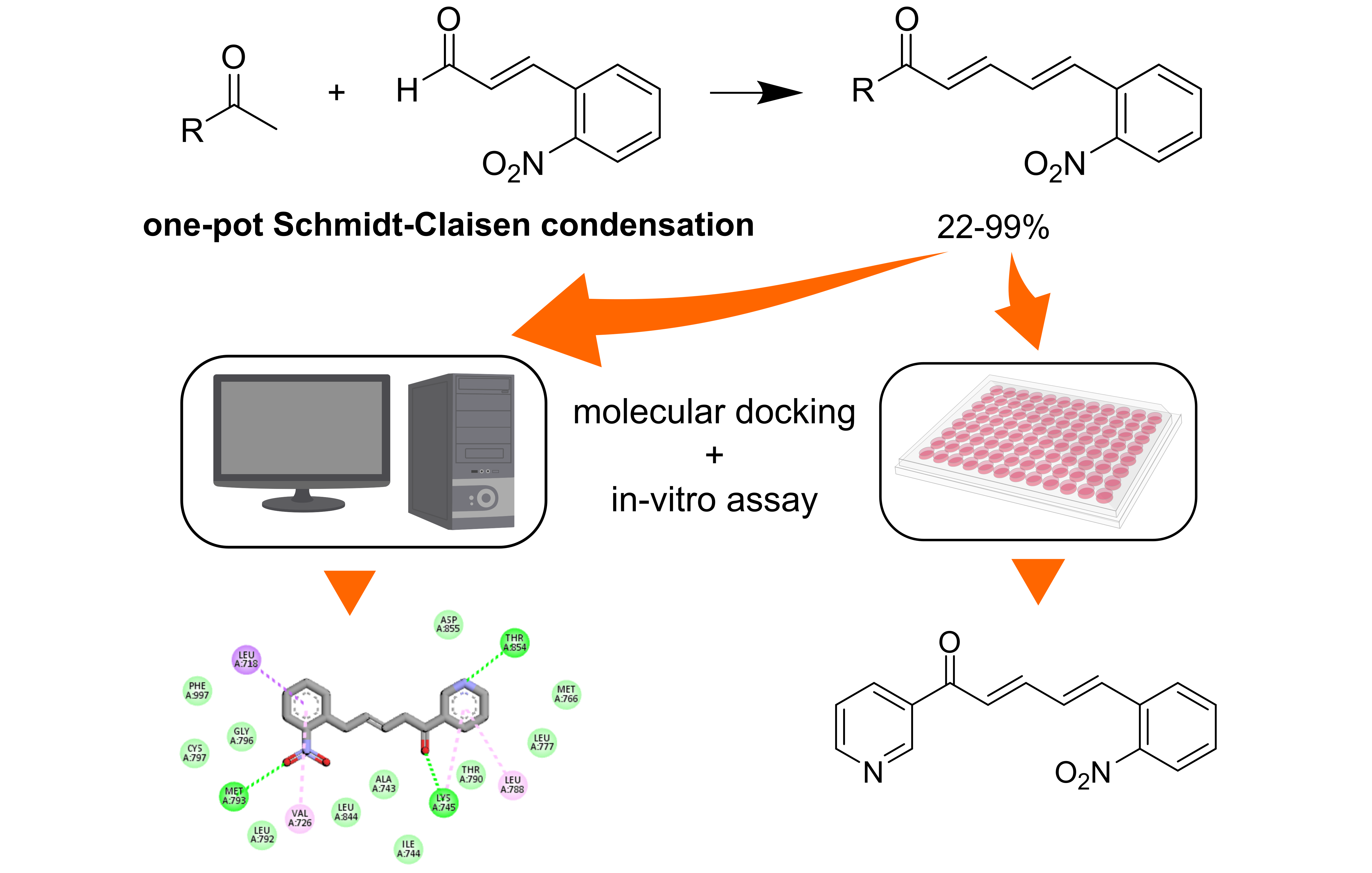
Conventional and systemic treatments of breast cancer are commonly found to expose unexpected side effects. Thus, a new drug design is intensively explored to anticipate the related risks. This work aimed to synthesize and characterize the chalcone derivatives based on a cinnamaldehyde skeleton (9 - 12). Further biological assay of the derivatives against MCF-7 breast cancer cell lines and their molecular docking analysis was also performed to obtain a more potential chemical structure for the active compounds. Four compounds of a 2-Nitrocinnamaldehyde-based scaffold, 1-(4-aminophenyl)-5-(2-nitrophenyl)-penta-2,4-dien-1-one (9), 1-(4-bromophenyl)-5-(2-nitrophenyl)-penta-2,4-dien-1-one (10), 5-(2-nitrophenyl)-1-(p-tolyl)-penta-2,4-dien-1-one (11), and 5-(2-nitrophenyl)-1-(pyridine-3-yl)-penta-2,4-dien-1-one (12) were successfully synthesized with a chemical yield of 29 - 99 %. The synthetic method employed a one-pot Claisen-Schmidt coupling reaction using a NaOH/KOH base catalyst, while their characterization was based on spectroscopic data and literature comparison. An in-vitro MTT bioassay against MCF-7 cancer cells revealed the IC50 values of the derivatives of 9, 10, 11, and 12 were 178, 376, >500, and 118.20 μg/mL, respectively. Molecular docking showed that the most potent compound in this series (12) had the lowest binding energy (–8.56 kcal/mol) and created an essential H-bond with hinge region residue, Met793. Docking-based structural optimization of 12 showed that incorporating 3 important pharmacophores commonly found in EGFR kinase inhibitors, fused heterocyclic, hydrophobic group, and propylmorpholine moiety, resulted in lower binding energy (–9.41 kcal/mol). Perhaps the modified 12 could exhibit a more potent toxicity against MCF-7.
HIGHLIGHTS
- Anti-MCF-7 breast cancer potential of new cinnamaldehyde derivatives.
- A one-pot Claisen-Schmidt coupling reaction of new chalcone derivatives.
- A bioinformatics study of a more potent structure modification.
GRAPHICAL ABSTRACT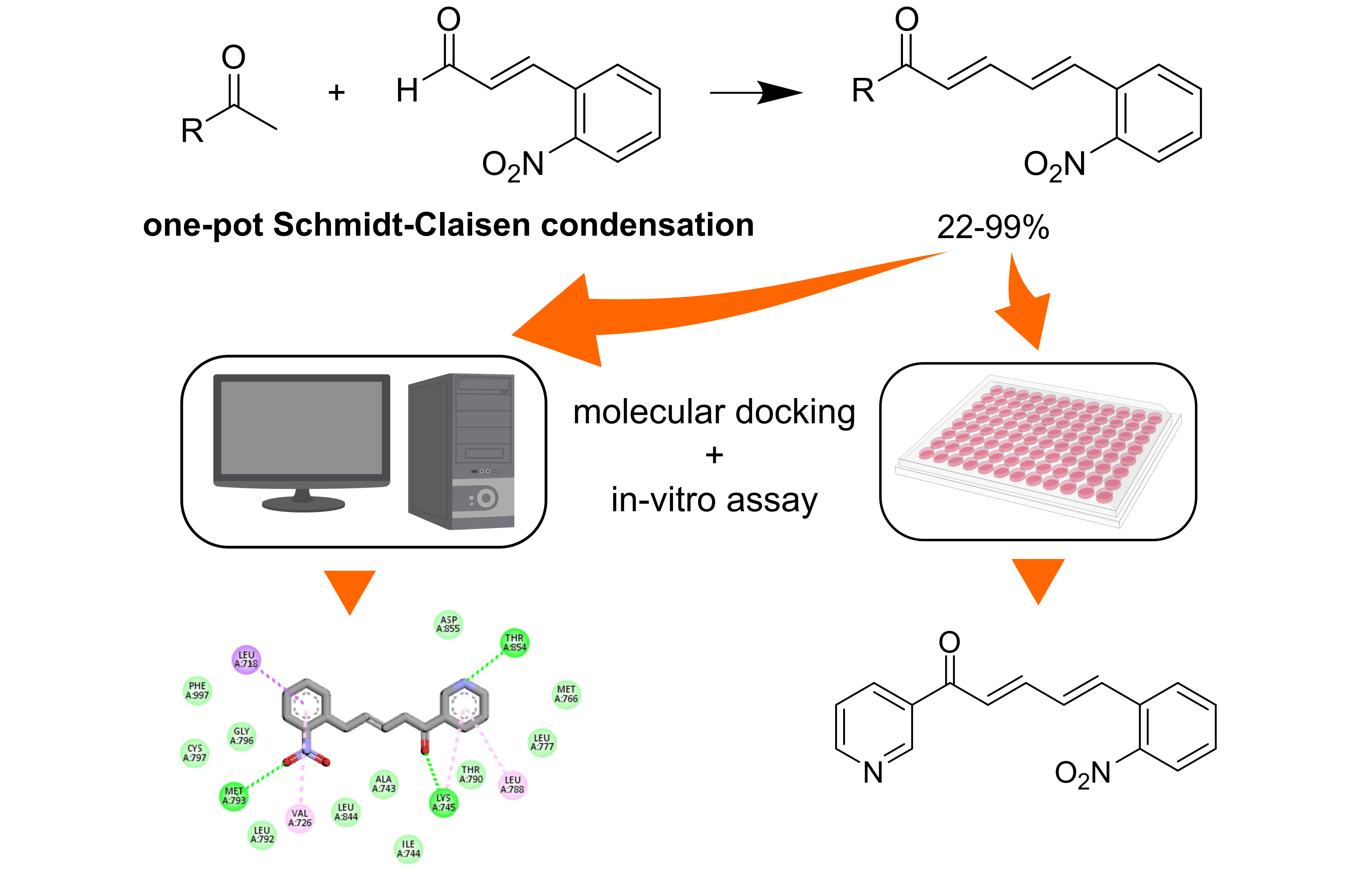
Downloads
References
J Ferlay, HR Shin, F Bray, D Forman, C Mathers and DM Parkin. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. International Journal of Cancer 2010; 127, 2893-2917.
B Costa, I Amorim, F Gärtner and N Vale. Understanding Breast cancer: From conventional therapies to repurposed drugs. European Journal of Pharmaceutical Sciences 2020; 151, 105401.
V Schirrmacher. From chemotherapy to biological therapy: A review of novel concepts to reduce the side effects of systemic cancer treatment (Review). International Journal of Oncology 2019; 54(2), 407-419.
S Haryanti and Y Widiyastuti. Cytotoxic activity of Indonesian plants on MCF-7 cell lines for traditional breast cancer treatment (in Indonesian). Media Penelitian dan Pengembangan Kesehatan 2017; 27(4), 247-254.
EB Elkaeed, HAAE Salam, A Sabt, GH Al-Ansary and WM Eldehna. Recent advancements in the development of anti-breast cancer synthetic small molecules. Molecules 2021; 26(24), 7611.
M Naufal, E Hermawati, YM Syah, AT Hidayat, IW Hidayat and J Al-Anshori. Structure-activity relationship study and design strategies of hydantoin, thiazolidinedione, and rhodanine-based kinase inhibitors: A two-decade review. ACS Omega 2024; 9, 4186-4209.
R Leechaisit, P Mahalapbutr, P Boonsri, K Karnchanapandh, T Rungrotmongkol, V Prachayasittikul, S Prachayasittikul, S Ruchirawat, V Prachayasittikul and R Pingaew. Discovery of novel naphthoquinone-chalcone hybrids as potent FGFR1 tyrosine kinase inhibitors: Synthesis, biological evaluation, and molecular modeling. ACS Omega 2023; 8(36), 32593-32605.
L Wang, W Fan, L Cui, N Yang, X Zhang, S Yu, Y Li and B Wang. Synthesis and biological activity evaluation of novel chalcone analogues containing a methylxanthine moiety and their N-acyl pyrazoline derivatives. Journal of Agricultural and Food Chemistry 2023; 71(49), 19343-19356.
K Mezgebe, Y Melaku and E Mulugeta. Synthesis and pharmacological activities of chalcone and its derivatives bearing n-heterocyclic scaffolds: A review. ACS Omega 2023; 8(22), 19194-19211.
C Zhuang, W Zhang, C Sheng, W Zhang, C Xing and Z Miao. Chalcone: A privileged structure in medicinal chemistry. Chemical Reviews 2017; 117, 7762-7810.
NAA Elkanzi, H Hrichi, RA Alolayan, W Derafa, FM Zahou and RB Bakr. Synthesis of chalcones derivatives and their biological activities: A review. ACS Omega 2022; 7(32), 27769-27786.
GD Yadav and DP Wagh. Claisen-Schmidt condensation using green catalytic processes: A critical review. ChemistrySelect 2020; 5(29), 9059-9085.
CB Patil, SK Mahajan and SA Katti. Chalcone: A versatile molecule. Journal of Pharmaceutical Sciences and Research 2009; 1(3), 11-22.
G Wang, W Liu, Z Gong, Y Huang, Y Li and Z Peng. Synthesis, biological evaluation, and molecular modelling of new naphthalene-chalcone derivatives as potential anticancer agents on MCF-7 breast cancer cells by targeting tubulin colchicine binding site. Journal of Enzyme Inhibition and Medical Chemistry 2020; 35, 139-144.
S Syam, SI Abdelwahab, MA Al-Mamary and S Mohan. Synthesis of chalcones with anticancer activities. Molecules 2012; 17(6), 6179-6195.
CW Mai, M Yaeghoobi, N Abd-Rahman, YB Kang and MR Pichika. Chalcones with electron-withdrawing and electron-donating substituents: Anticancer activity against TRAIL resistant cancer cells, structure-activity relationship analysis and regulation of apoptotic proteins. European Journal of Medical Chemistry 2014; 77, 378-387.
HO Saxena, U Faridi, JK Kumar, S Luqman, MP Darokar, K Shanker, CS Chanotiys, MM Gupta and AS Negi. Synthesis of chalcone derivatives on steroidal framework and their anticancer activities. Steroids 2007; 72(13), 892-900.
DJ Weldon, MD Saulsbury, J Goh, L Rowland, P Campbell, L Robinson, C Miller, J Christian, L Amis, N Taylor, C Dill, WJ Davis, SL Evans and E Brantley. One-pot synthesis of cinnamylideneacetophenones and their in vitro cytotoxicity in breast cancer cells. Bioorganic & Medicinal Chemistry Letters 2014; 24(15), 3381-3384.
A Zainuddin, SR Meilanie, Darwati, Kurniawan, Nurlelasari, T Herlina, AR Saputra, JA Anshori and T Mayanti. Cytotoxic triterpenoids from the stem barks of Dysoxylum arborescens and Dysoxylum excelsum against MCF-7 breast cancer cell. Sains Malaysiana 2020; 49(5), 989-994.
T Mayanti, SA Azahra, TP Syafriadi, N Nurlelasari, R Maharani, E Julaeha, U Supratman, SE Sinaga, S Ekawardhani and S Fajriah. Onoceranoid triterpenes of Lansium domesticum Corr. cv. Kokossan and their cytotoxicity against MCF-7 breast cancer cells. Trends in Sciences 2024; 22(1), 9000.
S Forli, R Huey, ME Pique, MF Sanner, DS Goodsell and AJ Olson. Computational protein-ligand docking and virtual drug screening with the AutoDock suite. Nature Protocols 2016; 11(5), 905-919.
AT Nielsen and WJ Houlihan. The aldol condensation. Wiley, New Jersey, 2011.
YS Kil, SK Choi, YS Lee, M Jafari and EK Seo. Chalcones from Angelica keiskei: Evaluation of their heat shock protein inducing activities. Journal of Natural Products 2015; 78, 2481-2487.
Y Ren, C Yuan, Y Qian, HB Chai, X Chen, M Goetz and AD Kinghorn. Constituents of an extract of Cryptocarya rubra housed in a repository with cytotoxic and glucose transport inhibitory effects. Journal of Natural Products 2014; 77(3), 550-556.
SL Gaonkar and UN Vignesh. Synthesis and pharmacological properties of chalcones: A review. Research on Chemical Intermediates 2017; 43(11), 6043-6077.
P Kulkarni. Calcium oxide catalyzed synthesis of chalcone under microwave condition. Current Microwave Chemistry 2015; 2(2), 144-149.
D Rocchi, JF González and JC Menéndez. Montmorillonite clay-promoted, solvent-free cross-aldol condensations under focused microwave irradiation. Molecules 2014; 19(6), 7317-7326.
T Liptaj, M Remko and J Polčin. Analysis of 1H-NMR spectra of cinnamaldehyde type model substances of lignin. Collection of Czechoslovak Chemical Communications 1980; 45, 330-334.
CR Polaquini, GS Torrezan, VR Santos, AC Nazaré, DL Campos, LA Almeida, IC Silva, H Ferreira, FR Pavan, C Duque and LO Regasini. Antibacterial and antitubercular activities of cinnamylideneacetophenones. Molecules 2017; 22(10), 1685.
H Zhang, H Jin, L Ji, K Tao, W Liu, HY Zhao and TP Hou. Design, synthesis, and bioactivities screening of a diaryl ketone-inspired pesticide molecular library as derived from natural products. Chemical Biology & Drug Design 2011; 78(1), 94-100.
P Prayong, S Barusrux and N Weerapreeyakul. Cytotoxic activity screening of some indigenous Thai plants. Fitoterapia 2008; 79(7-8), 598-601.
SK Liew, S Malagobadan, NM Arshad and NH Nagoor. A review of the structure-activity relationship of natural and synthetic antimetastatic compounds. Biomolecules 2020; 10(1), 138.
ML Go, X Wu and XL Liu. Chalcones: An update on cytotoxic and chemoprotective properties. Current Medical Chemistry 2005; 12(4), 483-499.
Y Shibahara, Y Miki, Y Onodera, S Hata, MSM Chan, CC Yiu, TY Loo, Y Nakamura, JI Akahira, T Ishida, K Abe, H Hirakawa, LW Chow, T Suzuki, N Ouchi and H Sasano. Aromatase inhibitor treatment of breast cancer cells increases the expression of let-7f, a microRNA targeting CYP19A1. Journal of Pathology 2012; 227(3), 357-366.
O Prakash, A Ahmad, VK Tripathi, S Tandon, AB Pant and F Khan. In silico assay development for screening of tetracyclic triterpenoids as anticancer agents against human breast cancer cell line MCF7. PLoS One 2014; 9(11), e111049.
XH Liao, DL Lu, N Wang, LY Liu, Y Wang, YQ Li, TB Yan, XG Sun, P Hu and TC Zhang. Estrogen receptor α mediates proliferation of breast cancer MCF-7 cells via a p21/PCNA/E2F1-dependent pathway. FEBS Journal 2014; 281, 927-942.
AK Shiau, D Barstad, PM Loria, L Cheng, PJ Kushner, DA Agard and GL Greene. The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell 1998; 95(7), 927-937.
S Massarweh, CK Osborne, CJ Creighton, L Qin, A Tsimelzon, S Huang, H Weiss, M Rimawi and R Schiff. Tamoxifen resistance in breast tumors is driven by growth factor receptor signaling with repression of classic estrogen receptor genomic function. Cancer Research 2008; 68(3), 826-833.
CH Yun, TJ Boggon, Y Li, MS Woo, H Greulich, M Meyerson and MJ Eck. Structures of lung cancer-derived EGFR mutants and inhibitor complexes: Mechanism of activation and insights into differential inhibitor sensitivity. Cancer Cell 2007; 11(3), 217-227.
K Aertgeerts, R Skene, J Yano, BC Sang, H Zou, G Snell, A Jennings, K Iwamoto, N Habuka, A Hirokawa, T Ishikawa, T Tanaka, H Miki, Y Ohta and S Sogabe. Structural analysis of the mechanism of inhibition and allosteric activation of the kinase domain of HER2 protein. Journal of Biological Chemistry 2011; 286(21), 18756-18765.
T Doi, H Takiuchi, A Ohtsu, N Fuse, M Goto, M Yoshida, N Dote, Y Kuze, F Jinno, M Fujimoto, T Takubo, N Nakayama and R Tsutsumi. Phase I first-in-human study of TAK-285, a novel investigational dual HER2/EGFR inhibitor, in cancer patients. British Journal of Cancer 2012; 106(4), 666-672.
T Ishikawa, M Seto, H Banno, Y Kawakita, M Oorui, T Taniguchi, Y Ohta, T Tamura, A Nakayama, H Miki, H Kamiguchi, T Tanaka, N Habuka, S Sogabe, J Yano, K Aertgeerts and K Kamiyama. Design and synthesis of novel human epidermal growth factor receptor 2 (HER2)/epidermal growth factor receptor (EGFR) dual inhibitors bearing a pyrrolo[3,2-d]pyrimidine scaffold. Journal of Medicinal Chemistry 2011; 54(23), 8030-8050.
AL Tökés, GY Litkei and L Szilágyi. N-heterocycles by cyclization of 2′-Nhr-Chalcones, 2′-Nhr-Chalcone dibromides and 2′-Nhr-α-Azidochalcones. Synthetic Communications 1992; 22, 2433-2445.
M El-Naggar, HRM Rashdan and AH Abdelmonsef. Cyclization of chalcone derivatives: Design, synthesis, in silico docking study, and biological evaluation of new quinazolin-2,4-diones incorporating five-, six-, and seven-membered ring moieties as potent antibacterial inhibitors. ACS Omega 2023; 8(30), 27216-27230.
A Voskienė and V Mickevičius. Cyclization of chalcones to isoxazole and pyrazole derivatives. Chemistry of Heterocyclic Compounds 2009; 45, 1485-1488.
S El-Kalyoubi, SA El-Sebaey, AA El-Sayed, MS Abdelhamid, F Agili and SM Elfeky. Novel pyrimidine Schiff bases and their selenium-containing nanoparticles as dual inhibitors of CDK1 and tubulin polymerase: Design, synthesis, anti-proliferative evaluation, and molecular modelling. Journal of Enzyme Inhibition and Medical Chemistry 2023; 38(1), 2232125.
S El-Kalyoubi, MM Khalifa, MT Abo-Elfadl, AA El-Sayed, A Elkamhawy, K Lee and AA Al-Karmalawy. Design and synthesis of new spirooxindole candidates and their selenium nanoparticles as potential dual Topo I/II inhibitors, DNA intercalators, and apoptotic inducers. Journal of Enzyme Inhibition and Medical Chemistry 2023; 38(1), 2242714.
ZS Alshehri, FF Alshehri, SF Belasy, EA El-Hefny, MS Aly, AA El-Sayed and NA Hassan. Biochemical and preclinical evaluation with synthesis and docking study of pyridopyrimidines and selenium nanoparticle drugs for cancer targeting. Current Nanoscience 2025; 21(6), 498-510.
LO El-Halaby, NFA El-Magd, SJ Almehmadi, AA El-Sayed, RR Khattab, S El-Kalyoubi and SM Elfeky. Synthesis, In-Vitro, In-Vivo screening, and molecular docking of disubstituted aminothiazole derivatives and their selenium nanoparticles as potential antiparkinson agents. Journal of Molecular Structure 2024; 1315, 138951.
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Walailak University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.






